How Inflammation in One Part of the Body Can Affect the Rest of the Body
Last reviewed: 24 March 2026
Audience: Patients, families, and non-specialist clinicians
Author: Aspergillosis.org editorial team
Many people think of inflammation as something that stays in one place: a painful joint, an inflamed lung, an irritated sinus, or a bowel flare. In reality, inflammation is often a whole-body process. Signals released at one site can travel through the blood, nervous system, and immune system, influencing other organs and changing how the body feels and functions overall.
This helps explain why a local health problem can sometimes lead to symptoms that seem much broader, such as fatigue, poor concentration, low mood, loss of appetite, aches, disturbed sleep, or worsening of other long-term conditions.
Key points
- Inflammation is not always confined to one organ or body part.
- Inflamed tissues release chemical messengers that can circulate throughout the body.
- The brain, heart, kidneys, liver, gut, lungs, and immune system all communicate with one another.
- This “cross-talk” can be helpful in short-term illness, but harmful when inflammation becomes prolonged.
- Ongoing inflammation is linked with fatigue, brain fog, low mood, cardiovascular strain, and worsening of other chronic diseases.
Table of contents
- What is inflammation?
- Why inflammation does not always stay local
- How the body communicates during inflammation
- Common whole-body effects of inflammation
- Why this matters in lung disease and aspergillosis
- Acute inflammation versus chronic inflammation
- What can help?
- When to seek medical advice
- Common questions
- References
What is inflammation?
Inflammation is part of the body’s defence system. It is one of the ways the immune system responds to infection, injury, irritation, allergens, or tissue damage. In the short term, inflammation is often helpful. It can help the body fight infection, clear damaged tissue, and begin repair.
But inflammation can also become too strong, too prolonged, or poorly controlled. When that happens, the effects may no longer stay limited to the original problem area.
Why inflammation does not always stay local
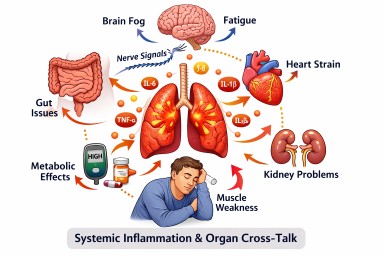
When tissue becomes inflamed, immune cells release small signalling proteins called cytokines and other inflammatory mediators. These act like chemical messages. Some stay nearby, but many enter the bloodstream and influence distant organs.
This is why inflammation in one part of the body can sometimes cause:
- tiredness or exhaustion
- feeling unwell or “washed out”
- poor concentration or “brain fog”
- worsening appetite
- sleep disruption
- higher strain on the heart or kidneys
- worsening of other inflammatory conditions
Researchers increasingly describe this as systemic inflammation or organ cross-talk. In other words, organs do not operate in isolation. They are part of an interconnected network.
How the body communicates during inflammation
1. Chemical messengers in the blood
Inflamed tissues can release cytokines such as interleukin-6 (IL-6), interleukin-1 beta (IL-1β), and tumour necrosis factor alpha (TNF-α). These may affect blood vessels, metabolism, the brain, the heart, and other immune cells.
These signals are useful during short-term illness, but if they remain elevated they may contribute to chronic symptoms and long-term health effects.
2. Organ-to-organ immune cross-talk
Modern immunology shows that the gut, liver, lungs, brain, heart, kidneys, and bone marrow can influence one another through immune signalling. A problem in one organ may therefore alter immune behaviour somewhere else.
This can be protective, but it can also become part of a vicious circle, especially in chronic disease.
3. Nerve signalling between the body and brain
Inflammation is not communicated only by blood. The nervous system also plays a role. Signals from inflamed tissues can travel through nerves, including the vagus nerve, to the brain. The brain then responds by adjusting immune activity and body-wide stress responses.
This helps explain why inflammation can affect fatigue, mood, motivation, sleep, and mental clarity.
4. Stress, hormones, and metabolism
Inflammation also interacts with the body’s hormonal and metabolic systems. This can influence energy use, blood sugar regulation, muscle strength, and appetite. Over time, chronic inflammation may put extra strain on the cardiovascular and kidney systems.
Common whole-body effects of inflammation
Fatigue
One of the most common effects of inflammation is fatigue. This is not simply feeling sleepy. It can be a profound lack of physical and mental energy. Many chronic inflammatory illnesses are associated with this kind of exhaustion.
Brain fog and mood changes
Inflammatory signals can affect the brain, contributing to reduced concentration, slowed thinking, low motivation, anxiety, or low mood. This does not mean symptoms are “all in the mind”. It means that immune activity can influence brain function.
Heart and blood vessel effects
Inflammation can make blood vessels less healthy over time and may contribute to a higher cardiovascular risk. This is one reason why long-standing inflammatory diseases are often linked to heart and circulatory problems.
Kidney effects
The kidneys are sensitive to inflammatory stress. In some conditions, long-term systemic inflammation can contribute to kidney damage or worsen existing kidney disease. Kidney disease itself can also increase inflammation, creating a two-way relationship.
Muscle weakness and reduced stamina
Ongoing inflammation can alter how muscles use energy and recover after activity. This may contribute to weakness, reduced exercise tolerance, and slower recovery after exertion.
Why this matters in lung disease and aspergillosis
For people with chronic lung conditions, including some forms of aspergillosis, inflammation in the airways or lungs may have effects beyond breathing alone. The lungs are not separate from the rest of the body.
Inflammation in the lungs may contribute to:
- general fatigue
- poor stamina
- sleep disruption
- brain fog
- loss of appetite
- worsening of other conditions
This can be especially relevant for people living with long-term inflammatory lung disease, repeated infections, allergic inflammation, or complex treatment burdens.
It is also one reason why patients sometimes feel that their symptoms are “bigger” than what would be expected from the lungs alone. Often, that experience is real and biologically plausible.
Acute inflammation versus chronic inflammation
Acute inflammation
This is the short-term response seen with infection, injury, or a sudden flare. It may cause fever, pain, swelling, and marked tiredness. Usually, it settles when the trigger is controlled.
Chronic inflammation
This is lower-grade or persistent inflammation that continues over time. It may be driven by chronic infection, immune dysregulation, ongoing tissue damage, obesity, autoimmune disease, long-term lung disease, or other medical problems. Chronic inflammation is often less dramatic but may have broader long-term effects.
What can help?
The right approach depends on the underlying cause. Broadly, management focuses on:
- identifying and treating the cause of inflammation where possible
- controlling infections or allergic triggers
- optimising treatment of the underlying disease
- supporting sleep, nutrition, and pacing of activity
- monitoring the effects on other organs when relevant
There is rarely a single quick fix for chronic inflammation. Good management usually means looking at the whole person, not just the inflamed organ.
When to seek medical advice
Please seek medical advice if inflammation-related symptoms are worsening or if you develop:
- new or severe breathlessness
- chest pain
- confusion or marked drowsiness
- new swelling, reduced urine output, or signs of dehydration
- persistent fevers
- rapid decline in energy, mobility, or daily functioning
If symptoms are sudden, severe, or alarming, seek urgent medical help.
Common questions
Does inflammation always damage the whole body?
No. Short-term, controlled inflammation is a normal and useful response. Problems are more likely when inflammation is severe, repeated, or persistent.
Can one inflamed organ affect another?
Yes. There is now strong evidence that organs influence one another through immune, vascular, metabolic, and nerve-based pathways.
Can inflammation cause fatigue even if blood tests are not dramatically abnormal?
Yes. Symptoms and blood markers do not always match perfectly. Some people experience substantial fatigue and other systemic symptoms even when routine blood tests are only mildly abnormal or intermittently raised.
Is this relevant to chronic lung disease?
Yes. Lung inflammation can have effects that go beyond breathing, including fatigue, reduced stamina, and wider body effects.
References
- Dou J, et al. The Interplay of Cross-Organ Immune Regulation in Inflammation and Cancer. MedComm. 2025.
- Jin H, Li M, et al. A body–brain circuit that regulates body inflammatory responses. Nature. 2024.
- Katkenov N, et al. Systematic Review on the Role of IL-6 and IL-1β in Cardiovascular Diseases. Journal of Cardiovascular Development and Disease. 2024.
- Nowak KL, et al. Targeting Inflammation in CKD. Current Opinion in Nephrology and Hypertension. 2025.
- Paganin W, et al. Inflammatory biomarkers in depression: a scoping review. 2024.
- Mehta NN, et al. IL-6 and Cardiovascular Risk: A Narrative Review. 2024.
- Che H, et al. Organ cross-talk: molecular mechanisms, biological functions and therapeutic opportunities. 2026.
Disclaimer: This article is for general information and education. It is not a substitute for personalised medical advice. If you are worried about worsening symptoms, new symptoms, or the effect of inflammation on your health, speak to your clinical team.
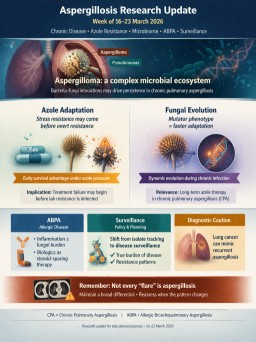
Aspergillosis Research Update (Week of 16–23 March 2026)
This week’s aspergillosis literature includes important new work on chronic pulmonary aspergillosis (CPA), aspergilloma microbiology, azole resistance evolution, biomarkers, allergic bronchopulmonary aspergillosis (ABPA), and surveillance. As usual, the most clinically useful papers for long-term aspergillosis care are prioritised.
Key points summary
- New evidence suggests that azole persistence and stress tolerance may develop before overt antifungal resistance becomes detectable.
- An aspergilloma appears to be more than a simple fungal ball: it may function as a complex microbial ecosystem involving bacterial adaptation and persistence.
- A new surveillance paper argues for moving beyond Aspergillus fumigatus strain surveillance toward clinical disease surveillance.
- ABPA review literature continues to reflect growing interest in biologic therapies as steroid-sparing treatment.
- A case report reminds clinicians that lung cancer can mimic recurrent aspergillosis, with potential for diagnostic delay.
- Biomarker studies in invasive pulmonary aspergillosis (IPA) are continuing, although most are not yet ready for routine clinical use.
Contents
- Chronic and structural disease
- Antifungal resistance and fungal evolution
- Diagnosis and biomarkers
- ABPA and allergic disease
- Surveillance, epidemiology and environment
- Case reports and diagnostic challenges
- Other relevant papers
1. Chronic and structural disease
Pseudomonas aeruginosa adaptation and persistence in the aspergilloma microbiome revealed by integrated multi-omics
Ribeiro MM, Liu C, Xu JF, Liang S, Goldman GH
G3 (Bethesda), 17 March 2026
PMID: 41843749
This is one of the most interesting papers this week for those focused on CPA and aspergilloma. The authors examine the microbial ecology of aspergilloma and show that Pseudomonas aeruginosa can adapt and persist within this environment. That matters because aspergilloma has often been thought of mainly as a fungal structure, whereas this paper supports the idea that it may be a more complex polymicrobial niche.
The study strengthens the view that chronic pulmonary aspergillosis may involve not only fungal persistence, but also bacterial-fungal interactions, biofilm-like behaviour, and long-term microbial adaptation. This may help explain why some patients remain symptomatic despite antifungal therapy, and why structural lung disease can be so difficult to stabilise.
Why it matters:
- Highly relevant to aspergilloma and CPA.
- Supports growing interest in the lung microbiome and mixed microbial communities.
- May eventually influence how we think about treatment failure, chronic symptoms, and combined antimicrobial strategies.
2. Antifungal resistance and fungal evolution
Evidence that increased azole persistence and stress resistance precede the in vivo evolution of azole resistance in Aspergillus fumigatus
Delbaje E, Pontes L, Savoldi M, Sedik S, Dichtl K, Hoenigl M, Lass-Flörl C, Silva Pereira C, Schreiber AZ, Rokas A, Lu L, Barbosa JCJ, Fill T, Dos Reis TF, Goldman GH
Microbiology Spectrum, 16 March 2026
PMID: 41837673
This is an important resistance paper. The authors provide evidence that azole persistence and stress resistance may emerge before formal azole resistance becomes established in vivo. In other words, the fungus may first become better at surviving azole exposure before developing the classical resistance patterns that laboratories can detect more easily.
That finding is highly relevant to patients with chronic aspergillosis receiving prolonged azole therapy. It suggests that the road to treatment failure may begin earlier than clinicians currently realise, and that traditional susceptibility testing may capture resistance only after important adaptive changes are already underway.
Why it matters:
- Very important for CPA management and long-term triazole treatment.
- Suggests that “susceptible” isolates may still show clinically relevant survival advantages.
- May help explain some cases of gradual loss of treatment response before overt resistance is identified.
Accelerated mutator phenotype in a clinical Aspergillus fumigatus isolate contributes to adaptive evolution
Song Y, Hokken MWJ, Zoll J, Venselaar H, Verweij PE, Melchers WJG, Rhodes J
Emerging Microbes & Infections, 16 March 2026
PMID: 41838943 |
PMCID: PMC12997362
This paper complements the study above. It describes a clinical Aspergillus fumigatus isolate with an accelerated mutator phenotype, meaning it acquires mutations more readily and can therefore adapt more quickly under selective pressure.
For chronic disease, especially where patients receive long courses of azole therapy, this is a concerning but important concept. Some fungal strains may be inherently more capable of adapting during treatment, which could contribute to the emergence of resistance, persistence, or other survival advantages.
Why it matters:
- Relevant to CPA, azole resistance, and treatment monitoring.
- Supports the idea that fungal evolution during chronic infection may be dynamic and patient-specific.
- May eventually help explain why some patients develop resistance more rapidly than others.
3. Diagnosis and biomarkers
Synergistic effects of S100 calcium-binding protein A12 combined with Pentraxin 3 in invasive pulmonary aspergillosis and their clinical application prospects
Zhou X, Hu X, Liu H
Frontiers in Cellular and Infection Microbiology, 20 March 2026
PMCID: PMC12999553
This paper focuses on invasive pulmonary aspergillosis rather than chronic disease. It explores whether combining host biomarkers such as S100A12 and Pentraxin 3 could improve diagnosis and perhaps risk stratification.
As with many biomarker studies, the concept is promising, but the clinical pathway remains uncertain. Biomarkers that reflect the host inflammatory response may ultimately complement fungal biomarkers and imaging, particularly in intensive care or immunocompromised settings.
Why it matters:
- More relevant to IPA than CPA or ABPA.
- Illustrates continued interest in host-response biomarkers.
- Potentially useful in the future, but not immediately practice-changing for routine aspergillosis care.
Diagnostic and prognostic value of serum miR-155 in chronic obstructive pulmonary disease
Wu Y, Zhang K, Zhong R, Wang W, Luo Z, Ma Z, Liang R, Wu X, Zou X
Scientific Reports, 20 March 2026
PMID: 41857172
This is not primarily an aspergillosis paper, but it includes findings relevant to invasive pulmonary aspergillosis in patients with chronic obstructive pulmonary disease (COPD). The authors report that serum miR-155 levels were lower in IPA than non-IPA patients within their study population.
That is potentially interesting because COPD is a major risk factor for IPA, and there is growing need for better tools to identify invasive fungal disease in such patients. However, this remains exploratory and should be viewed as an early biomarker signal rather than something ready for clinical use.
Why it matters:
- Relevant mainly for COPD-associated IPA risk.
- Adds to the biomarker literature, but is not yet directly applicable in routine practice.
- Limited immediate relevance for CPA and ABPA.
4. ABPA and allergic disease
Allergic bronchopulmonary aspergillosis in internal medicine
Chedal-Anglay C, Martin de Frémont G, Dupin C
Revue de Médecine Interne, 18 March 2026
PMID: 41856838
This review provides an overview of allergic bronchopulmonary aspergillosis, its diagnosis, and treatment. It reiterates that ABPA is a complex and often under-recognised inflammatory lung disease in which treatment may target inflammation, fungal burden, or both.
Importantly, the review reflects current momentum behind biotherapies (biologics) as a growing part of management. This is particularly relevant for patients in whom long-term corticosteroid exposure is problematic, ineffective, or poorly tolerated.
Why it matters:
- A useful review paper for ABPA education and clinical context.
- Supports the continuing move toward steroid-sparing treatment approaches.
- Relevant to current discussions about personalised management pathways in ABPA.
5. Surveillance, epidemiology and environment
From Aspergillus fumigatus pathogen surveillance to Aspergillus disease surveillance
van Grootveld R, van der Beek MT, Buil JB, Schoffelen AF, de Greeff SC, Bosch T, de Boer MGJ, Kuijper EJ, Verweij PE
Journal of Hospital Infection, 18 March 2026
PMID: 41862136
This is an important conceptual paper. Rather than focusing solely on surveillance of Aspergillus fumigatus as an organism, the authors argue for broader Aspergillus disease surveillance. That is a significant distinction: public health and healthcare systems may gain more by tracking actual disease burden, clinical phenotypes, antifungal resistance patterns, and outcomes than by looking only at isolates.
For services interested in national strategy, referral equity, or long-term burden of disease, this paper points toward a more mature surveillance model. It has particular relevance to discussions about CPA burden, referral pathways, registry development, and national service planning.
Why it matters:
- Important for policy, national strategy, and service development.
- Supports the case for stronger data systems around aspergillosis burden and outcomes.
- Potentially relevant to UK service planning and inequity mapping work.
A deep dive into the diversity of the Aspergillus community in the lakes of northern Iran
Kor M, Hedayati M, Abastabar M, Haghani I, Nabili M, Saravani A, Javidnia J, Brandão J, Moazeni M
Frontiers in Public Health, 20 March 2026
PMCID: PMC12999566
This environmental study analyses the diversity of Aspergillus species in lake environments and identifies potentially pathogenic species in water and sediment samples. It is not a clinical paper, but it adds to the wider evidence base showing that environmental reservoirs of Aspergillus are varied and widespread.
Such work contributes to broader understanding of exposure pathways and may be relevant to discussions around damp environments, environmental fungal burden, and risk in vulnerable individuals.
Why it matters:
- Mainly relevant as background epidemiology and environmental context.
- Useful for the bigger public-health picture of Aspergillus exposure.
- Limited immediate clinical impact for patient management.
6. Case reports and diagnostic challenges
Case Report: Lung squamous cell carcinoma mimicking recurrent aspergillosis in systemic lupus erythematosus
Yu J, Tang Y, Tian S, Zhu W, Dai Q
Frontiers in Medicine, 22 March 2026
PMCID: PMC13002587
This case report is particularly valuable because it highlights a major diagnostic pitfall: lung squamous cell carcinoma presenting as recurrent aspergillosis. In patients with complex lung disease or immunological disease, it can be tempting to interpret recurring pulmonary abnormalities through the lens of known infection, inflammation, or prior fungal disease. This paper is a reminder that alternative diagnoses, including malignancy, must remain in view.
For patients with chronic pulmonary abnormalities, repeated “flare” narratives can sometimes delay the recognition of another process. This has strong relevance for clinical vigilance in CPA and related diagnostic pathways.
Why it matters:
- Important reminder that not everything that looks like recurrent aspergillosis is aspergillosis.
- Relevant to diagnostic delay, differential diagnosis, and the overlap between fungal disease and cancer.
- Useful for clinician education and patient-facing discussion about why diagnosis can sometimes take time.
7. Other relevant papers
Genetic background and immune response in paracoccidioidomycosis: A systematic review and meta-analysis of single nucleotide variants
Coelho SDS, Fava WS, Burger E, Pereira-Latini AC, Pontillo A, Venturini J
PLoS Neglected Tropical Diseases, 19 March 2026
PMID: 41855184 |
PMCID: PMC13001940
This paper is not focused on aspergillosis, but it is relevant to the wider theme of host genetic susceptibility to fungal infection. It adds to the growing literature suggesting that inherited immune variation may partly shape vulnerability to invasive mycoses.
A Case of Disseminated Trichophytosis With Vascular Invasion and Multiple Ulcers: Case Report and Literature Review
Fujino K, Umemoto N, Kakurai M, Yabe H, Maekawa T, Harada K, Makimura K, Shibuya K, Demitsu T
Journal of Dermatology, 19 March 2026
PMID: 41853986
This case is relevant mainly because invasive aspergillosis was initially suspected. It is a useful reminder that other invasive fungal infections may enter the differential diagnosis in immunocompromised patients.
IFUCISTRATEGY: A Spanish Survey on the Management of Invasive Fungal Infection (IFI) in Critically Ill Patients
Zaragoza R, Estella Á, Nuvials X, Robles-Plaza M, Casado-Gómez A
Preprints.org, 17 March 2026
Preprint: PPR1166594
This preprint surveys management practices for invasive fungal infection in critically ill patients. It includes references to pulmonary aspergillosis and reflects continuing emphasis on early testing, bronchoalveolar lavage galactomannan, and timely treatment. As a preprint, it should be interpreted cautiously until peer review is complete.
TET2 germline mutation in a patient with sequential lymphoid malignancies: a novel case report
Mao X, Shen K, Wang J, Wang Z, Ao Q, Wang C, Xiao M
Annals of Hematology, 17 March 2026
PMID: 41843166 |
PMCID: PMC12995931
This paper is included as contextual evidence of aspergillosis occurring in a severely immunocompromised setting. Its relevance is mainly in reinforcing the ongoing burden of pulmonary aspergillosis in haematology patients.
Post-transplant Cyclophosphamide Reduces Bronchiolitis Obliterans Syndrome Risk Through Chronic Graft-versus-Host Disease Prevention: A Multicenter Cohort Study
Eggleston RH, Alkhateeb H, Pennington KM, Zhang Z, Torghabeh MH, Hogan WJ, Khera N, Roy V, Durani U, Yadav H
Chest, 16 March 2026
PMID: 41850483
This paper is not principally about aspergillosis, but may be of indirect relevance to post-transplant lung complications and immunosuppressed populations in whom fungal disease risk remains important.
Overall interpretation
This week’s literature is strongest in three areas: chronic disease ecology, fungal adaptation under azole pressure, and diagnostic complexity. For chronic pulmonary aspergillosis in particular, the most notable message is that disease behaviour may be shaped by more than the fungus alone. The aspergilloma paper supports a richer ecological model involving bacterial adaptation, while the resistance papers suggest that fungal survival under treatment may begin to change before classical resistance becomes obvious.
Together, these studies strengthen the case for thinking about chronic aspergillosis as a dynamic long-term host-microbe-environment problem, rather than a static fungal infection. At the same time, the lung cancer case report is an important reminder that persistent or recurrent disease patterns must still be reviewed critically, especially if the clinical course changes.
What seems most relevant this week?
- Most important for CPA: the aspergilloma microbiome paper and the two azole adaptation/resistance papers.
- Most important for ABPA: the ABPA review summarising diagnosis and evolving treatment approaches.
- Most important for service planning: the surveillance paper arguing for disease-level rather than pathogen-only monitoring.
- Most important diagnostic caution: lung cancer masquerading as recurrent aspergillosis.
References
- Ribeiro MM et al. PMID: 41843749
- Delbaje E et al. PMID: 41837673
- Song Y et al. PMID: 41838943
- Zhou X et al. PMCID: PMC12999553
- Wu Y et al. PMID: 41857172
- Chedal-Anglay C et al. PMID: 41856838
- van Grootveld R et al. PMID: 41862136
- Kor M et al. PMCID: PMC12999566
- Yu J et al. PMCID: PMC13002587
- Coelho SDS et al. PMID: 41855184
- Fujino K et al. PMID: 41853986
- Zaragoza R et al. Preprint: PPR1166594
- Mao X et al. PMID: 41843166
- Eggleston RH et al. PMID: 41850483
Building fitness with Aspergillosis
Last reviewed: 20 March 2026
Audience: Patients, carers, families, and non-specialists
Applies to: Allergic bronchopulmonary aspergillosis (ABPA), chronic pulmonary aspergillosis (CPA), Aspergillus bronchitis, severe asthma with fungal sensitisation (SAFS)
Why it feels so hard — and how to improve safely
Key points
- Feeling exhausted after activity is common in aspergillosis and does not mean you are failing.
- Breathlessness and fatigue are often due to lung changes, not just “lack of fitness”.
- Pushing too hard can make symptoms worse — pacing is essential.
- Small, steady increases in activity are more effective than big efforts.
- Respiratory physiotherapy can make a major difference.
- Some patients (e.g. with cavities or haemoptysis) need tailored, cautious approaches.
Why exercise feels so difficult
Many people with aspergillosis find that even simple activity can feel exhausting.
This is not just about fitness. It is often due to changes in how the lungs and body work, including:
- Inflamed or narrowed airways
- Mucus affecting airflow
- Reduced oxygen transfer
- Increased effort needed to breathe
In addition:
- Long-term illness can reduce muscle strength
- Treatments such as steroids may affect weight and energy
- Many people experience post-exertional fatigue (feeling worse after activity)
This means your body is working harder than it used to — even for everyday tasks.
Why “just doing more” can backfire
It is natural to think that pushing harder will improve fitness. However, in aspergillosis this often leads to a cycle:
- Doing too much on a “good day”
- Feeling exhausted afterwards
- Needing longer recovery
- Losing confidence
This is sometimes called a “boom and bust” cycle.
The goal is not to push harder — it is to build steadily and sustainably.
A safer way to build fitness
1. Find your baseline
Start with what you can do without feeling completely exhausted afterwards.
This may be much less than you expect — and that is normal.
2. Use the 70–80% rule
Only do about 70–80% of what you feel capable of.
This helps prevent setbacks and allows gradual improvement.
3. Break activity into smaller chunks
Short sessions (e.g. 10–15 minutes) with rest in between are often easier than one long effort.
4. Use breathing control
Pursed-lip breathing:
- Breathe in through your nose
- Breathe out slowly through pursed lips
This helps keep airways open and reduces breathlessness.
5. Add gentle strength work
Improving muscle strength can reduce breathlessness and improve stamina.
- Sit-to-stand from a chair
- Light squats (with support)
- Step-ups
Start small — 2–3 times per week is enough.
The role of respiratory physiotherapy
Respiratory physiotherapy can be one of the most helpful supports available.
A respiratory physiotherapist can help with:
- Breathing techniques to reduce breathlessness
- Airway clearance to manage mucus
- Tailored exercise plans at the right pace
- Confidence in what is safe to do
You may be offered this as pulmonary rehabilitation.
If you have not been referred, it is worth asking your GP or specialist team.
⚠️ Important safety advice (including bleeding risk)
Exercise is helpful for many people with aspergillosis — but it is not always appropriate to increase activity without guidance.
Be especially cautious if you have:
- Coughing up blood (haemoptysis) — current or recent
- An aspergilloma (fungal ball)
- Lung cavities
- A recent flare or worsening symptoms
Why this matters
In some forms of aspergillosis, blood vessels in the lungs can become fragile. Strain or increased pressure in the chest can increase the risk of bleeding.
When to stop and seek advice
- Coughing up blood (even small amounts)
- Sudden increase in breathlessness
- Chest pain or tightness
- Feeling significantly worse after activity
How to exercise more safely (if stable)
- Keep activity gentle and controlled
- Avoid heavy lifting or straining
- Avoid holding your breath during exertion
- Build activity gradually
If you are unsure, seek advice from your specialist team or a respiratory physiotherapist before increasing activity.
What progress really looks like
Improvement is rarely a straight line.
- Some days will be better than others
- Setbacks are common
- Progress may be slow but meaningful
This is normal in chronic lung conditions.
When to seek medical advice
Contact your healthcare team if you experience:
- New or worsening breathlessness
- Changes in cough or sputum
- Chest pain
- Coughing up blood
- Severe or persistent fatigue
Final message
Living with aspergillosis changes how your body responds to activity.
But improvement is possible — with the right approach.
Build slowly, pace yourself, and get the right support — especially from a respiratory physiotherapist.
In one sentence
Exercise can help — but it must be paced, personalised, and safe for your type of aspergillosis.
Author: Graham Atherton and ChatGPT draft support
For review by: National Aspergillosis Centre
Note: This article is for general information and does not replace medical advice.
A Drop of Blood, Real-Time Answers
Last reviewed: 20 March 2026
Audience: Patients, carers, families, and non-specialists
Topic: Point-of-care monitoring of antifungal drug levels
New bedside testing for antifungal drugs — and why patients welcome it
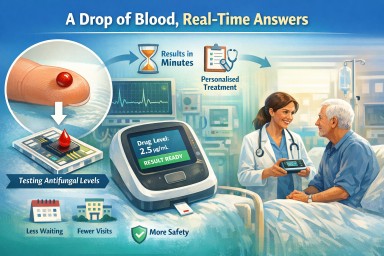
For many people taking antifungal medicines, blood tests are an important part of care. These tests help doctors check whether the amount of medicine in the body is too low, too high, or about right.
A new type of technology is being developed to do this much more quickly, using just a single drop of blood placed onto a specialised chip. Instead of sending blood away to a laboratory and waiting days for a result, this kind of test may be able to provide an answer much more quickly, sometimes during the clinic visit itself.
Patients in a recent focus group responded very positively to this idea. They welcomed not only the technology itself, but also what it could mean for their care: less waiting, less uncertainty, fewer trips to hospital, and more personalised treatment.
Key points
- A new test can measure antifungal drug levels from a drop of blood.
- The blood is placed on a specialised chip containing tiny sensors.
- Results may be available much faster than standard laboratory testing.
- This could help doctors adjust treatment more quickly and more precisely.
- Patients in a focus group strongly welcomed the technology.
- Reported benefits included less anxiety, fewer hospital visits, and more confidence in treatment decisions.
What is this new test?
This is a type of point-of-care test. That means it is designed to be used close to the patient, such as in a clinic or at the bedside, rather than sending the sample away to a central laboratory.
In this case, the aim is to measure the level of an antifungal drug in the blood from a very small sample, sometimes just a finger-prick drop. The drop of blood is placed onto a specialised chip. That chip contains tiny channels and sensors that can detect the amount of drug present.
People sometimes describe this type of system as a “lab on a chip” because it performs some of the work of a laboratory in a very small device.
How does the technology work?
The exact science varies between devices, but the general idea is similar.
- A small blood sample is taken.
This may be from a finger prick rather than a larger blood draw. - The blood is placed onto a specialised chip.
The chip is designed to handle a tiny volume of blood. - The blood moves through microscopic channels.
These channels guide the sample to the parts of the chip that do the measurement. - Sensors on the chip detect the antifungal drug.
These sensors are designed to recognise the drug or react to it in a measurable way. - A reader produces a result.
A connected device reads the signal from the chip and estimates the drug level.
Some systems use electrical signals, some use light, and some use chemical reactions. Patients do not need to understand all the engineering details to understand the main point: the chip is acting like a mini laboratory.
A simple way to think about it is this:
Instead of sending your blood sample to a distant laboratory, this technology brings part of the laboratory to your fingertip.
Why do antifungal drug levels matter?
Some antifungal medicines need careful monitoring because the “right” level can be quite important.
If the drug level is too low, the medicine may not work well enough. If the drug level is too high, side effects may become more likely.
This can be especially relevant for antifungal drugs such as:
- itraconazole
- voriconazole
- posaconazole
Drug levels can vary from person to person for many reasons, including:
- how well the body absorbs the medicine
- interactions with other medicines
- differences in liver function and metabolism
- changes in health over time
At present, monitoring usually involves sending blood to a laboratory. That works, but it can mean delays. Results may not come back quickly enough to guide decisions during the clinic appointment itself.
A faster bedside test could help clinicians make treatment decisions more quickly and could support more personalised care.
What did patients say about it?
In the patient focus group, this technology was widely welcomed. Patients were not only interested in the novelty of the test. They also recognised several practical benefits that could make day-to-day care easier and safer.
1. Faster results could reduce anxiety
Many patients described the stress of waiting for test results. Waiting can create a sense of uncertainty: Is the treatment working? Is the dose correct? Are side effects more likely?
A test that gives much quicker results was seen as reassuring. Instead of waiting days, patients liked the idea of getting answers much sooner, possibly while still in clinic.
2. Fewer visits could reduce the burden of care
For many people with chronic lung conditions or long-term illness, going to hospital is not a small task. Travel, parking, breathlessness, fatigue, mobility problems, and long waits can make even a short appointment exhausting.
Patients felt that a faster and simpler test could reduce some of this burden, especially if it could be built into a normal appointment or eventually be offered closer to home.
3. More personalised dosing felt important
Patients often understand from experience that medicines do not affect everyone in the same way. One person may tolerate a treatment well, while another may have side effects or absorb the medicine differently.
Because of this, patients valued the idea that treatment could be adjusted based on their own measured drug level, rather than relying only on standard dosing. This gave a stronger sense that care was being tailored to the individual.
4. Closer monitoring gave reassurance about safety
Antifungal drugs can be very helpful, but patients also know that some of them can have side effects and interactions. That can make treatment feel worrying, especially over longer periods.
Patients said that being able to check drug levels more quickly and more easily could help them feel safer. It suggested that treatment was being watched closely rather than left unchecked between appointments.
5. Immediate results could help patients feel more involved
Another important theme was involvement. Patients often feel that blood is taken, results disappear into the system, and decisions come later without much real-time discussion.
By contrast, a bedside result creates the possibility of discussing the number there and then. Patients felt this could help them better understand their treatment and feel more involved in decisions about dose changes and ongoing care.
6. It seemed to fit better with real life
Patients repeatedly emphasised that long-term treatment has to fit around real lives, not just clinic systems. Many welcomed the idea of a test that was quicker, simpler, and potentially more convenient.
In that sense, what patients welcomed was not just a chip or a machine, but a model of care that felt more responsive and more human-centred.
What could this mean for future care?
If this technology proves accurate, reliable, and affordable, it could support a different way of monitoring antifungal treatment.
Possible future benefits could include:
- drug level testing during the clinic appointment itself
- faster dose adjustment when levels are too high or too low
- closer monitoring when starting or changing treatment
- fewer repeat visits just to check blood levels
- potential future use in community settings or, one day, at home
It is important to be realistic. New technologies must be carefully tested before they become routine. They need to be shown to be accurate, dependable, and practical in real healthcare settings.
Even so, patients clearly recognised the potential. For them, this is not just about speed. It is about moving toward care that is:
- more responsive
- more personalised
- more convenient
- less anxiety-provoking
Common questions
Is this available now?
Usually not as a routine test in most healthcare settings. It is still being developed and studied, although interest in this type of monitoring is growing.
Will this replace ordinary blood tests?
Not immediately. Standard laboratory testing is still important. New bedside systems may first be used alongside existing methods while they are being evaluated and introduced.
Would this work for every antifungal drug?
Not necessarily. Some devices may be designed for specific drugs first. Wider use would depend on the technology and the evidence supporting it.
Could this be used at home?
Possibly one day, but that is likely to depend on how reliable, affordable, and easy to use the technology becomes. For now, clinic or bedside use is the more immediate possibility.
Why is a drop-of-blood test appealing to patients?
Because it may mean quicker answers, less uncertainty, fewer hospital trips, and more confidence that treatment decisions are based on what is happening in their own body.
When to seek medical advice
You should contact your healthcare team if you:
- develop new or worsening side effects from your antifungal medicine
- feel your treatment is not helping
- have concerns about drug interactions with other medicines
- are unsure whether to continue, stop, or change your medication
A new bedside test could support treatment decisions, but it would not replace medical advice. Symptoms, scans, blood tests, and clinical review would still matter.
Final thoughts
This new chip-based bedside technology may sound futuristic, but the reason patients welcomed it is very straightforward.
They saw the possibility of care that is faster, clearer, safer, and better adapted to real life.
In other words, this is about more than measuring a drug level from a drop of blood. It is about moving away from delayed, one-size-fits-all monitoring and toward real-time, personalised, patient-centred care.
In one sentence
A tiny chip and a drop of blood could help doctors adjust antifungal treatment more quickly — and patients believe that could make care less stressful, less burdensome, and more personal.
Author: Graham Atherton and ChatGPT draft support
For review by: National Aspergillosis Centre / relevant clinical or research reviewer
Note: This article is for general information and should not be used as a substitute for medical advice.
Understanding the Journey to Diagnosis (Start Here)
Last reviewed: 18 March 2026
Key points
- Aspergillosis can be difficult to diagnose because symptoms often overlap with more common illnesses.
- Long diagnostic journeys are common in many chronic and uncommon conditions, not just aspergillosis.
- Uncertainty does not mean symptoms are unreal.
- Diagnosis often becomes clearer over time, as symptoms, scans, tests, and patterns build up.
- Good communication, ongoing review, and specialist input can make a major difference.
Why we created this series
People living with aspergillosis often tell us that the hardest part was not only the illness itself, but the time before they had an answer. Many describe repeated symptoms, multiple appointments, courses of treatment that only partly helped, and a growing sense that something still did not fit.
This experience is common in conditions such as chronic pulmonary aspergillosis (CPA) and allergic bronchopulmonary aspergillosis (ABPA), but it is also seen much more widely across medicine.
We created this series to help make sense of that process in a way that is:
- clear and accessible for patients and carers
- respectful of clinicians working within complex systems
- honest about uncertainty, delay, and emotional impact
- useful for improving understanding on both sides
The articles in this series
1. Why Aspergillosis Is So Hard to Diagnose
This article explains why aspergillosis can be difficult to recognise, why medicine usually starts with more common explanations, and how diagnosis often depends on patterns building up over time rather than one single test.
Best for: understanding the overall process and why delays can happen.
2. When Symptoms Are Real but Answers Are Not: Understanding Uncertainty
This article looks at the emotional and communication side of the diagnostic journey, including why patients may feel unheard, what clinicians may mean when they speak cautiously, and why unexplained symptoms are still real symptoms.
Best for: understanding uncertainty, communication gaps, and the experience of not yet having answers.
3. Why Diagnosis Can Take Time — and Why You Are Not Alone
This article places the diagnostic journey in a wider context, showing that long paths to diagnosis are common in many chronic and complex conditions, and offering reassurance that patients are not alone in this experience.
Best for: reassurance, perspective, and recognising that this journey is shared by many others.
Who may find this series helpful
This series may be useful if you are:
- a patient with ongoing respiratory symptoms and no clear diagnosis yet
- a carer or family member trying to understand what your loved one is going through
- a person recently diagnosed with aspergillosis and looking back on a long journey
- a general practitioner, respiratory clinician, nurse, or allied health professional wanting to better understand the patient perspective
It may also help people with other chronic conditions, because many of the same themes—uncertainty, overlap of symptoms, repeated reassessment, and eventual recognition—are seen across a wide range of illnesses.
When to seek medical advice
Seek medical advice if you have ongoing or worsening symptoms that are not improving as expected, especially if they keep returning or no longer fit the original explanation.
Seek urgent medical advice if you have symptoms such as:
- coughing up blood
- rapidly worsening breathlessness
- new chest pain
- significant unexplained weight loss
- high fever or signs of severe infection
If you already have an underlying lung condition and your usual treatment no longer seems to be working, it is reasonable to ask whether the diagnosis needs to be reviewed.
Related resources
Why Diagnosis Can Take Time — and Why You Are Not Alone
Last reviewed: 18 March 2026
Key points
- Long diagnostic journeys are common in many chronic and rare conditions—not just aspergillosis.
- Delays do not mean your symptoms were not real or important.
- Diagnosis often becomes clear over time, as patterns develop.
- Many patients go through similar experiences before reaching answers.
- Specialist centres play an important role when conditions are complex.
You are not alone in this
One of the most important things to understand is this:
Long and difficult diagnostic journeys are common—especially in chronic or complex illness.
Many people living with conditions such as chronic pulmonary aspergillosis (CPA) or allergic bronchopulmonary aspergillosis (ABPA) describe months or years of symptoms before a clear diagnosis was made.
This experience, while frustrating, is not unusual—and it does not mean your care has failed.
This happens in many conditions
Aspergillosis is not unique in this respect.
Similar diagnostic journeys are seen in:
- chronic lung diseases
- autoimmune conditions
- rare infections
- conditions with overlapping symptoms (e.g. fatigue syndromes)
In all of these, the same pattern often occurs:
- early symptoms are non-specific
- common conditions are treated first
- tests may initially be normal
- the true pattern only becomes clear over time
Why time helps diagnosis
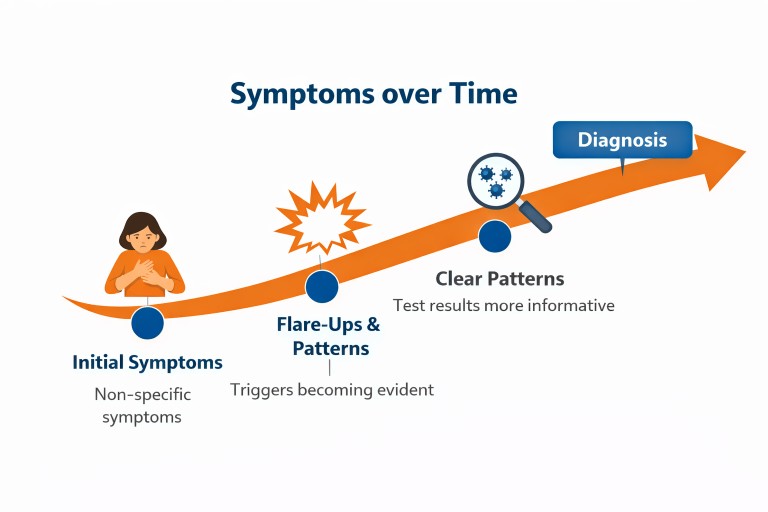
Although it can feel like delay, time often provides essential information.
Patterns emerge
- symptoms become more consistent
- flare-ups follow recognisable triggers
- response to treatment becomes clearer
Tests become more informative
- changes appear on imaging (e.g. CT scans)
- blood markers become more clearly abnormal
- microbiology results become more consistent
What was unclear early on can become much more visible later.
The turning point
Many patients describe a moment when things begin to change:
- symptoms no longer fit common conditions
- treatments stop working as expected
- a clinician recognises a pattern
This is often the point where less common diagnoses—including aspergillosis—are considered.
The role of specialist centres
Complex conditions are often best diagnosed in specialist centres with experience in that field.
In the UK, the National Aspergillosis Centre provides:
- expert multidisciplinary assessment
- access to specialist diagnostics
- experience in recognising patterns of disease
Referral to a specialist centre is often a key step in reaching a diagnosis.
The emotional impact
Long periods without answers can be deeply challenging.
Patients often describe:
- frustration and uncertainty
- feeling unheard or misunderstood
- loss of confidence in their own body
These reactions are entirely understandable.
Your experience is valid—even if the diagnosis took time to emerge.
Moving forward
Once a diagnosis is made, many patients feel a sense of relief—even if treatment is still needed.
A diagnosis provides:
- an explanation for symptoms
- a direction for treatment
- a clearer future plan
Even before diagnosis, it is important to remember:
You are still on a pathway—just not always a straight one.
Common questions
Does a long delay mean something was missed?
Not necessarily. Many conditions are only diagnosable once they have developed further.
Should I have been referred earlier?
Sometimes earlier referral is helpful, but it usually depends on how symptoms evolve over time.
Is this unique to aspergillosis?
No—this pattern is seen across many chronic and rare conditions.
Related pages
Why Aspergillosis Is So Hard to Diagnose
Last reviewed: 18 March 2026
Key points
- Aspergillosis is often difficult to diagnose because its symptoms can look very similar to those of more common conditions.
- Diagnosis usually depends on several pieces of evidence being brought together, rather than one simple test.
- Doctors are trained to consider common conditions first, because this is usually the safest and most efficient approach.
- This approach works well for many patients, but it can delay recognition of conditions such as aspergillosis.
- Delays are often caused by the way healthcare systems are organised, not by lack of care or effort from individual clinicians.
- Patients can help by keeping a clear record of symptoms, tests, treatments, and how things have changed over time.
Why diagnosis can be difficult
Aspergillosis is not a single illness but a group of conditions caused by Aspergillus, a mould commonly found in the environment. These include:
- Allergic bronchopulmonary aspergillosis (ABPA)
- Chronic pulmonary aspergillosis (CPA)
- Other airway and colonisation-related conditions
Diagnosis usually depends on combining:
- symptoms over time
- CT scan findings
- blood tests (including immunological tests)
- sputum microbiology
- clinical history
There is rarely a single “yes or no” test, which is why diagnosis can take time.
What the patient journey often looks like
Early symptoms
Symptoms such as cough, breathlessness, fatigue, or sputum are common across many conditions including bronchiectasis, asthma, and infection.
Treatment for common conditions
Initial treatment often includes antibiotics, inhalers, or steroids. These are appropriate first steps based on clinical guidelines such as those from the British Thoracic Society (BTS).
Ongoing symptoms
When symptoms persist or return, further investigation is usually needed.
The turning point
At some stage, fungal disease may be considered and tests for Aspergillus are performed.
Why doctors tackle common conditions first
Why do doctors start with common conditions?
Doctors treat common diseases first, prioritizing efficiency, patient safety, and high-probability outcomes. This approach, considering the most likely diagnosis first, helps manage patient health efficiently and effecctively before investigating rare or complex conditions.
This approach is safe and effective for most people, but conditions like aspergillosis can sit outside these usual pathways.
Where delays can happen
Overlap of symptoms
Symptoms overlap with many conditions, including tuberculosis and lung cancer.
No single definitive test
Diagnosis often requires combining multiple test results rather than relying on one.
Gradual disease progression
Conditions such as CPA may evolve over months or years.
Multiple conditions
Patients may have more than one lung condition at the same time.
Why this is often about the system, not the individual doctor
Healthcare systems are designed to manage large numbers of patients efficiently and safely. This means prioritising common conditions first.
However, aspergillosis often requires specialist input. In the UK, this may include referral to the National Aspergillosis Centre, which provides expert assessment and management.
International guidance from organisations such as ESCMID (European Society of Clinical Microbiology and Infectious Diseases) also highlights the complexity of fungal diseases.
What patients can do
- Keep a record of symptoms and treatments
- Ask when diagnosis should be reviewed
- Discuss whether further tests are needed
- Use trusted information sources such as our diagnosis guide
A more balanced way to think about delay
Diagnosis is often not a single event but a process that unfolds over time.
The goal is to recognise patterns earlier and ensure patients who need specialist input are identified sooner.
Common questions
Why was I treated for other conditions first?
Because those conditions are more common and more likely.
Should I ask about aspergillosis?
Yes, especially if symptoms are persistent or unusual—but it should be part of a broader discussion.
When to seek medical advice
- Persistent or worsening symptoms
- Coughing up blood
- Unexplained weight loss
References and further reading
When Symptoms Are Real but Answers Are Not: Understanding Uncertainty
Last reviewed: 18 March 2026
Key points
- Many people experience a period where symptoms are real but no clear diagnosis has been made.
- This can sometimes feel like being told “nothing is wrong” or “it is all in your head”.
- In most cases, this reflects uncertainty in the system, not disbelief from clinicians.
- Conditions such as aspergillosis can take time to become recognisable.
- Physical and psychological factors can overlap—but unexplained symptoms are still real symptoms.
- Clear communication and ongoing review are key to moving forward.
The experience many patients describe
During a long diagnostic journey, many people reach a point where they hear phrases such as:
- “Your tests are normal”
- “We can’t find anything serious”
- “It may be stress or anxiety contributing”
Even when these words are carefully chosen, they can feel like:
“Nothing is wrong” or “it’s all in my head”
This can be one of the most difficult parts of the journey—especially when symptoms are ongoing, disruptive, and clearly real.
Why this happens
This situation usually reflects the limits of current medical systems rather than a lack of concern.
Tests do not always give clear answers
Modern medicine relies heavily on tests. But for many conditions—including chronic pulmonary aspergillosis (CPA)—tests may:
- be negative early on
- show unclear or borderline results
- require interpretation over time
This creates a gap between:
- what the patient is experiencing
- what can currently be measured
Medicine is designed to provide answers
Clinicians are trained to explain symptoms and reassure patients. When no clear diagnosis is available, they may turn to explanations such as:
- stress
- anxiety
- functional symptoms
These are real and valid factors—but if introduced too early, they can feel like the search has stopped.
Time pressure
Short consultations can mean:
- less time to explain uncertainty
- less opportunity to validate patient experience
- simplified explanations that lose nuance
What doctors mean vs what patients hear

Often, the issue is not what is said—but how it is understood.
| What may be said | What may be heard |
|---|---|
| “Your tests are normal” | “Nothing is wrong” |
| “We haven’t found a cause yet” | “There is no cause” |
| “Stress may be contributing” | “It’s all in your head” |
Understanding this gap can help both patients and clinicians move forward more constructively.
A critical clarification
Not having a diagnosis is not the same as not having a disease.
Unexplained symptoms are still real symptoms.
Medicine does not always have immediate answers—especially for conditions that develop slowly or do not fit standard patterns.
Physical and psychological overlap
It is important to take a balanced view.
- Physical illness can lead to anxiety, fatigue, and distress
- Anxiety can worsen physical symptoms such as breathlessness
This relationship is two-way, not either/or.
The problem arises when psychological explanations replace further investigation, rather than sitting alongside it.
Why this matters in aspergillosis
Conditions such as aspergillosis often:
- develop gradually
- have non-specific symptoms
- require multiple tests over time
This makes periods of uncertainty more likely, particularly before a diagnosis such as allergic bronchopulmonary aspergillosis (ABPA) or CPA is confirmed.
You can read more about this in Why Aspergillosis Is So Hard to Diagnose.
What patients can do
- Keep a record of symptoms and how they change over time
- Ask what the current working diagnosis is
- Ask when the situation should be reviewed
- Share concerns clearly, but remain open to different explanations
Helpful questions include:
- “What else could this be?”
- “What would make you reconsider the diagnosis?”
- “When should we review this again?”
A shared understanding
This situation is not about:
- patients being dismissed
- clinicians not caring
It reflects a deeper reality:
Medicine does not always have immediate answers—especially for complex or evolving conditions.
The goal is to keep the process open, respectful, and moving forward.
Common questions
Does this mean doctors think I’m imagining symptoms?
No. It usually reflects uncertainty rather than disbelief.
Can stress really affect physical symptoms?
Yes—but this should not stop appropriate medical investigation.
What should I do if I feel dismissed?
Ask for clarification, a review plan, or a second opinion if needed.
Related pages
Aspergillosis Research Highlights - Week 11
Mid-March 2026 Literature Update
This update summarises notable recent publications mentioning aspergillosis, prioritising studies relevant to chronic pulmonary aspergillosis (CPA), allergic bronchopulmonary aspergillosis (ABPA), diagnostics, epidemiology and management.
Table of Contents
Key Highlights
- Pulmonary aspergilloma review published in CHEST: a substantial review summarises modern understanding of pulmonary aspergilloma, including diagnosis, haemoptysis risk and management.
- Rapid diagnostic technologies emerging: new CRISPR-based and LAMP molecular approaches show promise for faster identification of Aspergillus fumigatus and antifungal resistance.
- Nebulised antifungal therapy explored for ABPA: a case report describes inhaled antifungal therapy in a patient with treatment-limiting comorbidities.
- Microbiome disruption may increase susceptibility to Aspergillus infection: experimental data suggest antibiotics can impair neutrophil-mediated antifungal immunity.
- Global burden continues to emerge: recent studies again show Aspergillus infection in patients initially suspected of tuberculosis or pneumonia.
Clinical and Review Papers
Pulmonary Aspergilloma: Comprehensive Clinical Review
Seo C, Dumoulin E, Thornton CS.
Spore Wars: A Comprehensive Review of Pulmonary Aspergilloma and Its Clinical Management.
CHEST, 12 Mar 2026.
PubMed: https://pubmed.ncbi.nlm.nih.gov/41831523/
This review provides an updated overview of pulmonary aspergilloma, covering pathogenesis, imaging findings, haemoptysis risk and current management strategies.
- Aspergilloma develops in pre-existing lung cavities, often related to previous tuberculosis or other structural lung disease.
- The major complication is haemoptysis, which can be severe or life-threatening.
- Management may include monitoring, antifungal therapy, bronchial artery embolisation or surgical resection in selected patients.
Systematic Review Planned on ABPA Therapies
Nwankwo L, Maidment I, Periselneris J, Jackson DJ, Agarwal R, Asano K, Nuh A, Ni M, Shah A, Armstrong-James D.
Protocol for a systematic literature review and network meta-analysis of the evidence for therapies in allergic bronchopulmonary aspergillosis (ABPA).
Systematic Reviews, 11 Mar 2026.
PubMed: https://pubmed.ncbi.nlm.nih.gov/41814377/
This protocol sets out a planned network meta-analysis comparing therapies used in ABPA.
- Will assess the evidence for oral corticosteroids, antifungal therapy, biologics and combination approaches.
- Aims to address the continuing lack of strong comparative evidence between treatment strategies.
Diagnostics and Detection Advances
Rapid Antifungal Susceptibility Testing for Aspergillus fumigatus
Nozue S, Furuhashi K, Toguchi A, Ishikawa J, Nagura O, Yamashita K, Maekawa M, Iwaizumi M.
Rapid antifungal susceptibility testing for Aspergillus fumigatus using a loop-mediated isothermal amplification method.
Journal of Microbiological Methods, 12 Mar 2026.
PubMed: https://pubmed.ncbi.nlm.nih.gov/41831694/
This study describes a loop-mediated isothermal amplification (LAMP) method for rapid antifungal susceptibility testing.
- Designed to identify antifungal resistance more quickly than conventional culture-based testing.
- Potentially useful for early recognition of azole resistance.
CRISPR-Based Detection of Aspergillus fumigatus
Jiang Q, Zeng X, Zhang Q, Yang F, Lv T, Zhang Y, Wang J, Li F, Xu D.
Development and application of a rapid detection system for Aspergillus fumigatus based on ERA/CRISPR-Cas12a.
BMC Microbiology, 9 Mar 2026.
PubMed: https://pubmed.ncbi.nlm.nih.gov/41803678/
This paper reports a rapid molecular detection platform combining ERA amplification with CRISPR-Cas12a.
- Aims to detect A. fumigatus DNA rapidly and accurately.
- Represents the wider move toward faster molecular fungal diagnostics.
Sputum Galactomannan for Diagnosing IPA in COPD
Lan Y, Li H, Su D, Liao X, Zhang Q, Ma Q.
Clinical value of sputum galactomannan testing in the diagnosis of invasive pulmonary aspergillosis among chronic obstructive pulmonary disease patients.
European Journal of Medical Research, 9 Mar 2026.
PubMed: https://pubmed.ncbi.nlm.nih.gov/41803989/
This study evaluates sputum galactomannan as a less invasive diagnostic tool for invasive pulmonary aspergillosis in patients with COPD.
- Could provide useful diagnostic information when bronchoscopy is not feasible.
- May complement serum or bronchoalveolar lavage-based testing.
Immunology and Pathogenesis
Antibiotics and Susceptibility to Aspergillus Infection
Aufiero MA, Hohl TM.
Antibiotic-induced microbiota disruption impairs neutrophil-mediated immunity to respiratory Aspergillus fumigatus infection in mice.
mBio, 11 Mar 2026.
PubMed: https://pubmed.ncbi.nlm.nih.gov/41810941/
This experimental paper suggests that antibiotic-driven microbiome disruption can impair neutrophil responses and increase vulnerability to Aspergillus infection.
- Antibiotics altered microbial communities in ways that weakened antifungal immunity.
- The findings support a protective role for the microbiome in respiratory host defence.
Therapeutic Developments
Nebulised Antifungal Therapy in ABPA
Carrasco Sánchez M, Llopis Pastor E, García-Salmones Martín M.
Nebulised antifungal therapy in allergic bronchopulmonary aspergillosis in a patient with treatment-limiting comorbidities.
Medicina Clínica (Barcelona), 12 Mar 2026.
PubMed: https://pubmed.ncbi.nlm.nih.gov/41832814/
This case report describes use of nebulised antifungal therapy in ABPA where standard systemic treatment was limited by comorbidities.
- Suggests inhaled delivery may occasionally offer a practical workaround in selected patients.
- Evidence remains limited and this should still be viewed as a niche or exploratory approach.
Epidemiology and Public Health
Aspergillus Positivity in Patients With Tuberculosis-Like Symptoms
Ebong SMA, Kengne VN, Ayong MNA, Foko LPK, Ambono JLN, Ndzana GM, Baïdam MT, Youguitcha O, Abah OZA, Ayangma C, Koro FK.
Positivity rate of Aspergillus spp. in patients with tuberculosis-like symptoms in Yaoundé, Cameroon and antifungal resistance profile.
BMC Infectious Diseases, 9 Mar 2026.
PubMed: https://pubmed.ncbi.nlm.nih.gov/41803790/
This study found Aspergillus positivity in patients initially presenting with tuberculosis-like symptoms.
- Shows again how pulmonary aspergillosis can be confused with TB.
- Also assessed antifungal resistance patterns.
Pulmonary Fungal Infection in Pneumonia
Shokohi R, Mehraban Z, Darvishi F, Fatahinia M, Kiasat N.
Epidemiology and Clinical Features of Pulmonary Fungal Infections in Patients with Pneumonia: A Single-Center Study from Southwestern Iran.
Research Square preprint, 11 Mar 2026.
Preprint: https://www.researchsquare.com/article/rs-1164127
This preprint reports that aspergillosis was a significant component of pulmonary fungal infection among patients with pneumonia.
- Suggests fungal infection may be more common in severe respiratory disease than often recognised.
- As a preprint, findings should be interpreted cautiously until peer reviewed.
Aspergillosis in Complex Clinical Settings
Brain Abscess With Tuberculosis and Aspergillosis
Mirg S, Parihar J, Vibha D, Garg A, Singh G, Singh U, Sharma MC, Tripathi M.
Brain abscess with concurrent infection: tuberculosis and aspergillosis.
Practical Neurology, 13 Mar 2026.
PubMed: https://pubmed.ncbi.nlm.nih.gov/40784749/
This case report describes a rare brain abscess caused by concurrent tuberculosis and aspergillosis.
- Illustrates the complexity of diagnosis when multiple serious infections coexist.
- Highlights the need to keep fungal infection in the differential diagnosis.
Nosocomial Aspergillus calidoustus Infection in CAR-T Cell Therapy
Aubry A, Joris M, Choquet M, Kemp H, Bigot J, Braule B, Lemonnier D, Merlin-Brochart J, Lebon D, Maizel J, Guitard J, Chouaki T.
Nosocomial invasive Aspergillus calidoustus infection in a CAR-T cell-treated patient with concomitant Aspergillus fumigatus respiratory infection.
European Journal of Clinical Microbiology & Infectious Diseases, 12 Mar 2026.
PubMed: https://pubmed.ncbi.nlm.nih.gov/41817606/
This case highlights invasive infection by the rarer species Aspergillus calidoustus in a highly immunocompromised patient.
- Occurred alongside respiratory infection with A. fumigatus.
- Emphasises the diverse Aspergillus species that may affect profoundly immunosuppressed patients.
Additional Papers Mentioning Aspergillosis
These papers mention aspergillosis but are not primarily focused on it.
- Seffar L et al. Beyond Classification: An Antineutrophil Cytoplasmic Antibody-Associated Vasculitis Overlap Case. Cureus, 14 Mar 2026. PMC: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12983185/
- Jandric M et al. Continuous Renal Replacement Therapy for Patients With Sepsis in a Low-Resource Medical Intensive Care Unit (MICU): Incidence, Risk Factors, and Outcomes. Cureus, 14 Mar 2026. PMC: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12979951/
- Koh M et al. Understanding disease burden, challenges in current treatment strategies and call for action for management of severe asthma in Asia: a position statement from Asian respiratory experts. Frontiers in Allergy, 14 Mar 2026. PMC: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12979456/
- Mustafa J et al. Phytochemical profiling of Vitex negundo seeds via UHPLC-QTOF-MS/MS analyses with antimicrobial evaluation and in silico targeting of DNA Gyrase B and Secreted Aspartic Proteinase 2 (SAP2). PLoS One, 13 Mar 2026. PubMed: https://pubmed.ncbi.nlm.nih.gov/41824429/
- Mitchelmore P, Duggan S. Candida in the lung: Fact, fiction, friend or foe? PLoS Pathogens, 10 Mar 2026. PubMed: https://pubmed.ncbi.nlm.nih.gov/41805870/
- Xie Y, Zhang A, Wang Y, Wang R. Community-Acquired Pneumonia in Patients With Diabetes: Narrative Review. JMIR Diabetes, 10 Mar 2026. PubMed: https://pubmed.ncbi.nlm.nih.gov/41805689/
- Long MB et al. Design and rationale of the AIR-NET trial: a randomised, open-label, multifactorial, multicentre, adaptive platform trial using a range of repurposed anti-inflammatory treatments to improve outcomes in patients with bronchiectasis within the EMBARC clinical research network. ERJ Open Research, 9 Mar 2026. PubMed: https://pubmed.ncbi.nlm.nih.gov/41809869/
- Zablonski KG et al. Successful allogeneic stem cell transplant in a patient with a left ventricular assist device: a novel case report. Annals of Hematology, 11 Mar 2026. PubMed: https://pubmed.ncbi.nlm.nih.gov/41807548/
- He D et al. Amphotericin B promotes respiratory viral entry by enhancing late endosomal maturation and fusion via glucocerebrosidase-mediated ceramide remodeling. Nature Communications, 9 Mar 2026. PubMed: https://pubmed.ncbi.nlm.nih.gov/41803143/
Overall Trends
- Rapid molecular diagnostics for Aspergillus detection and resistance testing continue to advance.
- Global awareness of fungal lung disease is improving, especially in patients initially thought to have tuberculosis or bacterial pneumonia.
- Host immunity and microbiome research is expanding understanding of why some patients become vulnerable to Aspergillus disease.
- Alternative treatment approaches, such as inhaled antifungal therapy, are being explored in selected difficult cases.
Clinical Trials and Emerging Treatments for Chronic Aspergillosis
Last reviewed: 12 March 2026
Key points
- Only a small number of antifungal drug classes are currently available to treat aspergillosis.
- New treatments are needed because of drug resistance, side effects, drug interactions and the long-term burden of chronic disease.
- Research is now exploring not only new antifungal drugs, but also inhaled therapies, biologics, immune-modulating treatments and combination approaches.
- Most new drugs are first tested in invasive aspergillosis before being studied in chronic pulmonary aspergillosis (CPA) or allergic bronchopulmonary aspergillosis (ABPA).
- Clinical trials are essential for showing whether new treatments are safe and effective.
Table of contents
- Overview
- Why new treatments are needed
- How new treatments are developed
- Clinical trial phases
- Regulatory approval and NHS use
- New antifungal drugs in development
- Other emerging treatments for chronic aspergillosis
- Why new treatments are often tested in invasive aspergillosis first
- How to find clinical trials
- Common questions
- Further information
- Author and review information
Overview
Treatment options for aspergillosis have improved over time, but there are still important limitations. Only a small number of antifungal drug classes are available, some fungi develop resistance to existing medicines, and some patients cannot tolerate treatment because of side effects or drug interactions.
This is particularly important in chronic aspergillosis, where treatment may need to continue for months or years. Research is therefore focused not only on new antifungal drugs, but also on better drug delivery systems, immune-based treatments, biologic therapies and combinations of treatments.
Clinical trials are the main way that researchers test whether these new approaches are safe and effective.
Why new treatments are needed
New treatments for chronic aspergillosis are needed for several reasons:
- the number of available antifungal drug classes is limited,
- Aspergillus can develop resistance to azole antifungals,
- some patients experience significant side effects or important drug interactions,
- long-term treatment can be difficult to sustain,
- chronic disease may continue to affect symptoms, lung function and quality of life even when treatment is helping.
Because chronic pulmonary aspergillosis (CPA), allergic bronchopulmonary aspergillosis (ABPA) and related conditions can behave differently, researchers are exploring a wider range of therapies than in the past.
How new treatments are developed
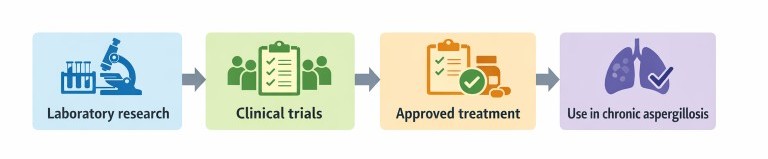
Before a new treatment can be used routinely, it must pass through several stages of development.
| Stage | Description |
|---|---|
| Basic research | Scientists study the disease and identify targets that could be affected by a new drug or treatment approach. |
| Drug discovery | Researchers screen chemical compounds or modify promising molecules to find potential treatments. |
| Pre-clinical studies | Potential treatments are tested in laboratory systems and sometimes animal models to assess activity and safety. |
| Application to begin human trials | Researchers apply to regulators and ethics committees for permission to test the treatment in people. |
Clinical trial phases
Clinical trials are usually carried out in phases.
| Phase | Purpose |
|---|---|
| Phase 0 / Phase I | Small studies, often in healthy volunteers, to understand how the treatment behaves in the body, including absorption, distribution and safe dose ranges. |
| Phase II | Studies in patients with the disease to identify useful doses and gather early information on effectiveness and side effects. |
| Phase III | Larger studies comparing the new treatment with existing care to assess effectiveness and safety more accurately. |
| Phase IV | Post-marketing studies that monitor how the treatment performs in real-world use after approval. |
Regulatory approval and NHS use
If a treatment performs well in trials, the manufacturer can apply for approval from a medicines regulator such as the European Medicines Agency (EMA) or the U.S. Food and Drug Administration (FDA).
In the United Kingdom, a treatment may also be assessed by the National Institute for Health and Care Excellence (NICE) to determine whether it should be funded for routine use in the NHS.
Even when a drug is not recommended for routine NHS use, doctors may sometimes apply for individual funding if they believe it could benefit a particular patient.
New antifungal drugs in development
Many new antifungal drugs are first developed for severe invasive fungal infections and may later be studied in chronic conditions such as CPA or ABPA.
Olorofim
Olorofim is a novel antifungal from a completely new class called the orotomides. It targets fungal pyrimidine synthesis, a pathway not affected by current azole, echinocandin or polyene antifungals.
Rezafungin
Rezafungin is an echinocandin designed to have a longer duration of action and improved pharmacokinetic properties compared with older drugs in the same class.
Ibrexafungerp
Ibrexafungerp belongs to a new group of antifungals called triterpenoids. It acts on fungal cell wall synthesis in a way that is similar to echinocandins, but its structure is different and it can be given orally.
Fosmanogepix
Fosmanogepix is a first-in-class antifungal that blocks production of a molecule needed for fungal cell wall construction and self-regulation.
Oteseconazole
Oteseconazole is one of the newer tetrazole agents designed to improve selectivity and reduce side effects compared with traditional azoles.
Encochleated Amphotericin B
This is a reformulated version of amphotericin B designed to improve delivery and reduce toxicity.
ATI-2307
ATI-2307 is an arylamidine antifungal that interferes with mitochondrial function in fungal cells.
Other emerging treatments for chronic aspergillosis
Although new antifungal drugs are an important area of research, scientists are also exploring other ways to treat chronic forms of aspergillosis such as chronic pulmonary aspergillosis (CPA) and allergic bronchopulmonary aspergillosis (ABPA). These approaches aim to improve treatment effectiveness, reduce side effects, or target the immune response to the fungus.
Inhaled antifungal treatments
One area of research is the development of antifungal medicines that can be delivered directly to the lungs using inhalers or nebulisers. Delivering medication directly to the lungs may allow higher drug concentrations at the site of disease while reducing side effects elsewhere in the body.
Examples being explored include inhaled or nebulised formulations of amphotericin B, itraconazole and voriconazole.
Immune-modulating therapies
In some patients with aspergillosis, the immune response to the fungus plays an important role in how the disease develops or persists. Researchers are studying treatments that help modify the immune response rather than directly killing the fungus.
Examples include therapies that may enhance antifungal immunity or reduce harmful inflammation.
Biologic therapies
Biologic drugs that target specific immune pathways are already used to treat severe asthma and allergic disease. Some of these medicines are now being studied or used in fungal-related airway disease.
Examples include drugs targeting immunoglobulin E (IgE) or eosinophilic inflammation, such as omalizumab, mepolizumab, benralizumab, dupilumab and tezepelumab. These may be particularly relevant in ABPA or severe asthma with fungal sensitisation (SAFS).
Combination therapies
Future treatment strategies may combine different approaches, for example antifungal medication together with biologic therapy, inhaled therapy or immune-modulating treatment. Combination treatment may improve outcomes in patients whose disease does not respond fully to a single treatment alone.
Research is ongoing to determine which combinations are most effective and safest for patients with chronic aspergillosis.
Why new treatments are often tested in invasive aspergillosis first
Many new antifungal drugs are first tested in patients with invasive aspergillosis before being studied in chronic forms of the disease such as chronic pulmonary aspergillosis (CPA) or allergic bronchopulmonary aspergillosis (ABPA).
There are several reasons for this:
- Disease progression is faster. In invasive infections the illness progresses quickly, so researchers can more easily measure whether a new treatment is working.
- Clearer treatment outcomes. Invasive infections often have well-defined clinical outcomes such as survival or clearance of infection.
- Smaller studies can provide useful results. Because invasive infections are severe, treatment effects may be detected in smaller numbers of patients.
Chronic forms of aspergillosis usually progress more slowly. This means clinical trials often need to run for longer periods and include larger numbers of patients to demonstrate whether a treatment is effective.
Once a new antifungal drug has shown benefit in invasive disease, researchers may then study how it performs in chronic conditions such as CPA or ABPA.
Specialist centres such as the National Aspergillosis Centre contribute to research that helps evaluate new treatments for chronic forms of aspergillosis and improve care for patients living with these conditions.
How to find clinical trials
Clinical trials involving human participants must be registered publicly for ethical and regulatory reasons.
You can search for ongoing or completed studies at:
This database allows you to search for studies by disease, location, treatment or trial status.
Not all studies test new drugs. Some trials investigate diagnostics, biomarkers, new ways of using existing medicines, or observational registries that help researchers understand disease patterns over time.
If you are interested in taking part in a clinical trial, discuss this with your specialist respiratory team.
Common questions
Why are new treatments needed if antifungal drugs already exist?
Current antifungal drugs help many patients, but they do not work for everyone. Some fungi develop resistance, some patients experience side effects or interactions, and chronic disease can remain difficult to control.
Are all new treatments new antifungal drugs?
No. Research now includes new antifungal drugs, inhaled treatments, biologics, immune-modulating therapies and combination approaches.
Why are there more trials in invasive aspergillosis than CPA?
Invasive aspergillosis progresses more quickly, so trial results can often be measured sooner and with fewer patients. CPA usually changes more slowly, which makes trials longer and more difficult to run.
Can patients with chronic aspergillosis join clinical trials?
Sometimes, yes. Eligibility depends on the trial design, the type of aspergillosis, previous treatment and other health factors. Your specialist team can advise whether there may be suitable studies.
Do clinical trials always involve testing a completely new drug?
No. Some studies test new doses, new combinations, new formulations such as inhaled treatment, or new diagnostic approaches.
Further information
You may find these pages helpful:
- Antifungal medicines for aspergillosis
- Biologics and eosinophilic asthma
- Inhalers and nebulisers
- Chronic pulmonary aspergillosis (CPA)
- Allergic bronchopulmonary aspergillosis (ABPA)
More information about the medicine development process can be found through major medicines regulators, clinical trial registries, and specialist respiratory teams.
Author and review information
Author: Aspergillosis Website Editorial Team
Audience: Patients, carers, GPs and non-specialists
Last reviewed: 12 March 2026