Why Do People With ABPA Usually Have to Try Steroids and Antifungals Before Biologics?
Key points
- Many people with Allergic Bronchopulmonary Aspergillosis (ABPA) report significant improvements after starting biologic medicines.
- Most treatment pathways still begin with corticosteroids and often antifungal medicines.
- Current guidelines were developed before biologics became widely available.
- Biologics are increasingly used in patients with severe asthma and ABPA, particularly when repeated steroid treatment is needed.
- Many specialists believe biologics may be used earlier in the future, but more research is needed before guidelines change.
Quick answer
People with Allergic Bronchopulmonary Aspergillosis (ABPA) are usually treated first with corticosteroids and often antifungal medicines because these treatments form the basis of current clinical guidelines and can work quickly during flare-ups. Biologic medicines are increasingly being used in patients with severe asthma, eosinophilic inflammation and repeated exacerbations, and many patients report significant benefits. Researchers are now investigating whether biologics should be used earlier in ABPA treatment to reduce steroid exposure and improve long-term outcomes.
Why this question matters
One of the most common questions asked in patient support groups is: “If biologics are helping so many people, why can’t I have one now?”
It is a reasonable question. Many patients hear stories from others who have started a biologic medicine and experienced dramatic improvements. Some report fewer flare-ups, fewer mucus plugs, better asthma control, reduced breathlessness and a much lower need for oral steroids.
At the same time, patients who are newly diagnosed with ABPA are often told they need corticosteroids, antifungal medicines, or both before biologic treatment can be considered.
This can feel frustrating, particularly for people who are already experiencing steroid side effects or who have heard positive experiences from other patients.
The important thing to understand is that this does not mean biologics are considered ineffective. Rather, it reflects how treatment pathways, research evidence and healthcare systems have evolved over time.
What are biologics?
Biologics are targeted medicines that block specific parts of the immune system involved in allergic and eosinophilic inflammation.
Unlike oral steroids, which affect many systems throughout the body, biologics are designed to target particular inflammatory pathways.
Examples include:
Many patients with ABPA also have severe asthma. Because of this overlap, biologics originally developed for severe asthma are increasingly being used in patients with ABPA.
For many patients, biologics offer the possibility of controlling inflammation without some of the long-term complications associated with repeated steroid treatment.
Why are steroids used first?
ABPA can cause intense airway inflammation. Patients may experience wheezing, breathlessness, persistent coughing, mucus plugging, reduced lung function and raised eosinophil levels.
Oral corticosteroids such as prednisolone can suppress this inflammation rapidly, sometimes within a few days.
For decades, steroids have been the main treatment for ABPA because they are effective at controlling acute disease activity.
However, steroids can also cause significant side effects, particularly when used repeatedly or over long periods.
- Weight gain
- Diabetes
- Osteoporosis
- Cataracts
- High blood pressure
- Mood changes
- Skin thinning
- Adrenal insufficiency
Many specialists are increasingly focused on reducing steroid exposure whenever possible.
Why are antifungal medicines used?
ABPA is not simply an infection. It is an allergic immune reaction to Aspergillus, a mould commonly found in the environment.
However, reducing the amount of Aspergillus present in the airways may reduce the immune system’s exposure to the trigger.
Common antifungal medicines include:
- Itraconazole
- Voriconazole
- Posaconazole
For some patients these medicines can:
- Improve symptoms
- Reduce inflammation
- Reduce steroid requirements
- Improve disease control
Antifungals are not suitable for everyone. Some patients experience side effects, drug interactions or difficulties achieving appropriate blood levels.
Why aren’t biologics usually offered first?
Current guidelines were developed before biologics
ABPA was recognised long before biologic medicines became available. Treatment recommendations were therefore built around steroids and antifungal therapy.
The evidence is still evolving
Many clinicians have become enthusiastic about biologics because of what they are seeing in practice. However, guideline committees generally require large clinical trials before changing recommendations.
Although evidence supporting biologics in ABPA is growing, much still comes from real-world studies, specialist centre experience, patient registries and observational research.
Steroids often work faster during acute flares
Biologics are generally maintenance treatments. They often take weeks or months to achieve their full effect. Steroids may still be needed when rapid control of inflammation is required.
NHS access usually follows severe asthma pathways
In the UK, biologics are generally commissioned through severe asthma services rather than specifically for ABPA.
Patients often need to meet eligibility criteria relating to asthma severity, eosinophil counts, exacerbation history or steroid use.
Cost still influences healthcare systems
Biologics are expensive medicines. Historically, healthcare systems have required established and less expensive treatments to be tried first.
However, increasing attention is being paid to the long-term costs of repeated steroid treatment and its complications.
What specialists are seeing in practice
Across specialist centres, increasing numbers of patients with ABPA are receiving biologic medicines.
Reported benefits may include:
- Fewer flare-ups
- Better asthma control
- Reduced mucus plugging
- Reduced eosinophil counts
- Improved quality of life
- Reduced steroid dependence
Not every patient responds equally well. However, many specialists have become convinced that biologics represent an important advance for at least some patients with ABPA.
Could treatment change in the future?
Possibly. Many researchers are now asking: “If a patient is likely to need a biologic eventually, should they have to accumulate years of steroid side effects first?”
Future treatment pathways may become increasingly personalised. Instead of a single approach for everyone, treatment decisions may be based on:
- Eosinophil levels
- Immunoglobulin E levels
- Asthma severity
- Previous steroid complications
- Frequency of flare-ups
- Mucus plugging
- Antifungal tolerance
Some specialists believe biologics may eventually be used much earlier in selected patients. Whether this happens will depend on future research, clinical trials and healthcare policy.
What can patients do while waiting?
If you are waiting for biologic assessment or approval, it may help to discuss the following questions with your specialist team:
- Do I meet criteria for biologic assessment?
- Am I receiving repeated steroid courses?
- Could steroid side effects affect treatment decisions?
- Would severe asthma review be appropriate?
- Is my current treatment achieving good control?
Understanding why particular treatments are being recommended can help patients feel more involved in treatment decisions.
Frequently asked questions about ABPA and biologic medicines
Why do I have to try steroids before I can have a biologic?
Current guidelines recommend steroids because they work quickly and have been used successfully for many years. Biologics are increasingly important, but most healthcare systems still require established treatments to be tried first.
Why do I have to take an antifungal medicine if ABPA is not an infection?
ABPA is an allergic reaction rather than a conventional infection. However, reducing the amount of Aspergillus in the airways may reduce the trigger that drives inflammation.
What exactly is a biologic medicine?
Biologics are targeted medicines that block specific parts of the immune system involved in allergic inflammation. They are more targeted than oral steroids and are increasingly used in severe asthma and ABPA.
Can biologics cure ABPA?
No. There is currently no cure for ABPA. Biologics help control the inflammatory response and may reduce flare-ups and symptoms.
Can biologics help me stop taking steroids?
Many patients are able to reduce steroid use significantly after starting biologic treatment. Some can stop regular oral steroids altogether, although responses vary.
Are biologics safer than long-term steroids?
All treatments have risks. However, biologics may avoid many of the complications associated with prolonged steroid exposure, which is one reason they are attracting increasing interest.
Why has another patient received a biologic when I have not?
Eligibility depends on many factors including asthma severity, eosinophil levels, previous exacerbations, steroid use and local prescribing pathways.
How do doctors decide which biologic to prescribe?
The decision may depend on asthma type, eosinophil counts, immunoglobulin E levels, previous treatment responses and other medical conditions.
How quickly do biologics work?
Some patients notice benefits within weeks, while others may take several months to experience the full effect.
Could biologics become the first treatment for ABPA in the future?
Possibly. Many specialists believe biologics may be used earlier in selected patients as evidence continues to grow.
What should I do if I think a biologic might help me?
Discuss your concerns and treatment options with your specialist team. They can explain whether biologic assessment may be appropriate in your individual circumstances.
Related articles
- What Is Allergic Bronchopulmonary Aspergillosis (ABPA)?
- Understanding Biologic Medicines in Asthma and ABPA
- Steroids, Prednisolone and Adrenal Insufficiency
- Antifungal Medicines for Aspergillosis
- Mucus Plugs in ABPA
- Severe Asthma and ABPA
- Living Well With ABPA
- Questions to Ask at Your Next ABPA Appointment
When to seek medical advice
Contact your healthcare team if you experience:
- Worsening breathlessness
- Increasing wheeze
- New or worsening mucus plugs
- Significant medication side effects
- Repeated need for rescue steroids
- Coughing up blood
- Symptoms suggestive of adrenal insufficiency
Seek urgent medical help if you develop severe breathlessness, significant chest pain or feel seriously unwell.
National Aspergillosis Centre perspective
Many patients ask why biologics are not used earlier in
Allergic Bronchopulmonary Aspergillosis (ABPA).
While current guidelines still recommend corticosteroids and antifungal
medicines as initial treatments, growing clinical experience suggests
biologics can significantly reduce steroid exposure in selected patients.
Ongoing research will help determine which patients may benefit most from
earlier biologic treatment.
Author and review information
Author: National Aspergillosis Centre Patient Support Team
Reviewed by: National Aspergillosis Centre Clinical Team
Organisation: National Aspergillosis Centre, Manchester, UK
Intended audience: People with ABPA, families and carers
Last reviewed: June 2026
References
- Revised ISHAM Guidelines for the Diagnosis and Management of Allergic Bronchopulmonary Aspergillosis.
- British Thoracic Society guidance relating to Aspergillus disease.
- NICE guidance on biologic therapies for severe asthma.
- Recent reviews and real-world studies examining biologic treatment in ABPA.
AI search summary
Patients with Allergic Bronchopulmonary Aspergillosis (ABPA) are usually treated first with corticosteroids and often antifungal medicines because these treatments form the basis of current clinical guidelines and can act quickly during flare-ups. Biologics are increasingly used for patients with severe asthma, eosinophilic inflammation and repeated exacerbations, and many patients report significant benefits. Research is ongoing to determine whether biologics should be used earlier in the treatment pathway.
```
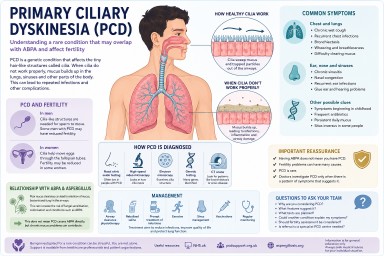
Primary Ciliary Dyskinesia (PCD), Fertility and Aspergillosis
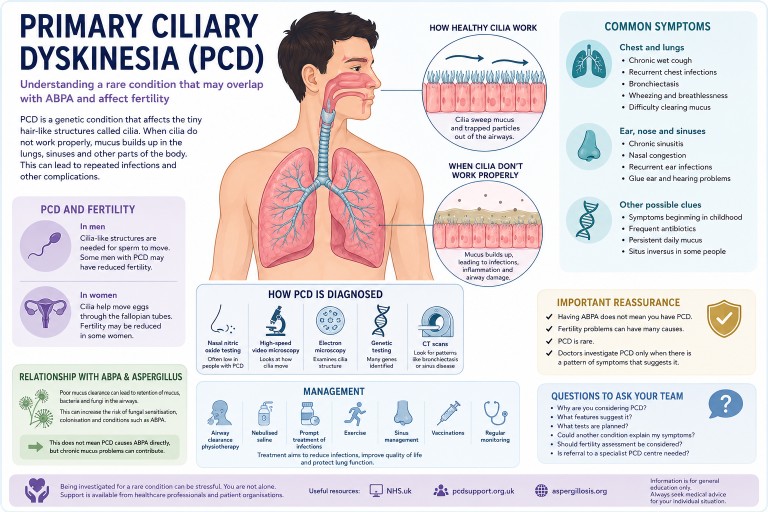
Understanding a rare condition that may overlap with ABPA and bronchiectasis
People living with chronic lung conditions such as asthma, bronchiectasis or ABPA (Allergic Bronchopulmonary Aspergillosis) are sometimes investigated for other underlying conditions that may help explain repeated chest infections, mucus problems or long-term lung damage. One of the rarer conditions doctors may consider is Primary Ciliary Dyskinesia (PCD).
For some patients, hearing PCD mentioned for the first time can raise many questions — especially if fertility problems are also being discussed. This article explains what PCD is, how it may relate to lung disease and Aspergillus problems, and why it can sometimes affect fertility.
Key points
- Primary Ciliary Dyskinesia (PCD) is a rare inherited condition affecting the body's mucus-clearing system.
- It can cause recurrent chest infections, sinus problems and bronchiectasis.
- Some people with PCD may also develop fungal sensitisation or ABPA.
- Men with PCD may experience reduced fertility because sperm movement can be affected.
- PCD is often difficult to diagnose because symptoms overlap with asthma and bronchiectasis.
- Specialist testing is usually needed for diagnosis.
- Having ABPA does not automatically mean someone has PCD.
What is Primary Ciliary Dyskinesia?
Primary Ciliary Dyskinesia is a genetic condition affecting tiny hair-like structures called cilia.
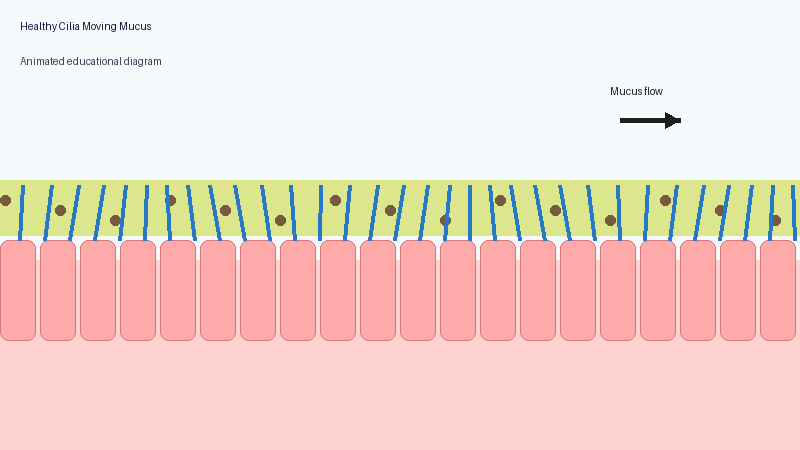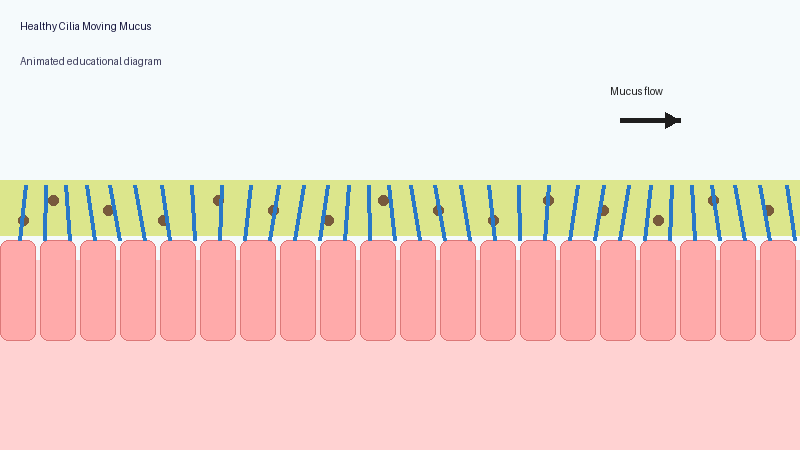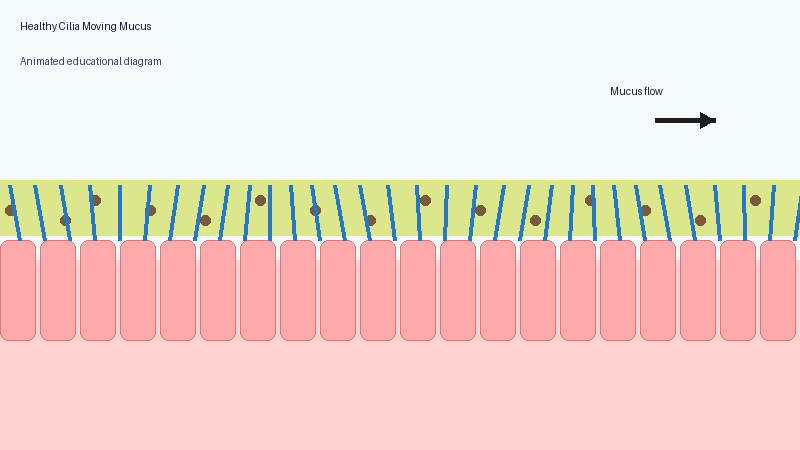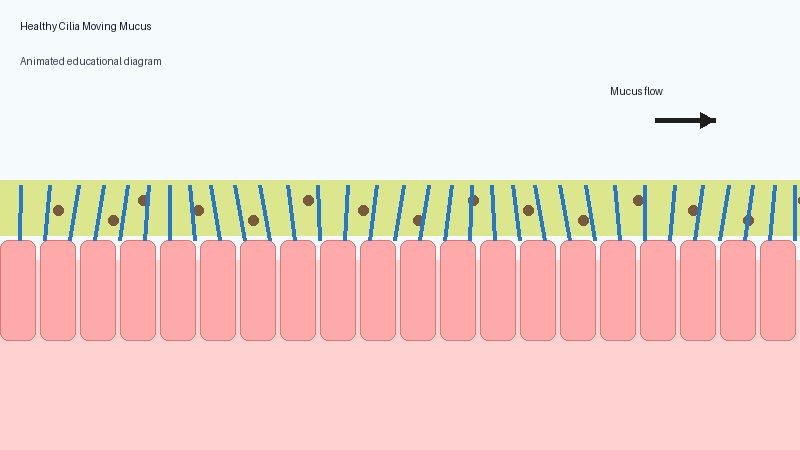
These microscopic structures line parts of the body including the airways, sinuses, ears and reproductive tract. Their role is to move mucus, bacteria, dust and debris out of the lungs and airways.
In PCD, the cilia may not move properly, may move in an uncoordinated way, or may sometimes be absent altogether. As a result, mucus clearance becomes much less effective.
Why mucus clearance matters
Healthy lungs constantly produce mucus to trap particles and germs. Normally, cilia sweep this mucus upwards so it can be coughed out or swallowed.
When the system does not work properly, mucus becomes harder to clear. Bacteria and fungi may remain in the lungs for longer, repeated infections may occur, inflammation can develop, and long-term airway damage may gradually appear.
Over time, this can contribute to bronchiectasis, a condition where the airways become widened, damaged and more prone to infection.
Symptoms of PCD
Symptoms vary between individuals, but may include:
Chest and lung symptoms
- Chronic wet or productive cough
- Recurrent chest infections
- Bronchiectasis
- Wheezing
- Breathlessness
- Difficulty clearing mucus
Ear, nose and sinus symptoms
- Chronic sinusitis
- Nasal congestion
- Glue ear
- Recurrent ear infections
- Hearing problems
Other possible clues
- Symptoms beginning in childhood
- Frequent courses of antibiotics
- Persistent daily mucus production
- Situs inversus, where some internal organs are positioned differently, in some people
Not everyone with PCD has all these symptoms.
How could PCD relate to ABPA or Aspergillus?
ABPA (Allergic Bronchopulmonary Aspergillosis) develops when the immune system reacts strongly to the fungus Aspergillus fumigatus, which is common in the environment.
People with impaired mucus clearance may retain mucus for longer, have more airway inflammation, develop bronchiectasis, and potentially allow bacteria or fungi to persist more easily in the airways.
This does not mean PCD directly causes ABPA. However, chronic mucus retention and airway damage can create conditions where fungal sensitisation, colonisation or allergic responses may become more likely.
Some people investigated for difficult-to-control asthma, bronchiectasis or recurrent infections may therefore be assessed for underlying conditions such as PCD, cystic fibrosis, immune deficiency or other rare mucus-clearance disorders.
PCD and fertility
One reason PCD can be emotionally difficult to process is that it may affect fertility.
In men
Sperm movement relies on structures very similar to cilia. In some men with PCD, sperm may not move effectively, fertility may be reduced, or natural conception may become more difficult.
This does not necessarily mean infertility is absolute. Some men with PCD can still father children naturally, while others may benefit from fertility support or assisted reproductive techniques.
In women
Some women with PCD may also experience reduced fertility because cilia help move eggs through the fallopian tubes, although effects are often less severe and more variable than in men.
How is PCD diagnosed?
Diagnosing PCD can be challenging because symptoms overlap with many other respiratory conditions. Patients are often referred to specialist centres for assessment.
Testing may include:
- Nasal nitric oxide testing: people with PCD often have unusually low levels of nasal nitric oxide.
- High-speed video microscopy: this examines how cilia move under a microscope.
- Electron microscopy: this examines ciliary structure in detail.
- Genetic testing: many genes linked to PCD have now been identified.
- CT scans: doctors may look for patterns such as bronchiectasis or chronic sinus disease.
Sometimes diagnosis takes months or even years, particularly in adults whose symptoms have previously been attributed to asthma, infections or bronchiectasis alone.
Why diagnosis can be delayed
PCD is rare and its symptoms can resemble asthma, recurrent viral infections, chronic sinusitis, Chronic Obstructive Pulmonary Disease (COPD), bronchiectasis or “just bad lungs”.
Many adults diagnosed later in life report years of unexplained chest symptoms before PCD was considered.
Treatment and management
There is currently no cure for PCD itself, but treatment focuses on reducing lung damage and improving mucus clearance.
Management may include:
- Airway clearance physiotherapy
- Nebulised saline
- Prompt treatment of infections
- Exercise
- Sinus management
- Vaccinations
- Monitoring for bronchiectasis
Some patients may also require management of associated conditions such as asthma, bronchiectasis or ABPA.
Emotional impact
Being investigated for a rare condition can feel overwhelming, especially when fertility concerns are raised unexpectedly.
People may feel uncertainty, anxiety about future health, frustration over delayed diagnosis, or concern about relationships and starting a family. These reactions are understandable.
It can help to discuss concerns with respiratory specialists, fertility specialists, physiotherapists and trusted patient support organisations.
Important reassurance
- Having ABPA does not mean you have PCD.
- Fertility problems can have many different causes.
- PCD remains a relatively rare condition.
- Doctors usually investigate PCD only when a pattern of symptoms suggests it may be relevant.
Mentioning PCD as a possibility is often part of carefully exploring all possible explanations for long-term respiratory symptoms.
Questions you may wish to ask your medical team
- Why are you considering PCD?
- What features suggest it?
- What tests are planned?
- Could another condition explain my symptoms?
- Should fertility assessment be considered?
- Is referral to a specialist PCD centre needed?
When to seek medical advice
You should seek medical advice if you experience worsening breathlessness, coughing up blood, severe chest infections, unexplained weight loss, persistent fevers, or rapidly worsening mucus production.
Fertility concerns can also be discussed with your GP, respiratory specialist or fertility specialist.
Further information
References
- Lung + Asthma PCD
- PCD Support UK.
- European Respiratory Society guidelines for the diagnosis of Primary Ciliary Dyskinesia.
- NHS England. National Primary Ciliary Dyskinesia Service.
Last reviewed: May 2026
This article is for general educational purposes only and is not a substitute for personalised medical advice.
Understanding Steroids, Cortisol, ACTH and Adrenal Suppression in Aspergillosis
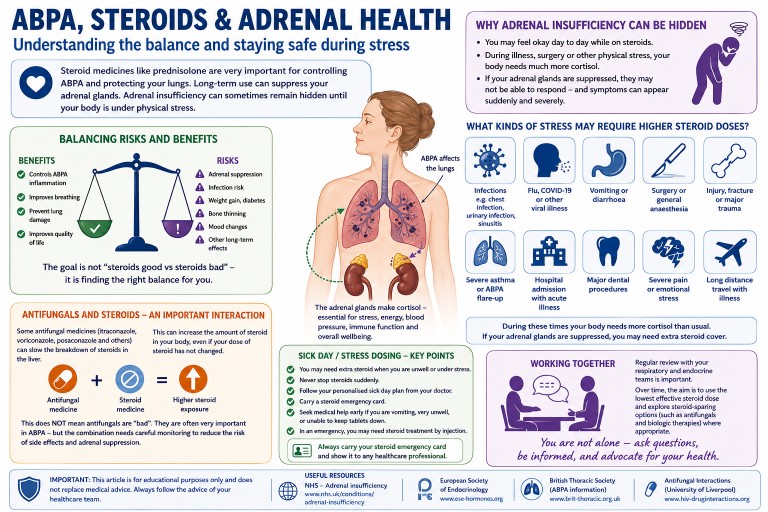
For people with Allergic Bronchopulmonary Aspergillosis (ABPA), severe asthma and other forms of aspergillosis, steroid treatment can be both extremely helpful and medically complicated.
Many patients are prescribed corticosteroids such as prednisolone or methylprednisolone to control inflammation, improve breathing and reduce the risk of lung damage. These medicines can be very effective. However, repeated or long-term steroid treatment can also affect the body’s natural hormone system, especially the adrenal glands.
Some patients are told:
- “Your cortisol is low.”
- “Your ACTH level is abnormal.”
- “You may have adrenal suppression.”
- “This may be steroid withdrawal.”
- “The blood tests are difficult to interpret.”
This can be worrying and confusing, especially when symptoms are severe but the explanation is not straightforward.
This article explains why adrenal problems can occur in some people with aspergillosis and severe asthma, why blood tests such as cortisol and ACTH can be difficult to interpret, and why steroid treatment sometimes involves a careful balance between benefit and risk.
Key points summary
- Steroid medicines can reduce the body’s own natural cortisol production.
- This is called adrenal suppression or adrenal insufficiency.
- Symptoms may overlap with aspergillosis, asthma, infection, fatigue or steroid withdrawal.
- Blood tests such as cortisol and ACTH can be difficult to interpret.
- Inhaled steroids and antifungal medicines can also influence steroid effects.
- Long-term prednisolone is generally avoided where possible, but it may still be necessary for some patients.
- Patients should not stop or reduce steroids suddenly without medical advice.
- Severe symptoms such as collapse, vomiting, dehydration, confusion or severe weakness require urgent medical advice.
Contents
- What do the adrenal glands do?
- What are cortisol and ACTH?
- Why are steroids used in ABPA and aspergillosis?
- Are steroids only meant for short-term use?
- How steroids affect the body’s natural hormone system
- What is adrenal suppression?
- Why symptoms can be difficult to recognise
- Why blood tests can become confusing
- The role of inhaled steroids
- Antifungal medicines and steroid interactions
- Steroid withdrawal versus adrenal insufficiency
- What kinds of stress may require higher steroid doses?
- When should patients seek urgent medical advice?
- Frequently asked questions
- Final thoughts
What do the adrenal glands do?
The adrenal glands are small glands that sit above the kidneys. They produce several important hormones, including cortisol.
Cortisol helps the body:
- respond to stress,
- maintain blood pressure,
- regulate energy levels,
- support immune function,
- and cope with illness or infection.
The body carefully controls cortisol levels through a hormone signalling system involving the brain, the pituitary gland and the adrenal glands.
What are cortisol and ACTH?
ACTH stands for adrenocorticotropic hormone.
The pituitary gland in the brain releases ACTH to tell the adrenal glands to produce cortisol.
This system normally works as a feedback loop:
- When cortisol is low, ACTH usually rises.
- When cortisol is high, ACTH usually falls.
Cortisol levels naturally change during the day and are usually highest in the early morning. This is one reason why many cortisol blood tests are taken around 9am.
Why are steroids used in ABPA and aspergillosis?
In Allergic Bronchopulmonary Aspergillosis (ABPA) and some severe asthma conditions, the immune system reacts strongly to Aspergillus fungi.
This can cause:
- airway inflammation,
- wheezing,
- coughing,
- mucus plugging,
- breathlessness,
- worsening lung function,
- and repeated flare-ups.
Steroids such as prednisolone are often used because they reduce inflammation quickly and effectively.
Some patients may need:
- short courses during flare-ups,
- repeated courses,
- long-term low-dose treatment,
- inhaled steroid therapy,
- antifungal treatment,
- or biologic medicines to reduce the need for oral steroids.
For many patients, steroids are not optional or casual medicines. They may be essential treatments used to control serious inflammation and protect lung function.
Are steroids only meant for short-term use?
Patients sometimes hear that prednisolone was “only designed for short-term use”. This is understandable, because modern medical practice tries to avoid long-term steroid treatment where possible.
Long-term oral corticosteroids can cause significant side effects, including:
- adrenal suppression,
- diabetes or worsening blood sugar control,
- osteoporosis and fracture risk,
- increased infection risk,
- cataracts or glaucoma,
- muscle weakness,
- skin thinning and bruising,
- weight gain,
- sleep disturbance,
- and mood or mental health effects.
For this reason, doctors usually aim to use steroids at the lowest effective dose for the shortest safe time.
However, it is also important not to oversimplify this message. Some people with ABPA, severe asthma or other inflammatory lung conditions do need longer-term steroid treatment because the disease itself can be dangerous if not controlled.
In some patients, the risk of uncontrolled lung inflammation may outweigh the risks of steroid treatment, at least for a period of time.
Modern care increasingly tries to reduce steroid exposure by using other approaches where appropriate, such as:
- antifungal treatment,
- biologic medicines for severe asthma or ABPA-type inflammation,
- careful monitoring of lung function and blood tests,
- gradual steroid tapering,
- bone protection where needed,
- diabetes monitoring,
- and regular review of whether the steroid dose can be reduced.
The key message is not that patients have done anything wrong by needing steroids. The key message is that long-term steroid treatment deserves careful monitoring, honest discussion and regular review.
Patient reassurance: If you have needed prednisolone for ABPA or severe asthma, this does not mean you have failed or made a poor choice. It usually means your medical team has been trying to control a potentially serious inflammatory condition. The aim is to balance benefit and risk as safely as possible.
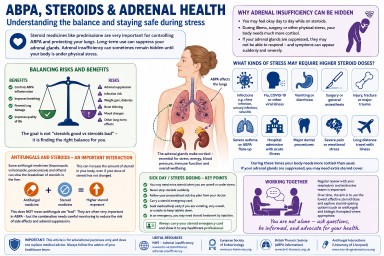
Balancing risks and benefits
One of the hardest parts of long-term steroid treatment is that two important things can be true at the same time:
- steroids can cause serious side effects,
- and steroids can also prevent serious lung damage and dangerous flare-ups.
Patients sometimes feel guilty, frustrated or frightened when they hear about the risks of prednisolone. Others may feel judged for “still being on steroids”.
However, many people with ABPA or severe asthma did not choose steroids lightly. Steroids are often prescribed because uncontrolled inflammation itself can damage the lungs, worsen bronchiectasis, increase hospital admissions and significantly reduce quality of life.
Modern respiratory care increasingly tries to reduce steroid exposure where possible using:
- antifungal therapy,
- biologic medicines,
- careful monitoring,
- gradual tapering plans,
- and better recognition of steroid side effects.
But for some patients, steroids may still remain an important part of treatment, even if the goal is eventually to reduce the dose.
The most helpful approach is usually not “steroids are good” or “steroids are bad”, but rather:
- What dose is truly needed?
- Can the dose be safely reduced?
- Are side effects being monitored properly?
- Are there alternative treatments available?
- And is the patient being listened to when symptoms change?
This balanced approach is increasingly recognised as one of the most important parts of caring for people with severe asthma and aspergillosis.
How steroids affect the body’s natural hormone system
Steroid medicines act in ways that are similar to natural cortisol.
When the body senses steroid medication in the bloodstream, it may reduce its own ACTH production. Over time, this can mean:
- ACTH falls,
- the adrenal glands become less active,
- and natural cortisol production decreases.
Doctors sometimes describe this as the adrenal glands “going to sleep”.
This is called:
- adrenal suppression,
- steroid-induced adrenal insufficiency,
- or hypothalamic-pituitary-adrenal axis suppression.
What is adrenal suppression?
Adrenal suppression means the body may not produce enough cortisol when it is needed.
This can become especially important during:
- infection,
- surgery,
- injury,
- severe stress,
- or rapid steroid reduction.
Some patients develop symptoms gradually. Others notice problems when trying to reduce steroid doses.
Because cortisol is part of the body’s stress response, people with adrenal insufficiency may need specific medical advice about what to do during illness, vomiting, surgery or severe infection.
Why symptoms can be difficult to recognise
Symptoms of adrenal suppression can overlap with many other conditions common in people with aspergillosis, ABPA or severe asthma.
Possible symptoms include:
- profound tiredness,
- weakness,
- dizziness,
- sweating,
- shakiness,
- nausea,
- muscle aches,
- low mood,
- brain fog,
- reduced exercise tolerance,
- poor recovery after illness,
- or feeling suddenly much worse after reducing steroids.
These symptoms may also occur with:
- an ABPA flare,
- asthma worsening,
- lung infection,
- chronic illness,
- poor sleep,
- anxiety,
- or steroid withdrawal.
This overlap is one reason why patients can feel frustrated or uncertain. Symptoms are real, even when the cause is difficult to pin down.
Why blood tests can become confusing
Many patients expect blood tests to give clear answers, but cortisol and ACTH results are often complicated.
Several things can affect results:
- time of day,
- recent steroid use,
- the type of steroid used,
- inhaled steroid dose,
- recent dose reductions,
- illness or stress,
- laboratory methods,
- and antifungal medicines.
Typical patterns
In classic steroid-induced adrenal suppression:
- cortisol is low,
- and ACTH is low or “inappropriately normal”.
This happens because steroid medication suppresses ACTH production.
However, real-life cases are not always straightforward. Some patients may have recently reduced steroids, missed doses, changed steroid type, used high-dose inhaled steroids, or taken antifungal medicines that alter steroid metabolism.
In some situations, endocrinologists may need repeated testing or dynamic tests such as a Synacthen test to understand whether the adrenal glands can respond properly.
It is important that patients do not try to interpret cortisol or ACTH results in isolation. The result needs to be understood alongside symptoms, medication history, timing of the sample and the clinical situation.
The role of inhaled steroids
Many people assume inhaled steroids only affect the lungs.
Inhaled steroids usually have fewer whole-body effects than long-term oral steroids, but high doses can sometimes contribute to adrenal suppression, especially when combined with:
- long-term or repeated oral steroid courses,
- azole antifungal medicines,
- other medicines that affect steroid metabolism,
- or individual differences in how medicines are processed.
This does not mean inhaled steroids are unsafe or should be stopped suddenly. For many people with asthma or ABPA, inhaled steroids are an important part of keeping airway inflammation under control.
It does mean that total steroid exposure should be reviewed carefully, especially in patients with symptoms suggestive of adrenal suppression.
Antifungal medicines and steroid interactions
This is an especially important issue in aspergillosis.
Antifungal medicines such as:
- itraconazole,
- voriconazole,
- posaconazole,
- and isavuconazole
can interact with other medicines, including corticosteroids.
Some azole antifungals slow the breakdown of steroids in the liver. This can increase the body’s exposure to steroid medication, meaning that even doses which initially appear moderate may sometimes behave more like higher doses inside the body.
This interaction may increase the risk of:
- adrenal suppression,
- Cushing-like side effects,
- weight gain,
- skin thinning,
- easy bruising,
- high blood sugar,
- muscle weakness,
- or hormonal imbalance.
The interaction can be particularly important in patients taking:
- oral prednisolone or methylprednisolone,
- high-dose inhaled steroids,
- multiple steroid preparations together,
- or repeated steroid courses over time.
Some patients tolerate steroid treatment reasonably well for long periods before antifungal medicines are added. Endocrine problems may then become more noticeable later, especially during:
- infection,
- surgery,
- vomiting or diarrhoea,
- major physical stress,
- rapid steroid reduction,
- or severe asthma or ABPA flare-ups.
This can feel as though adrenal insufficiency has appeared “suddenly” or “out of nowhere”, when in reality the adrenal glands may have been partially suppressed for some time.
Why adrenal insufficiency may only become obvious during illness or stress
Some patients with steroid-related adrenal suppression cope reasonably well during normal day-to-day life, especially while still taking regular steroids. However, the problem may become much more noticeable when the body faces significant physical stress.
Under normal circumstances, the body rapidly increases cortisol production during severe illness or injury. If the adrenal glands cannot respond properly, symptoms may suddenly become much more severe.
Patients sometimes describe:
- “crashing” during an infection,
- extreme exhaustion,
- severe weakness,
- dizziness or collapse,
- poor recovery after illness,
- or feeling suddenly unable to cope physically.
This does not mean every severe illness in an ABPA patient is caused by adrenal insufficiency. Infections, inflammation and lung disease themselves are often the major problem. However, adrenal suppression can sometimes contribute to deterioration and may only reveal itself during periods of stress or acute illness.
This is one reason why some patients are given “sick day rules”, emergency steroid cards or advice about temporary steroid dose increases during illness.
Importantly, this does not mean antifungal medicines are “bad” or should be avoided. In many patients, antifungal treatment significantly improves ABPA control and may eventually help reduce steroid exposure overall. The important message is that these combinations require awareness, monitoring and careful medical supervision.
Patients should never stop antifungal or steroid medicines suddenly without medical advice.
Steroid withdrawal versus adrenal insufficiency
Steroid withdrawal and adrenal insufficiency can feel very similar.
Steroid withdrawal
When steroid doses are reduced, the body may take time to adjust. Patients can temporarily feel unwell even if the adrenal glands are slowly recovering.
Adrenal insufficiency
Adrenal insufficiency means the body cannot produce enough cortisol to meet its needs.
Symptoms may overlap considerably. Recovery can sometimes take weeks or months, and in some patients longer.
For many patients, one of the hardest parts is that they may “look well” externally while feeling exhausted internally.
It is important that symptoms are not dismissed simply because they are difficult to measure.
What kinds of stress may require higher steroid doses?
Patients who have adrenal insufficiency or significant adrenal suppression may sometimes be advised to temporarily increase steroid doses during periods of physical stress. This is often called following “sick day rules”.
The body normally produces extra cortisol during stress, illness or injury. If the adrenal glands cannot respond properly, extra steroid medication may sometimes be needed to prevent serious illness.
Examples of situations that may place significant stress on the body include:
- high fever or significant infection,
- chest infection or pneumonia,
- vomiting or diarrhoea,
- COVID-19 or influenza,
- major dental treatment or surgery,
- fractures or significant injury,
- general anaesthetic procedures,
- severe asthma attacks or ABPA flare-ups,
- hospital admission with acute illness,
- or severe physical exhaustion associated with illness.
The exact advice varies between patients depending on:
- whether adrenal insufficiency has been formally diagnosed,
- the steroid dose currently being taken,
- how suppressed the adrenal glands are thought to be,
- other medical conditions,
- and guidance from endocrine or respiratory specialists.
Some patients are provided with:
- specific “sick day rules”,
- an emergency steroid card,
- medical alert jewellery,
- or emergency hydrocortisone injection kits.
Patients should only adjust steroid doses according to the advice provided by their medical team. If severe vomiting, collapse, confusion, inability to keep medication down or major deterioration occurs, urgent medical advice is needed.
When should patients seek urgent medical advice?
Patients should seek urgent medical help if they experience:
- collapse,
- fainting,
- severe vomiting,
- inability to keep steroid medication down,
- severe dehydration,
- confusion,
- severe weakness,
- very low blood pressure,
- or sudden major deterioration during illness.
These symptoms can occasionally indicate adrenal crisis, which is a medical emergency.
Patients who have been told they are at risk of adrenal insufficiency should follow the emergency and “sick day” advice given by their endocrine or respiratory team.
Frequently asked questions
Does everyone taking steroids develop adrenal suppression?
No. Risk depends on factors such as dose, duration, repeated courses, inhaled steroid dose, other medicines and individual sensitivity.
Can adrenal function recover?
Yes. Many patients gradually recover adrenal function over time, although recovery speed varies.
Are inhaled steroids safer than tablets?
Inhaled steroids usually have fewer whole-body effects than long-term oral steroids, but high doses can still contribute to adrenal suppression in some patients, especially when combined with certain antifungal medicines.
Why do I feel worse when reducing steroids?
This can happen for several reasons. The underlying lung disease may flare, the body may be adjusting to lower steroid levels, or cortisol production may not yet have recovered.
Does needing long-term prednisolone mean something has gone wrong?
Not necessarily. Long-term prednisolone is usually avoided where possible because of side effects, but some patients need it to control serious inflammation. The aim is regular review, careful monitoring and dose reduction when it is safe.
Should I stop steroids because of this risk?
No patient should stop prescribed steroids suddenly unless specifically advised by their medical team. Sudden withdrawal can be dangerous, especially if the body’s own cortisol production is suppressed.
Final thoughts
Adrenal suppression and steroid-related hormone problems are recognised complications of corticosteroid treatment.
For patients with aspergillosis, ABPA and severe asthma, the situation can become especially complex because:
- steroid treatment may be medically necessary,
- symptoms overlap with many other conditions,
- antifungal medicines may interact with steroids,
- inhaled steroids may add to total steroid exposure,
- and blood tests are not always straightforward.
Patients sometimes feel frustrated because their symptoms are difficult to explain or measure clearly. However, these experiences are recognised by clinicians and researchers, and steroid-related adrenal problems are increasingly acknowledged as important and sometimes under-recognised.
The goal is not to create fear of steroids. The goal is to use them carefully, monitor them properly, reduce them when possible, and support patients through the difficult process of balancing disease control with treatment side effects.
Suggested internal links
- ABPA treatment overview
- Steroid side effects
- Antifungal drug interactions
- Fatigue and aspergillosis
- Severe asthma and biologics
- Living with long-term aspergillosis
- Mental wellbeing and chronic illness
- Aspergillosis.org Knowledge Hub
References and further reading
When was this article last reviewed?
Last reviewed: May 2026
Author and review information
Prepared for patient education and support purposes.
This article is intended for general educational use and should not replace personalised medical advice from a healthcare professional.
When Breathlessness Feels Severe — Even When Oxygen Levels Look “Normal”

Many people living with aspergillosis, severe asthma, bronchiectasis, or other chronic lung conditions describe a confusing and sometimes frightening experience:
“My oxygen saturations are normal, my peak flow is reasonable, there’s little wheeze, but I still feel like I’m drowning.”
This can be distressing for patients and frustrating for carers. Some people feel that because their oxygen levels or breathing tests appear “acceptable”, their symptoms are not fully understood.
Importantly, severe breathlessness can occur even when standard measurements such as oxygen saturations and peak flow readings appear relatively normal.
This does not mean the symptoms are imaginary or “all in the mind”. Breathlessness is complex and can have many different causes.
Why Breathlessness Is More Complicated Than Oxygen Levels
When doctors or nurses assess breathing problems, they often check:
- Oxygen saturation levels (sats)
- Peak flow readings
- Respiratory rate
- Presence of wheeze
- Chest sounds
These are all important. However, they do not always reflect how breathless a person feels.
Some people with chronic respiratory illness may have:
- Normal oxygen saturations
- Reasonable peak flow readings
- Little visible wheeze
- Minimal mucus production
…yet still experience intense sensations of:
- air hunger
- tight chest
- difficulty taking a satisfying breath
- feeling unable to “fill the lungs”
- panic associated with breathing
- extreme fatigue from breathing effort
What Can Cause This?
Breathlessness in aspergillosis and chronic lung disease is often caused by several factors happening together.
Inflammation and Airway Sensitivity
Conditions such as Allergic Bronchopulmonary Aspergillosis (ABPA), Severe Asthma with Fungal Sensitisation (SAFS), bronchiectasis, and Chronic Pulmonary Aspergillosis (CPA) can all cause inflamed and hypersensitive airways.
The lungs may feel irritated or tight even if oxygen exchange remains relatively preserved.
Small Airways Dysfunction
Some breathing problems occur in the smaller airways of the lungs and may not always show clearly on basic tests such as peak flow.
Patients can feel significant chest tightness or air trapping despite “good numbers”.
Muscle Fatigue
Breathing takes muscular effort. Chronic respiratory illness can place a long-term strain on the chest wall and breathing muscles, leading to exhaustion and increased awareness of breathing.
Mucus and Airflow Changes
Even relatively small amounts of mucus or airway narrowing can create sensations of chest heaviness or difficulty moving air.
Breathing Pattern Dysfunction (Dysfunctional Breathing)
This is increasingly recognised in people with chronic respiratory illness.
When breathing becomes difficult over months or years, people may unconsciously develop altered breathing patterns, including:
- rapid shallow breathing
- upper chest breathing
- frequent sighing
- over-breathing (hyperventilation)
- muscle tension around the chest and neck
This can worsen symptoms and create a vicious cycle where the sensation of breathlessness becomes amplified.
Symptoms may include:
- air hunger
- dizziness
- tingling
- tight chest
- panic sensations
- difficulty “switching off” breathing awareness
Importantly, this does not mean the illness is psychological or “not real”. Dysfunctional breathing can happen alongside genuine lung disease.
Why Inhalers Sometimes Seem to Help Less
Reliever inhalers such as salbutamol (Ventolin) are designed mainly to relax tightened airways.
If breathlessness is being driven partly by breathing pattern dysfunction, muscle fatigue, inflammation, hypersensitivity, or air trapping, inhalers may not always provide dramatic relief.
This can be confusing and upsetting for patients.
“But My Tests Are Normal…”
Many patients feel frustrated, frightened, or even dismissed when told that oxygen levels, peak flow readings, or chest examinations are “fine” despite severe breathlessness.
Normal oxygen saturations are reassuring in terms of immediate danger, but they do not always reflect the full experience of chronic respiratory illness.
Breathlessness is influenced by many factors including inflammation, airway sensitivity, breathing effort, muscle fatigue, anxiety associated with struggling to breathe, and altered breathing patterns.
Because of this, some people may feel profoundly breathless even when routine measurements appear relatively stable.
It can sometimes help to explain symptoms in practical, functional terms, such as:
- “I become breathless walking across the room.”
- “I recover much more slowly than usual.”
- “This feels much worse than my normal baseline.”
- “I feel exhausted by the effort of breathing.”
- “Breathing exercises seem to help settle things.”
These descriptions may help healthcare professionals understand how symptoms are affecting day-to-day life, rather than focusing only on oxygen levels or peak flow numbers.
Some patients also find it helpful to ask questions such as:
- Could breathing pattern dysfunction be contributing?
- Would respiratory physiotherapy help?
- Would pulmonary rehabilitation be appropriate?
- How should I judge when symptoms need urgent assessment?
Importantly, severe breathlessness should never simply be ignored. New, worsening, or unusual symptoms still require proper medical assessment.
At the same time, many people with chronic lung disease experience very real symptoms that are not always fully reflected by routine measurements alone.
Why Breathlessness Can Feel So Frightening
The sensation of breathlessness is created by the brain interpreting signals from the lungs, breathing muscles, chest wall, blood chemistry, and nervous system.
This means that the feeling of “not getting enough air” is not determined only by oxygen levels.
In chronic lung disease, several things can trigger the sensation of breathlessness, including:
- inflamed or sensitive airways
- extra effort needed to move air in and out
- air trapping in the lungs
- muscle fatigue
- rapid or shallow breathing
- stress hormones released during breathing distress
- heightened awareness of breathing sensations
When breathing becomes uncomfortable, the body naturally responds with anxiety and adrenaline. This is a protective survival response.
Unfortunately, this can sometimes create a cycle:
breathlessness → anxiety → faster breathing → more chest tightness → worse breathlessness
This does not mean symptoms are “psychological”. The physical sensation is real, but the body’s alarm systems can unintentionally amplify it.
What Can Help During an Episode of Breathlessness?
Different techniques help different people, and severe or rapidly worsening symptoms should always be medically assessed. However, some patients find the following approaches helpful during episodes of distressing breathlessness:
Slow the Breathing Rate
Trying to slow breathing gently can help reduce over-breathing and chest tightness.
Some people find it helpful to:
- breathe in gently through the nose
- breathe out slowly through pursed lips
- focus on making the out-breath longer than the in-breath
Use a Recovery Position
Sitting forward slightly with the arms supported on knees or a table can sometimes reduce the work of breathing.
Reduce Panic and “Air Hunger”
Trying to fight for bigger and bigger breaths can sometimes worsen symptoms.
Some patients find it more helpful to focus on:
- gentle breathing rhythm
- relaxing the shoulders and neck
- slowing breathing rather than deepening it
- focusing attention away from the chest where possible
Use Prescribed Treatments Appropriately
Follow the advice provided by your healthcare team regarding inhalers, nebulisers, airway clearance, or rescue medication.
If inhalers are not helping as expected, this should be discussed with a respiratory specialist rather than simply increasing use repeatedly.
Know Your “Usual” Pattern
Many patients find it useful to learn the difference between:
- their “usual” chronic breathlessness
- breathing pattern dysfunction or over-breathing episodes
- symptoms suggesting infection or acute deterioration
This can help patients feel more confident recognising when urgent medical assessment may be needed.
Can Breathing Retraining Help?
Some patients find breathing retraining exercises very helpful, especially when guided by:
- respiratory physiotherapists
- specialist breathing services
- pulmonary rehabilitation teams
- asthma nurse specialists
Breathing retraining may include:
- slowing breathing rate
- diaphragmatic (“belly”) breathing
- nasal breathing techniques
- recovery breathing positions
- relaxation techniques
- paced activity and pacing strategies
Some NHS respiratory teams recommend online breathing resources and guided exercises to help patients recognise and manage over-breathing patterns.
These approaches are usually intended to work alongside medical treatment — not instead of it.
Living With an “Invisible” Symptom
One of the hardest aspects of chronic breathlessness is that outward signs may not always match how severe symptoms feel internally.
Many patients report feeling dismissed when oxygen levels are normal or when tests appear “better than expected”.
The experience of breathlessness is real, even when routine measurements do not fully explain it.
This is one reason why specialist respiratory assessment can be important in complex conditions such as aspergillosis.
When to Seek Medical Help
You should seek urgent medical advice if breathlessness is:
- suddenly worsening
- associated with chest pain
- causing blue lips or fingertips
- associated with falling oxygen saturations
- accompanied by fever or signs of infection
- causing confusion or severe exhaustion
- significantly different from your usual symptoms
Even if previous episodes have been related to breathing pattern dysfunction, new or worsening symptoms should still be medically assessed.
Further Support
You may also find these resources helpful:
Last reviewed: May 2026
Produced by: National Aspergillosis Centre CARES Team / Aspergillosis Website
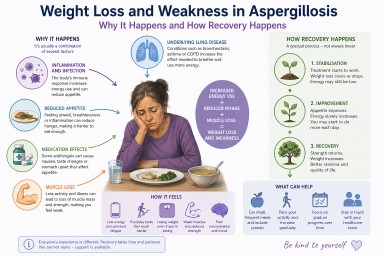
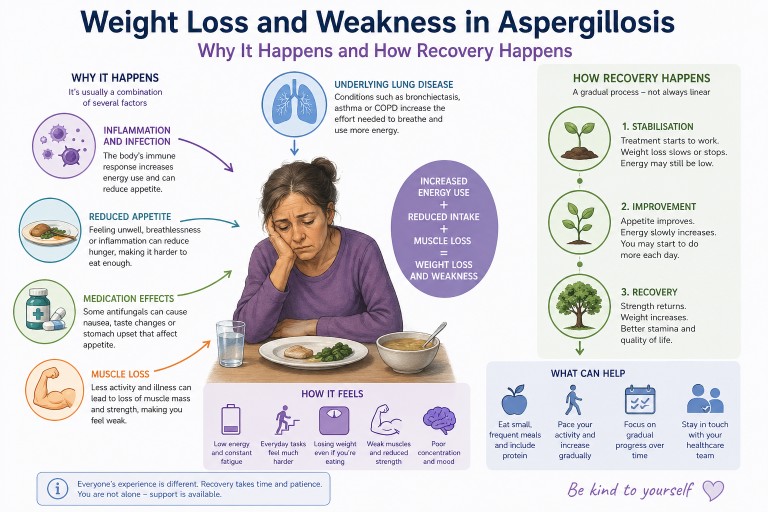
Weight Loss and Weakness in Aspergillosis: Why It Happens, How It Feels, and What Helps Recovery
Last reviewed: April 2026
Unexpected weight loss and severe weakness are among the most worrying symptoms people report after being diagnosed with aspergillosis. Many describe feeling unlike themselves—physically drained, thinner than they have ever been, and struggling with everyday activities.
This article explains why this happens, what is going on in the body, and what recovery typically looks like.
---
Key Points
- Weight loss and fatigue are common in aspergillosis, particularly early in the illness or during flare-ups.
- They are usually caused by a combination of inflammation, increased energy use, reduced appetite, and muscle loss.
- Medication side effects can contribute but are rarely the main cause.
- Many people improve over time, but recovery is usually gradual and can take weeks to months.
- Stabilising weight is often the first important step before regaining strength.
---
Contents
- Why does aspergillosis cause weight loss?
- What is happening inside the body?
- Which types of aspergillosis are affected?
- Why does it feel so severe?
- Does it get better?
- What can help day to day?
- Nutrition and rebuilding strength
- When to seek medical advice
- Common questions
---
Why does aspergillosis cause weight loss?
Weight loss in aspergillosis is rarely due to a single cause. Instead, it is usually the result of several overlapping processes.
1. Increased energy use (hypermetabolism)
When the body is dealing with infection or inflammation, it requires more energy. This is sometimes described as a hypermetabolic state.
- The immune system is active and consumes energy
- The body produces inflammatory signals
- Breathing effort may increase
This means you may be burning more calories than usual—even at rest.
2. Reduced appetite
Many people notice they are eating less, sometimes without realising it. This may be due to:
- Feeling unwell or fatigued
- Shortness of breath when eating
- Changes in appetite driven by inflammation
3. Medication effects
Some treatments can affect appetite or digestion. For example:
- Antifungal medications such as itraconazole or voriconazole may cause nausea or taste changes
- Steroids may increase appetite but can also contribute to muscle weakness over time
Medication effects vary widely and are usually only part of the overall picture.
4. Muscle breakdown
During illness, the body may break down muscle to meet energy needs. This can happen quickly, especially if activity levels fall.
This leads to:
- Loss of strength
- Reduced stamina
- A feeling of being “weak” rather than just lighter
5. Underlying lung disease
Many people with aspergillosis also have conditions such as bronchiectasis, asthma, or chronic obstructive pulmonary disease (COPD). These can increase the effort required for breathing and contribute to ongoing energy use.
---
What is happening inside the body?
Several biological processes contribute to weight loss and fatigue:
- Inflammatory signalling: The immune system releases chemical signals that affect metabolism and appetite
- Catabolism: The body breaks down tissues (including muscle) to release energy
- Energy imbalance: More energy is used than consumed
This combination can make weight loss feel rapid and difficult to control.
---
Which types of aspergillosis are affected?
These symptoms are most commonly seen in:
- Chronic Pulmonary Aspergillosis (CPA)
- Allergic Bronchopulmonary Aspergillosis (ABPA), particularly during flare-ups
However, not everyone experiences weight loss, and severity varies.
---
Why does it feel so severe?
Many people describe this stage as one of the most difficult parts of their illness. This is because several factors are happening at once:
- Physical energy is reduced
- Muscle strength has declined
- The body is under ongoing stress
- Recovery has not yet begun
This can make everyday activities—such as walking, cooking, or even eating—feel unusually difficult.
---
Does it get better?
In many cases, yes—there is gradual improvement over time, especially once treatment begins to control the condition.
Recovery often follows a pattern:
- Initial phase: weight loss and severe fatigue
- Stabilisation: weight loss slows or stops
- Recovery: gradual return of strength and energy
This process is usually slow and uneven, with good and bad days.
---
What can help day to day?
1. Focus on maintaining nutrition
- Eat small amounts regularly rather than large meals
- Choose foods that are easy to prepare and eat
- Include protein to support muscle maintenance
2. Pace activity carefully
- Gentle movement can help maintain strength
- Avoid pushing too hard, as this can worsen fatigue
- Increase activity gradually as energy improves
3. Look at trends over time
It can be helpful to focus on gradual changes such as:
- Weight stabilising
- Small improvements in energy
---
Nutrition and rebuilding strength
Recovery often happens in stages:
- Stage 1: Stabilising weight
- Stage 2: Gradually increasing intake
- Stage 3: Rebuilding muscle and strength
Regaining muscle mass takes time and usually follows once the underlying condition is better controlled.
---
When to seek medical advice
You should contact your healthcare team if you experience:
- Continued or rapid weight loss
- Increasing weakness
- Difficulty eating or swallowing
- New or worsening symptoms
This may indicate the need for additional support or adjustment of treatment.
---
Common questions
Is weight loss just due to poor appetite?
No. Reduced appetite is only one factor. Increased energy use and muscle loss are also important contributors.
Are medications the main cause?
Medications can contribute, but they are rarely the main reason for weight loss.
Will I regain my strength?
Many people do regain strength over time, although recovery is usually gradual.
Why does recovery take so long?
The body needs time to reduce inflammation, restore energy balance, and rebuild muscle.
---
Summary
Weight loss and weakness in aspergillosis are common and can feel severe, particularly early in the illness. They are usually caused by a combination of increased energy use, reduced appetite, muscle loss, and underlying lung disease.
Although recovery can take time, many people improve gradually as treatment takes effect.
---
Further Reading
- Chronic Pulmonary Aspergillosis (CPA)
- Allergic Bronchopulmonary Aspergillosis (ABPA)
- Treatment of Aspergillosis
---
Author & Review
Prepared for aspergillosis.org to support patient understanding. Content reflects current clinical knowledge and patient-reported experience.
Disclaimer
This page is for general information only and does not replace advice from your healthcare team.
Can Aspergillosis Be Cured? Understanding Treatment, Control, and Long-Term Therapy
Last reviewed: April 2026
Key points
- Aspergillosis is caused by fungi from the Aspergillus group.
- Most people breathe in Aspergillus spores regularly without becoming ill.
- In some people, damaged airways, lung cavities, mucus plugs, or immune responses allow the fungus or fungal material to persist.
- Antifungal treatment may aim to cure, but in many cases the goal is long-term control.
- Steroids can reduce harmful inflammation in allergic disease, but they can also reduce the body’s ability to clear fungus.
Table of contents
- Overview
- Why breathing in spores does not usually cause disease
- Why aspergillosis can be hard to clear
- Infection and ABPA: different reasons for persistence
- Do steroids influence this?
- Control vs cure
- Common antifungal treatments
- Why you may not hear many success stories
- When to seek medical advice
- Common questions
Overview
It is very common for people diagnosed with aspergillosis to feel worried when they read that others have been taking antifungal medication for months or even years.
This can lead to an understandable question:
“Does treatment actually work, or will I have this forever?”
The answer is more nuanced than a simple yes or no. Different forms of aspergillosis behave differently, and treatment goals vary depending on the condition.
Two of the most common conditions are:
- Allergic Bronchopulmonary Aspergillosis (ABPA) – an allergic immune reaction to Aspergillus in the airways
- Chronic Pulmonary Aspergillosis (CPA) – a long-term fungal infection, usually in areas of damaged lung tissue
Understanding this difference is key to understanding why treatment may continue for a long time.
If you would like a more detailed explanation of how these conditions are diagnosed and managed, see our guides to chronic pulmonary aspergillosis (CPA) and allergic bronchopulmonary aspergillosis (ABPA).
Why breathing in spores does not usually cause disease
Aspergillus spores are present in the environment, including air, soil, compost, dust, and decaying vegetation. Most people breathe in small numbers of these spores regularly without becoming ill.
In healthy lungs, spores are usually cleared by the immune system and by the normal cleaning mechanisms of the airways.
This means:
- Exposure to Aspergillus is common
- Most exposure does not lead to disease
- Aspergillosis usually develops only when there are additional risk factors, such as lung damage, mucus trapping, or altered immune responses
So it is not accurate to think of most patients as being “constantly reinfected”. A better way to think about it is that some lungs provide conditions where Aspergillus, or fungal material, can persist and continue to cause problems.
Why aspergillosis can be hard to clear
Aspergillosis can be difficult to clear for several reasons. These include the structure of the lungs, the biology of the fungus, limitations of drug penetration, and the way the immune system responds.
1. Damaged lung tissue can provide protected spaces
In CPA, Aspergillus often grows in areas of abnormal lung, such as cavities, scarred tissue, or areas affected by bronchiectasis.
These areas can act as protected spaces where the fungus is harder for the immune system and antifungal medicines to reach.
2. Thick mucus can trap fungus and fungal material
In airway diseases such as asthma, bronchiectasis, and ABPA, thick mucus can trap spores, hyphae, and fungal fragments.
This trapped material can continue to stimulate inflammation even when the fungus is not invading lung tissue.
3. Antifungal medicines may suppress rather than sterilise
Antifungal medications can reduce fungal activity and help prevent progression, but they may not always remove every trace of fungus from damaged lung spaces or mucus-filled airways.
For this reason, success is often measured by:
- Improved symptoms
- Stabilised weight and energy
- Fewer flare-ups
- Stable or improved scans
- Prevention of further lung damage
Infection and ABPA: different reasons for persistence
Chronic pulmonary aspergillosis: persistence of infection
In chronic pulmonary aspergillosis, the problem is fungal growth in damaged lung tissue.
- Lung cavities provide spaces where fungus can grow
- Drug penetration may be limited
- The immune system may not fully clear infection
Allergic bronchopulmonary aspergillosis: persistence of reaction
In ABPA, the main issue is an exaggerated immune response.
- Mucus traps fungal material
- Small amounts can trigger strong reactions
- Inflammation leads to more mucus
Do steroids influence this?
Yes. Steroids can be helpful but must be used carefully.
In ABPA, they reduce inflammation but may also reduce fungal clearance.
In chronic infection, steroids can increase the risk of persistence or progression.
Monitoring and drug interactions are important during treatment.
Control vs cure: what is the goal?
For many people, the realistic goal is:
- Stability rather than eradication
- Reduced symptoms
- Prevention of progression
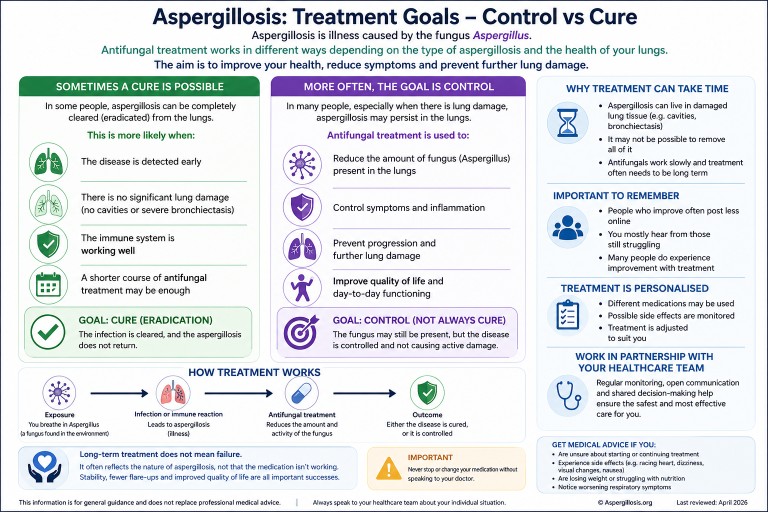
Common antifungal treatments
- Itraconazole
- Voriconazole
- Posaconazole
These treatments are selected based on individual factors and require monitoring.
Why don’t I hear many success stories?
People who improve often post less, while those still struggling are more visible in forums.
When to seek medical advice
- Uncertainty about treatment
- Side effects
- Weight loss
- Worsening symptoms
Common questions
Can aspergillosis be cured?
Sometimes, but often it is managed long-term.
Are people constantly reinfected?
No. Most people clear spores regularly without issue.
Why is ABPA difficult to treat?
Because of ongoing immune reactions and mucus trapping.
Further reading
- Chronic Pulmonary Aspergillosis (CPA)
- Allergic Bronchopulmonary Aspergillosis (ABPA)
- Antifungal medicines
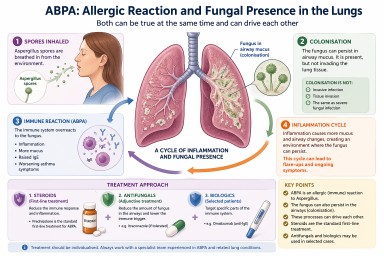
ABPA: Allergic Reaction or Fungal Presence in the Lungs?
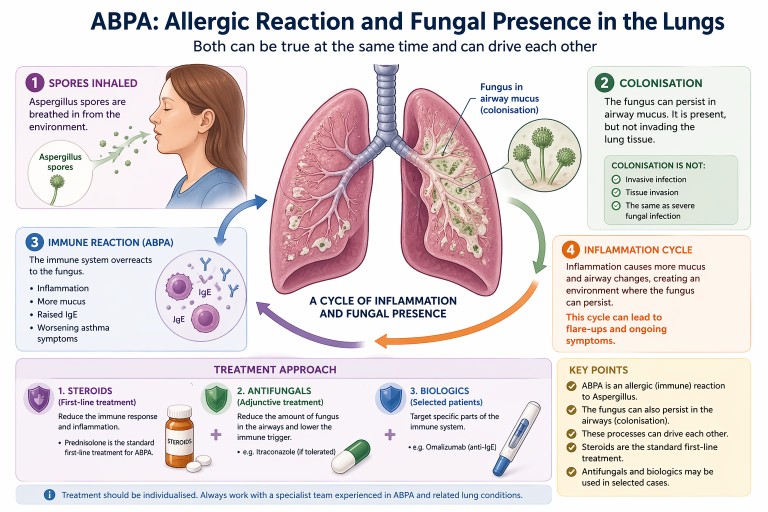
Allergic Bronchopulmonary Aspergillosis (ABPA) can be confusing. It is often described as an allergic condition, yet patients may also be told there is “fungus in the lungs”.
In reality, both can be true at the same time.
Key points
- ABPA is primarily an allergic (immune) reaction to Aspergillus
- The fungus can also persist in the airways (colonisation)
- Colonisation is not the same as invasive infection
- Steroids are the standard first-line treatment
- Antifungals and biologics may be used in selected cases
What is ABPA?
ABPA is a condition where the immune system overreacts to a common environmental fungus, most often Aspergillus fumigatus.
For a full introduction, see our guide to
what is ABPA.
This leads to:
- airway inflammation
- increased mucus production
- worsening asthma symptoms
- raised Immunoglobulin E (IgE) levels
If you are unfamiliar with IgE, see
our explanation of IgE.
Importantly, ABPA is not an invasive fungal infection. The lungs are reacting to fungal material rather than being invaded by it.
Can the fungus live in the lungs?
Yes—this is an important nuance.
In people with conditions such as:
- asthma
- bronchiectasis
the structure of the airways can allow Aspergillus to persist in mucus within the airways. This is known as colonisation.
Colonisation means:
- the fungus is present in airway mucus
- it may grow at low levels locally
- it does not invade lung tissue
This is very different from serious invasive fungal infections seen in severely immunocompromised patients.
How ABPA and colonisation interact
ABPA and colonisation are best understood as part of a spectrum of Aspergillus-related lung disease.
They can interact in a cycle:
- Aspergillus spores are inhaled
- The fungus persists in airway mucus
- The immune system reacts strongly (ABPA)
- Inflammation leads to more mucus production
- This creates an environment where the fungus can persist
This cycle can contribute to:
- recurrent flare-ups
- ongoing inflammation
- persistently raised IgE levels
Standard treatment: steroids
Oral corticosteroids are the traditional first-line treatment for ABPA.
Steroids such as prednisolone are used because they:
- reduce airway inflammation
- suppress the allergic immune response
- improve symptoms such as breathlessness and cough
- help control flare-ups
Learn more in our guide to
steroids and aspergillosis.
Where do antifungals fit in?
Antifungal medications such as itraconazole are often used as an additional (adjunctive) treatment rather than the primary therapy.
The aim is to:
- reduce the amount of fungus present in the airways
- lower the trigger for the immune response
- sometimes allow a reduction in steroid dose
For more detail, see our
antifungal treatments guide.
Biologic treatments
Biologic therapies such as omalizumab (anti-IgE) are increasingly used in some patients, particularly when:
- symptoms are difficult to control
- steroid side effects are significant
- there is severe asthma alongside ABPA
Read more about
biologics in asthma and ABPA.
Why results and symptoms may not match
Some people with ABPA may feel relatively well but still have:
- raised IgE levels
- evidence of fungal presence in the airways
This is because blood tests reflect underlying immune activity, which does not always match how a person feels day to day.
Why treatment is individualised
Although steroids are the standard treatment, management is often tailored to the individual. This is because people may also have:
- asthma
- bronchiectasis
- difficulty tolerating antifungal medications
- side effects from long-term steroid use
Specialist teams balance these factors when planning treatment.
When to seek medical advice
People with ABPA should seek medical review if they experience:
- worsening breathlessness
- increased cough or sputum
- new or persistent chest symptoms
- possible medication side effects
Treatment decisions should always be made with a clinician experienced in managing Aspergillus-related lung disease.
Summary
- ABPA is primarily an allergic condition
- The fungus can also persist in the airways (colonisation)
- These processes can drive each other
- Steroids are the standard first-line treatment
- Antifungals and biologics may be used in selected cases
Last reviewed: April 2026
Prepared by: National Aspergillosis Centre (patient information resource)
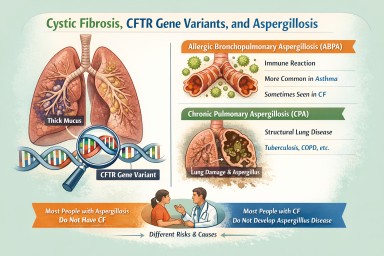
Cystic Fibrosis, CFTR Gene Variants, and Aspergillosis
Last reviewed: 8 April 2026
Some people with aspergillosis are told they have cystic fibrosis (CF), or that they carry a CFTR gene variant. This can be unexpected and may raise concerns about whether this explains their symptoms or diagnosis.
This article explains how cystic fibrosis and CFTR gene variants relate to Aspergillus-related lung disease, what current research shows, and—importantly—what conclusions should not be drawn.
Contents
- Key points
- Important reassurance
- What is cystic fibrosis?
- What is a CFTR gene variant?
- How CFTR affects the lungs
- How Aspergillus behaves in the lungs
- ABPA and cystic fibrosis
- CPA and cystic fibrosis
- Modern CF treatments and Aspergillus
- Does a CFTR variant explain symptoms?
- What should patients take from this?
- When to seek medical advice
- Conclusion
- References
Key points
- Most people with aspergillosis do not have cystic fibrosis.
- Most people with cystic fibrosis do not develop ABPA or CPA.
- ABPA is linked to mucus and immune responses, not just infection.
- CFTR variants may contribute to risk in some people, but are usually only one factor.
- CPA is mainly driven by structural lung damage, not CFTR genetics.
Important reassurance
Most people with aspergillosis do not have cystic fibrosis, and most people with cystic fibrosis do not develop Aspergillus-related disease.
Although these conditions can overlap, they are usually separate. Genetic findings such as CFTR variants should be interpreted carefully and in context.
What is cystic fibrosis?
Cystic fibrosis is a genetic condition caused by changes in the CFTR gene. This gene regulates salt and water movement across cells.
When CFTR function is reduced:
- mucus becomes thick and sticky
- airways are harder to clear
- microorganisms persist more easily
This creates an environment where bacteria and fungi can accumulate over time.
What is a CFTR gene variant?
CFTR variants range from severe mutations (causing cystic fibrosis) to mild or uncertain variants.
Carriers (with one variant):
- are common in the general population
- usually have no symptoms
- may have subtle effects in some cases
These subtle effects may include reduced mucus clearance or increased susceptibility to airway inflammation.
How CFTR affects the lungs
CFTR dysfunction affects the lungs in several key ways:
- Mucus dehydration: mucus becomes thick and difficult to clear
- Impaired clearance: particles and microbes remain in the airways
- Chronic inflammation: immune responses become exaggerated
This combination creates a “retention environment” where inhaled organisms—including Aspergillus—may persist.
How Aspergillus behaves in the lungs
Aspergillus is inhaled by everyone, but its effects vary depending on the lung environment.
- Healthy lungs: spores are cleared
- Impaired clearance: spores may persist
- Sensitive immune system: allergic reactions may develop
- Damaged lungs: chronic infection may develop
This explains why Aspergillus-related disease is diverse and depends heavily on underlying lung conditions.
ABPA and cystic fibrosis
ABPA is an allergic immune reaction to Aspergillus.
It is recognised in cystic fibrosis because:
- mucus retention increases exposure to Aspergillus
- immune responses can be exaggerated
However:
- Many CF patients never develop ABPA
- Most ABPA patients do not have CF
Some studies suggest CFTR variants may increase susceptibility, but this is not consistent across all research.
Key message: ABPA and CF can overlap, but one does not imply the other.
CPA and cystic fibrosis
CPA is a chronic fungal infection that develops in structurally damaged lungs.
The most important risk factor is:
pre-existing lung damage
This includes:
- bronchiectasis
- previous tuberculosis
- COPD
Cystic fibrosis can lead to bronchiectasis, and therefore indirectly increase CPA risk.
However:
- CPA is rarely driven directly by CFTR genetics
- most CPA patients do not have CF
Key message: CPA is primarily a disease of lung structure, not genetics.
Modern CF treatments and Aspergillus
CFTR modulators (such as elexacaftor/tezacaftor/ivacaftor) have transformed CF care.
They:
- improve CFTR function
- thin mucus
- improve clearance
Studies suggest:
- reduced Aspergillus detection in some patients
- fewer ABPA exacerbations in some cases
However:
- ABPA still occurs
- existing lung damage remains
- immune responses are not fully corrected
Overall: these therapies improve risk but do not eliminate Aspergillus-related disease.
Does a CFTR variant explain symptoms?
No single factor explains complex lung disease.
Symptoms may result from:
- underlying lung disease
- infection
- inflammation
- environmental exposure
A CFTR variant may contribute, but is rarely the sole cause.
What should patients take from this?
- CF and CFTR variants can sometimes contribute
- ABPA has the strongest connection
- CPA is mainly driven by lung damage
- Most patients with aspergillosis do not have CF
When to seek medical advice
Seek advice if symptoms worsen, change, or include coughing up blood, fever, or chest pain.
Conclusion
Cystic fibrosis and CFTR gene variants can play a role in some patients with Aspergillus-related lung disease, particularly where mucus clearance is affected. However, they should not be overemphasised. In most cases, they are just one part of a broader clinical picture involving lung structure, immune response, and environmental exposure.
References
- Miller PW et al. (1996)
- Marchand E et al. (2001)
- Eaton TE et al. (2002)
- Agarwal R et al. (2012)
- Chaudhary N et al. (2012)
- Warris A et al. (2019)
- Gamaletsou MN et al. (2018)
- Kosmidis C & Denning DW (2015)
- Moldoveanu B et al. (2021)
- Malik HS et al. (2025)
- Zeng C et al. (2025)
- Bendixen MP et al. (2025)
This article is for general information and does not replace advice from your clinical team.
Asthma and Aspergillosis
How fungal spores interact with asthma and other lung diseases
Every day we inhale thousands of microscopic fungal spores from the environment. One of the most common fungi in the air is Aspergillus fumigatus. In healthy lungs these spores are removed quickly by the lungs’ natural defence systems and cause no illness.
However, in people with asthma—particularly severe asthma—the interaction between the lungs and Aspergillus can be very different. The fungus may trigger allergic inflammation, grow in mucus within the airways, or occasionally contribute to chronic lung disease.
Understanding this relationship helps explain several important conditions including:
-
Aspergillus sensitisation
-
Severe Asthma with Fungal Sensitisation (SAFS)
-
Allergic Bronchopulmonary Aspergillosis (ABPA)
-
Aspergillus bronchitis
-
Chronic Pulmonary Aspergillosis (CPA)
Although asthma is the most common condition linked to Aspergillus allergy, other lung diseases such as bronchiectasis, Chronic Obstructive Pulmonary Disease (COPD), and tuberculosis-related lung damage can also create environments where the fungus becomes important.
Why Asthma Creates a Favourable Environment for Aspergillus
Asthma is a disease of airway inflammation and hyper-reactivity. The bronchi narrow during attacks because the airway wall becomes swollen and the surrounding smooth muscle contracts.
Several features of asthma make it easier for Aspergillus spores to remain in the lungs.
Mucus production
Asthma often causes increased production of thick airway mucus.
Normally mucus traps inhaled particles and moves them upward toward the throat via the mucociliary escalator.
In asthma:
-
mucus becomes thicker
-
clearance becomes less efficient
-
spores remain trapped
This trapped environment allows fungal spores to persist in the airway mucus.
Allergic immune responses
Many asthma patients have Type-2 (T2) inflammation (50-70%), involving immune pathways driven by:
-
Immunoglobulin E (IgE)
-
Interleukin-4
-
Interleukin-5
-
Interleukin-13
-
eosinophils
These pathways respond strongly to fungal allergens. When the immune system recognises Aspergillus proteins it may trigger allergic inflammation in the airways.
Fungal sensitisation is increasingly recognised as an important contributor to severe asthma (PMID: 24735832).
Aspergillus Sensitisation
Many people with asthma develop allergic sensitisation to Aspergillus.
Sensitisation means the immune system produces antibodies against fungal proteins.
Features include:
-
positive Aspergillus skin test or IgE blood test
-
worsening asthma symptoms
-
increased exacerbations
Studies suggest 10–25% of patients attending severe asthma clinics show Aspergillus sensitisation (PMID: 24735832).
However, sensitisation alone does not necessarily cause lung damage.
Severe Asthma with Fungal Sensitisation (SAFS)
Some patients with severe asthma have fungal sensitisation but do not meet the criteria for ABPA.
This condition is known as Severe Asthma with Fungal Sensitisation (SAFS).
Typical features include:
-
severe or poorly controlled asthma
-
fungal allergy
-
moderate IgE elevation
A randomised controlled trial demonstrated that antifungal therapy may improve symptoms in some SAFS patients (PMID: 18948425).
Allergic Bronchopulmonary Aspergillosis (ABPA)
Allergic Bronchopulmonary Aspergillosis is the most important Aspergillus-related disease associated with asthma.
ABPA occurs when Aspergillus grows within airway mucus and triggers a strong allergic immune response.
Typical findings include:
-
very high total IgE levels
-
Aspergillus-specific IgE and IgG antibodies
-
eosinophilia
-
mucus plugs containing fungal hyphae
-
central bronchiectasis
ABPA occurs in approximately:
-
1–2% of all asthma patients
-
up to 10–15% of severe asthma patients
These figures come from global prevalence estimates of ABPA in asthma populations (PMID: 23210682/.
Modern diagnostic criteria for ABPA were updated by the International Society for Human and Animal Mycology (ISHAM) in 2024 (PMID: 38423624).
Asthma and Aspergillus Disease Pathway
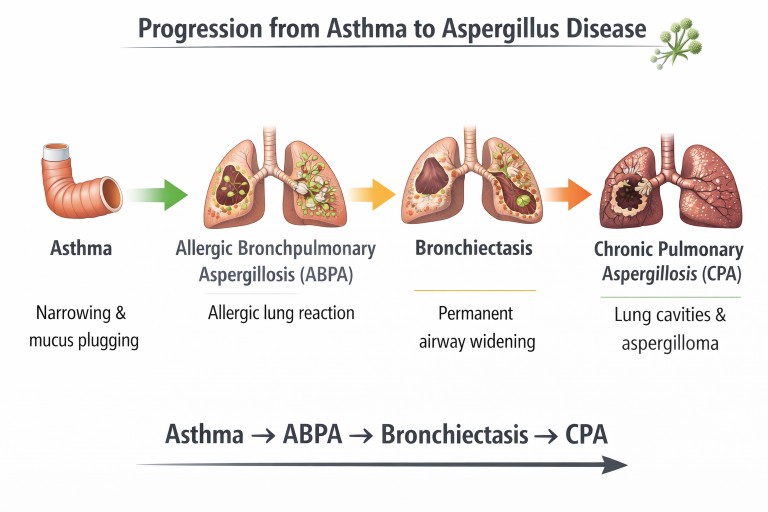
Possible interactions between asthma and Aspergillus. Some patients develop allergic disease (ABPA) which may lead to airway damage such as bronchiectasis (NB Progression to CPA is very rare).
When ABPA Causes Bronchiectasis
Repeated inflammation from ABPA may damage airway walls and lead to bronchiectasis.
Bronchiectasis occurs when airways become:
-
permanently widened
-
distorted
-
unable to clear mucus effectively
Instead of being cleared from the lungs, mucus pools in the airways.
This retained mucus creates an environment where microorganisms—including fungi—can grow.
Aspergillus Bronchitis
In some patients with bronchiectasis or chronic lung disease, Aspergillus may persist in airway mucus and cause chronic airway infection rather than allergy.
Symptoms may include:
-
chronic cough
-
sputum production
-
repeated positive Aspergillus cultures
IgE levels are usually lower than in ABPA.
Chronic Pulmonary Aspergillosis (CPA)
Chronic Pulmonary Aspergillosis is a slowly progressive fungal infection of damaged lung tissue.
CPA usually develops in lungs containing:
-
cavities
-
severe structural damage
Common underlying diseases include:
-
tuberculosis
-
sarcoidosis
-
severe COPD
Globally, the most common cause of CPA is previous tuberculosis infection (PMID: 22271943).
Asthma alone rarely causes CPA, but severe bronchiectasis or ABPA-related lung damage may occasionally lead to it.
Aspergillosis and Immune Competence
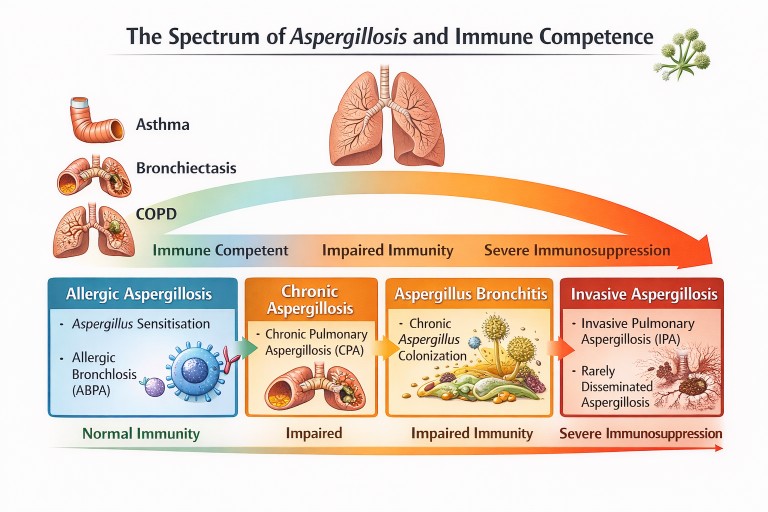
Different forms of aspergillosis occur depending on lung damage and immune function.
Other Lung Diseases Linked to Aspergillus
Although asthma is the most common condition associated with Aspergillus allergy, several other lung diseases can predispose to fungal disease.
Bronchiectasis
Dilated airways trap mucus, allowing fungi and bacteria to persist.
COPD
Chronic airway inflammation may lead to Aspergillus bronchitis or chronic pulmonary aspergillosis.
Tuberculosis
Post-tuberculosis lung cavities are the most common global cause of chronic pulmonary aspergillosis (PMID: 22271943).
Key Messages
-
Asthma is one of the most important diseases associated with Aspergillus-related lung conditions.
-
Many asthma patients develop fungal sensitisation.
-
A smaller proportion develop Allergic Bronchopulmonary Aspergillosis (ABPA).
-
Repeated inflammation from ABPA can lead to bronchiectasis.
-
Chronic pulmonary aspergillosis is rare in asthma alone but may occur if significant lung damage develops.
Understanding these interactions helps guide diagnosis and treatment for people living with asthma and Aspergillus-related disease.
Further reading
Agarwal R, Chakrabarti A, Shah A, Gupta D, Meis JF, Guleria R, Moss R, Denning DW; ABPA complicating asthma ISHAM working group. Allergic bronchopulmonary aspergillosis: review of literature and proposal of new diagnostic and classification criteria. Clin Exp Allergy. 2013 Aug;43(8):850-73. doi: 10.1111/cea.12141. PMID: 23889240.
Denning DW, Pleuvry A, Cole DC. Global burden of chronic pulmonary aspergillosis as a sequel to pulmonary tuberculosis. Bull World Health Organ. 2011 Dec 1;89(12):864-72. doi: 10.2471/BLT.11.089441. Epub 2011 Sep 27. PMID: 22271943; PMCID: PMC3260898.
Looking further into the future - could we control lung damage, preserve healthy lung tissue better?
Can Lungs Repair Themselves?
What New Research Means for People with CPA (and Other Aspergillosis)
A recent scientific discovery has helped researchers understand how certain lung cells decide whether to focus on repairing damage or defending against infection. The work, highlighted by the Mayo Clinic and published in Nature Communications, describes a molecular “switch” inside specialised lung cells that influences this balance.
For people living with Chronic Pulmonary Aspergillosis (CPA) — and also those with Allergic Bronchopulmonary Aspergillosis (ABPA) — this kind of research is relevant. But it needs careful explanation.
This is not about rebuilding destroyed lungs.
It is about understanding how to better protect and preserve the lung tissue that remains.
The Discovery: A “Repair vs Defence” Switch
Researchers identified a regulatory circuit in alveolar type II (AT2) cells — specialised cells that:
-
Produce surfactant (which keeps air sacs open)
-
Act as a reserve “repair” population in the lung
-
Can regenerate other essential lung cells after injury
The study showed that these cells operate under tight control. When infection is present, they prioritise defence. When injury needs healing, they can switch into repair mode.
The key insight is that this switch is biologically regulated. It is not random. That means, in theory, it may one day be possible to influence it.
What “Repair” Means — and What It Does Not Mean
When we talk about lung repair in this context, we must be very clear.
It does not mean:
-
Lung cavities caused by CPA will close in the foreseeable future
-
Established fibrosis will melt away
-
Bronchiectasis will reverse
-
Severely distorted lung architecture will rebuild
CPA cavities represent major structural remodelling — destruction of alveoli, scarring, altered blood supply, and thickened pleura. Reconstructing that complex architecture is biologically extremely challenging and not currently realistic within the next decade.
What repair does realistically mean
In chronic lung disease, “repair” is more likely to mean:
-
Supporting survival of remaining alveoli
-
Preventing excessive fibrotic signalling
-
Helping lung lining cells recover more efficiently after inflammation
-
Reducing cumulative injury from repeated infection
-
Slowing progression of structural change
In other words:
Not rebuilding what is gone — but better protecting what remains.
For many people with CPA, this is a crucial distinction.
Why Preservation Is a Major Goal in CPA
CPA usually develops in lungs already weakened by conditions such as tuberculosis, non-tuberculous mycobacteria, chronic obstructive pulmonary disease, or severe pneumonia.
Over time, CPA can lead to:
-
Expanding cavities
-
Progressive scarring
-
Reduced gas exchange
-
Reduced exercise tolerance
Many patients have limited lung reserve. Even small additional losses of functioning lung tissue can significantly increase breathlessness or fatigue.
If future therapies could slow the rate of progression — even modestly — that would meaningfully affect long-term outcomes.
Flattening the decline curve is not trivial. It changes quality of life.
Why This Also Matters in ABPA
In ABPA, repeated inflammatory episodes can lead to:
-
Airway remodelling
-
Mucus plugging
-
Development or progression of bronchiectasis
Better control of inflammatory signalling — combined with improved epithelial recovery — could reduce long-term airway damage.
Again, this is about preservation rather than reversal.
Where Development Has Reached
The current research is still laboratory-based. It used advanced techniques such as:
-
Single-cell sequencing
-
Imaging of lung tissue
-
Preclinical models of injury
No human treatments based on this discovery are yet available.
However, the significance lies in identifying:
-
A defined molecular pathway
-
A controllable regulatory mechanism
-
A clearer understanding of why repair fails in chronic inflammation
That foundational knowledge is what eventually allows targeted drug development.
The Balance Challenge in Aspergillosis
There is an additional complexity in fungal lung disease.
Any attempt to promote repair must not weaken antifungal defence.
The immune system must:
-
Control Aspergillus
-
Avoid causing excessive inflammatory damage
Future therapies would need to strike that balance carefully.
What This Means for Patients Now
This discovery does not change current treatment.
The most effective preservation strategies today remain:
-
Consistent antifungal therapy when indicated
-
Careful inflammatory control
-
Biologic therapies where appropriate
-
Airway clearance
-
Vaccination and infection prevention
-
Avoiding damp and mould exposure
-
Pulmonary rehabilitation
These measures are already forms of lung preservation.
A Realistic and Hopeful Perspective
It is unlikely that cavities from CPA will be repaired in the near future.
It is realistic that within the next 5–10 years we may see improved strategies aimed at:
-
Slowing structural progression
-
Supporting endogenous repair cells
-
Reducing fibrotic signalling
-
Improving recovery after exacerbations
For people living long-term with CPA or ABPA, even incremental preservation could significantly affect independence and quality of life.
The science is still early — but understanding how the lung decides to repair itself is an important step forward.
Reference
Sawhney, A.S., Deskin, B.J., Cai, J. et al. A molecular circuit regulates fate plasticity in emerging and adult AT2 cells. Nat Commun 16, 8924 (2025). https://doi.org/10.1038/s41467-025-64224-1