Clinical Trials and Emerging Diagnostics for Aspergillus Infections
How new diagnostic technologies may improve detection of Aspergillus lung infections
Last reviewed: March 2026
Author: Graham Atherton
Key points
- Diagnosing Aspergillus lung infections can be challenging because symptoms often resemble other lung diseases.
- Doctors usually combine CT scans, blood tests and microbiology tests to make a diagnosis.
- Researchers are developing new diagnostics that detect fungal DNA, fungal molecules, or drug-resistant strains.
- Emerging tools include PCR tests, antigen detection, rapid tests, sequencing technologies and artificial intelligence imaging.
- This page focuses on Aspergillus infections rather than allergic conditions such as Allergic Bronchopulmonary Aspergillosis, Severe Asthma with Fungal Sensitisation, or Aspergillus bronchitis.
- Many of these newer technologies are being studied in clinical trials and research programmes worldwide.
Table of contents
- 1. Important note: infection vs allergy
- 2. Why better diagnostics are needed
- 3. How Aspergillus infections are diagnosed today
- 4. Detecting the immune response vs detecting the fungus
- 5. Simple diagram: two ways of looking for Aspergillus disease
- 6. DNA testing (PCR diagnostics)
- 7. Fungal antigen detection
- 8. Rapid diagnostic tests
- 9. Breath diagnostics
- 10. Sequencing technologies and the lung microbiome
- 11. Detecting antifungal resistance
- 12. Artificial intelligence and CT imaging
- 13. Flowchart: how doctors diagnose chronic Aspergillus infection
- 14. The future diagnostic pathway
- 15. Common questions patients ask
- 16. When to seek medical advice
- 17. References
1. Important note: infection vs allergy
This article focuses on diagnosing Aspergillus infections of the lungs, particularly:
- Chronic Pulmonary Aspergillosis
- Aspergillus nodules
- Aspergilloma (fungal ball)
- Subacute invasive aspergillosis
These conditions involve the fungus growing in lung tissue or in pre-existing lung cavities.
This is different from allergic Aspergillus disease, such as:
- Allergic Bronchopulmonary Aspergillosis (ABPA)
- Severe Asthma with Fungal Sensitisation (SAFS)
- Aspergillus bronchitis (which overlaps with airway disease rather than the classic cavity-forming infections discussed here)
In allergic disease, diagnosis focuses more on immune reactions, such as IgE antibodies, eosinophils, allergy testing and markers of Type 2 inflammation.
This page therefore focuses mainly on diagnostics for Aspergillus infection rather than allergy. A separate article can cover immune diagnostics in allergic Aspergillus disease.
2. Why better diagnostics are needed
Chronic Pulmonary Aspergillosis often develops in people who already have damaged lungs, for example from:
- tuberculosis
- Chronic Obstructive Pulmonary Disease
- bronchiectasis
- sarcoidosis
- previous severe lung infections
- lung surgery
Symptoms may include:
- chronic cough
- breathlessness
- fatigue
- weight loss
- coughing up blood
- chest discomfort
These symptoms are not specific, meaning they can occur in many other lung diseases.
Aspergillus is also very common in the environment, so detecting it in sputum does not always mean it is causing disease. Sometimes it may simply be present without invading tissue or causing progressive damage.
For these reasons, diagnosing chronic aspergillosis usually requires multiple tests interpreted together, rather than relying on one result alone.
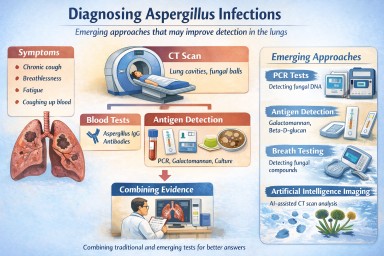
3. How Aspergillus infections are diagnosed today
Doctors usually combine three main types of evidence.
| Diagnostic method | What it shows |
|---|---|
| CT scan | Structural lung changes such as cavities, nodules or fungal balls |
| Blood tests | The body's immune response to Aspergillus |
| Microbiology tests | Evidence of the fungus itself |
The most important blood test for many patients with Chronic Pulmonary Aspergillosis is Aspergillus IgG antibodies.
CT scans are also critical because they can reveal features such as:
- lung cavities
- fungal balls
- nodules
- thickened cavity walls
- progressive lung destruction or scarring
Microbiology may include sputum culture, bronchoscopy samples, microscopy, molecular testing and antigen detection.
No single test is perfect. Doctors usually look at the whole picture: symptoms, scan findings, blood tests, microbiology, and how things change over time.
4. Detecting the immune response vs detecting the fungus
Some diagnostic tests detect how the body reacts to Aspergillus, while others try to detect the fungus itself.
Immune response tests
Examples include:
- Aspergillus IgG antibodies
- general inflammatory markers
These tests show that the immune system has encountered Aspergillus, but they do not always prove that the fungus is currently active, growing, or causing ongoing damage.
Direct fungal detection
Other tests look for components of the fungus itself.
| Test | What it detects |
|---|---|
| PCR | Aspergillus DNA |
| Galactomannan | Fungal cell wall molecules |
| Beta-D-glucan | Fungal structural components |
| Culture | Growth of the fungus in the laboratory |
These tests can sometimes provide more direct evidence that fungal material is present.
Why not rely only on the immune response?
Immune-response tests are extremely useful, but they are indirect. They tell us what the body is doing, not necessarily what the fungus is doing at that moment.
Antibodies may remain raised for quite a long time, may change only slowly, and may vary from person to person. Some patients also produce weaker immune responses than others.
By contrast, tests that detect fungal DNA or fungal cell wall components may sometimes give a better sense that fungal material is actually present in the lungs.
The footprints analogy
A useful way to understand this difference is:
- Immune response tests are like seeing footprints in the snow – they show that someone has been there.
- Direct fungal tests are like seeing the person themselves – they show that the organism is present.
In practice, doctors usually combine both kinds of evidence to make the diagnosis more reliable.
5. Simple diagram: two ways of looking for Aspergillus disease
Two broad diagnostic approaches
| Approach | Examples | Main question |
|---|---|---|
| Looking for the body's response | Aspergillus IgG, inflammation markers | Has the immune system reacted to Aspergillus? |
| Looking for the fungus itself | PCR, galactomannan, Beta-D-glucan, culture | Is fungal material present? |
| Looking for damage caused in the lungs | CT scan | Has Aspergillus or another disease caused structural lung change? |
This is why diagnosis is usually based on a combination of evidence rather than any single test.
6. DNA testing (PCR diagnostics)
Polymerase Chain Reaction, usually shortened to PCR, detects the DNA of Aspergillus fungi.
These tests can be performed on samples such as:
- sputum
- bronchoscopy samples
- lung tissue
Advantages of PCR include:
- faster results than fungal culture
- detection of very small amounts of fungus
- possible detection even when cultures are negative
However, PCR also has challenges. It may detect fungal material even when it is not clearly causing disease, and methods are not yet fully standardised across all laboratories.
Even so, PCR is one of the most important emerging tools in fungal diagnostics and is increasingly used in specialist centres.
7. Fungal antigen detection
Some tests look for molecules released by fungal cells. These are often called antigen tests.
Galactomannan
Galactomannan is a molecule found in the cell wall of Aspergillus.
It is already widely used in diagnosing invasive aspergillosis and is being studied more closely in chronic forms of disease as well.
Beta-D-glucan
Beta-D-glucan is a structural component found in the cell walls of many fungi.
Raised levels can suggest fungal infection somewhere in the body, although it is not specific for Aspergillus alone.
Researchers are interested in how these markers might be used alongside CT, antibody testing and PCR to improve accuracy.
8. Rapid diagnostic tests
Researchers are also developing rapid antigen tests that can detect fungal molecules within minutes.
These tests work in a similar way to a simple strip test and may offer:
- quick results
- minimal laboratory equipment
- possible use in clinics with fewer resources
These tests are promising, but they still need careful evaluation to show how accurate and reliable they are in real patients with chronic Aspergillus disease.
9. Breath diagnostics
Fungi release small chemicals called volatile organic compounds. Some experimental devices aim to detect these compounds in exhaled breath.
If successful, breath testing could provide:
- completely non-invasive testing
- rapid screening
- repeat testing over time without invasive procedures
This remains an early and experimental field, but it is attractive because it could make testing much easier for patients.
10. Sequencing technologies and the lung microbiome
Modern sequencing technologies can analyse all microbial DNA in a sample.
This means they may identify:
- fungi
- bacteria
- viruses
These approaches may help doctors understand mixed infections and the broader lung microbiome, especially in people with bronchiectasis or complex chronic lung disease.
In the future, sequencing may help explain why some patients have persistent symptoms or repeated flares involving more than one organism.
11. Detecting antifungal resistance
Some strains of Aspergillus fumigatus have developed resistance to azole antifungal drugs.
Newer diagnostic tests can detect genetic mutations linked to drug resistance, especially in the CYP51A gene.
Earlier detection of resistance could help doctors:
- choose effective antifungal drugs sooner
- avoid ineffective treatment
- improve long-term outcomes
This is an important area of research because antifungal resistance is a growing international concern.
12. Artificial intelligence and CT imaging
Artificial intelligence is increasingly being explored as a way to analyse CT scans more precisely.
These systems may eventually help doctors:
- detect subtle lung changes earlier
- measure cavity size or progression more consistently
- monitor disease over time
- compare scans more accurately
Artificial intelligence is unlikely to replace expert radiologists or specialist teams, but it may become a useful support tool.
13. Flowchart: how doctors diagnose chronic Aspergillus infection
Typical diagnostic pathway
Symptoms
persistent cough, breathlessness, fatigue, weight loss, coughing up blood
↓
CT scan
looking for cavities, fungal balls, nodules, scarring or progressive damage
↓
Blood tests
especially Aspergillus IgG and general inflammatory markers
↓
Microbiology
sputum, bronchoscopy, culture, PCR, antigen tests
↓
Clinical interpretation
combining symptoms, imaging, blood tests and microbiology
↓
Diagnosis and follow-up
deciding whether this is infection, colonisation, another lung condition, or mixed disease
This flowchart is simplified, but it shows the basic principle: diagnosis depends on combining evidence.
14. The future diagnostic pathway
In the future, diagnosing Aspergillus infection may involve several rapid tests used together.
A possible pathway could look like this:
Symptoms → CT scan → fungal antigen test → PCR confirmation → resistance testing
This approach could allow:
- faster diagnosis
- more personalised treatment
- earlier detection of complications
- better treatment monitoring
The aim is not necessarily to replace older tests, but to make the whole diagnostic process earlier, clearer and more precise.
15. Common questions patients ask
Why can Aspergillus infection take time to diagnose?
Because no single test can confirm the disease on its own. Doctors usually need to combine scan findings, blood tests, microbiology and the clinical history.
Why do some tests detect antibodies while others detect the fungus?
Antibody tests show how the immune system has reacted. Molecular and antigen tests try to show whether fungal material is present. Both are useful, but they answer slightly different questions.
Why is a CT scan so important?
A CT scan shows whether there is structural lung damage such as cavities, nodules or fungal balls. This helps doctors judge whether Aspergillus is likely to be causing disease rather than simply being present.
Are these new diagnostics available now?
Some are already used in specialist centres, but many are still being evaluated in clinical trials and research studies.
Will these newer tests replace existing blood tests?
Probably not completely. More likely, they will be used alongside existing tests to improve accuracy and speed.
16. When to seek medical advice
You should seek medical advice if you have symptoms such as:
- persistent cough
- worsening breathlessness
- unexplained fatigue
- weight loss
- coughing up blood
- new or worsening chest pain
These symptoms can have many causes, but they should be assessed properly, especially if you already have underlying lung disease.
17. References
- Denning DW, Cadranel J, Beigelman-Aubry C, Ader F, Chakrabarti A, Blot S, Ullmann AJ, Dimopoulos G, Lange C; European Society for Clinical Microbiology and Infectious Diseases and European Respiratory Society. Chronic pulmonary aspergillosis: rationale and clinical guidelines for diagnosis and management. Eur Respir J. 2016 Jan;47(1):45-68. doi: 10.1183/13993003.00583-2015. PMID: 26699723.
- Kosmidis C, Denning DW. The clinical spectrum of pulmonary aspergillosis. Thorax. 2015 Mar;70(3):270-7. doi: 10.1136/thoraxjnl-2014-206291. Epub 2014 Oct 29. PMID: 25354514.
- Takazono T, Izumikawa K. Recent Advances in Diagnosing Chronic Pulmonary Aspergillosis. Front Microbiol. 2018 Aug 17;9:1810. doi: 10.3389/fmicb.2018.01810. PMID: 30174658; PMCID: PMC6107790.
Author and review information
This page is intended for patients, carers, and non-specialist clinicians. It provides general educational information and should not replace individual medical advice.
Managing fatigue and energy in aspergillosis and allergic fungal lung disease
Key points
- Many people with aspergillus-related lung conditions experience extreme tiredness after physical or mental exertion.
- This fatigue can last hours or even several days.
- Breathing, immune activity, inflammation and sometimes hormone imbalance all use extra energy.
- Activity can temporarily increase airway inflammation and mucus.
- Managing energy carefully (“spoon theory”) can help prevent fatigue crashes.
Contents
- Why people with lung disease feel exhausted after exertion
- What is happening in the lungs and body
- Managing energy: the “spoon theory”
- Practical ways to manage energy
- Nutrition and adrenal health
- Warning signs you are running out of energy
- Why fatigue in lung disease is different from normal tiredness
- Daily energy management checklist
- Can this fatigue be treated?
- When to talk to your doctor
Why people with lung disease feel exhausted after exertion
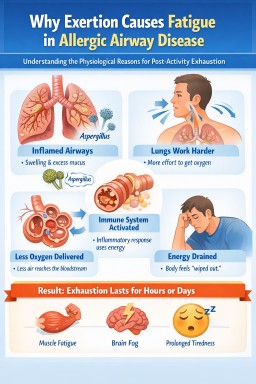
Healthy lungs have a large reserve capacity. When we exercise or do physical work, breathing becomes deeper and faster, but the lungs can usually cope easily.
In people with aspergillosis or allergic airway disease, the airways may already be:
- inflamed
- narrowed
- filled with mucus
- sensitive to allergens such as Aspergillus fumigatus
When the body demands more oxygen during activity, the lungs must work much harder to supply it. Activities that seem minor to other people may therefore require much greater effort from the body.
What is happening in the lungs and body?
Inflamed airways
Many aspergillus-related conditions involve inflammation in the airways. The immune system releases chemicals that cause:
- swelling of airway walls
- increased mucus production
- greater airway sensitivity
During exertion, breathing becomes faster and deeper, which can irritate these inflamed airways further.
Mucus affecting airflow
Inflamed airways often produce extra mucus. This mucus can partly block airflow and lead to:
- coughing
- wheezing
- breathlessness
- uneven airflow within the lungs
This may reduce how efficiently oxygen enters the bloodstream. Doctors sometimes refer to this as ventilation–perfusion mismatch.
Breathing uses more energy
In healthy people, breathing uses only a small fraction of the body's energy. In lung disease, breathing may require much more effort.
Additional muscles may assist breathing, including:
- chest muscles
- neck muscles
- shoulder muscles
These muscles can become fatigued during activity, just like leg muscles after exercise.
Immune system activity
If the immune system reacts to fungal proteins or allergens, it releases signalling chemicals called cytokines. These chemicals can produce symptoms similar to mild illness, including fatigue, brain fog and muscle aches.
Delayed inflammation after activity
Some people notice that fatigue appears later rather than immediately. Physical effort can trigger inflammation that develops over 12–48 hours, increasing mucus production, airway irritation and tiredness.
This explains why people sometimes feel worse the day after a busy day.
Managing energy: the “spoon theory”
Many people with chronic illness find it helpful to think about their energy using the idea of spoon theory.
In this idea:
- each spoon represents a small unit of energy
- you start the day with a limited number of spoons
- each activity uses some of those spoons
Because breathing and inflammation already use energy, people with lung disease may begin the day with fewer spoons available.
Example of spoon use
| Activity | Possible energy use |
|---|---|
| Getting dressed | 1 spoon |
| Showering | 2–3 spoons |
| Cooking a meal | 2 spoons |
| Doctor’s appointment | 3–4 spoons |
| Busy social day | Many spoons |
If too many spoons are used early in the day, the body may run out of energy, leading to exhaustion lasting hours or even days.
Practical ways to manage energy
Plan activities around your best time of day
| Time of day | Suggested activities |
|---|---|
| Morning | Errands or appointments |
| Midday | Light household tasks |
| Afternoon | Quieter activities |
| Evening | Rest and recovery |
Break tasks into smaller steps
Large tasks can overwhelm the lungs and muscles. Instead of doing everything at once:
- clean one room at a time
- cook in stages
- prepare things earlier in the day
Use the 50–70% rule
Try to stop activity when you reach about half to two-thirds of your limit. Stopping early often prevents the fatigue crash that can occur later.
Use breathing techniques
Pursed-lip breathing
- breathe in through your nose
- breathe out slowly through gently pursed lips
Rhythmic breathing
Match breathing with movement, for example when climbing stairs.
Keep mucus moving
Mucus increases the work of breathing. Helpful strategies include:
- airway clearance techniques
- staying well hydrated
- gentle movement
- using inhalers or nebulisers as prescribed
Maintain gentle regular activity
Although exertion can cause fatigue, complete inactivity can worsen the problem. Gentle activity such as walking or pulmonary rehabilitation exercises helps maintain muscle strength.
Protect sleep
- maintain a regular sleep routine
- clear mucus before bedtime if needed
- avoid heavy exertion late in the evening
Nutrition and adrenal health
Nutrition and energy
Good nutrition helps support energy levels. Helpful strategies include:
- eating regular meals
- including protein for muscle repair (eggs, fish, dairy, beans or nuts)
- eating complex carbohydrates for steady energy
- drinking enough fluids
Some people find that smaller, more frequent meals reduce breathlessness compared with large meals.
Important nutrients
- protein
- vitamin D
- iron
- B vitamins
Doctors may check for deficiencies if fatigue is severe.
Adrenal insufficiency
Some patients who have taken long-term steroid medications may develop adrenal insufficiency. The adrenal glands normally produce cortisol, which helps regulate energy and stress responses.
Symptoms may include:
- severe fatigue
- dizziness
- muscle weakness
- difficulty recovering after exertion
Patients with adrenal insufficiency usually take hydrocortisone replacement therapy and should follow their doctor’s advice carefully.
Warning signs you are running out of energy
- breathing becomes faster or more difficult
- increased coughing or mucus
- arms or legs feel heavy
- dizziness or weakness
- difficulty concentrating
- chest tightness or wheezing
When these warning signs appear, it is usually best to stop and rest before continuing.
Why fatigue in lung disease is different from normal tiredness
Fatigue in lung disease is not simply normal tiredness. Several factors occur at the same time:
- breathing requires more energy
- the immune system may be active
- oxygen exchange may be less efficient
- nutrition and hormone balance may influence recovery
Because of this combination, fatigue may appear suddenly and last longer than expected.
Daily energy management checklist
Pacing and activity
- spread activities across the day
- stop before exhaustion
- plan demanding tasks when energy is highest
- allow recovery time after busy days
Breathing and airway care
- use breathing techniques during exertion
- perform airway clearance if needed
- take inhalers or nebulisers as prescribed
- stay well hydrated
Nutrition and medication
- eat regular meals
- include protein for muscle strength
- take medications as prescribed
- follow sick-day rules if you have adrenal insufficiency
Sleep and recovery
- maintain a regular sleep routine
- clear mucus before bedtime
- rest when warning signs appear
Can this fatigue be treated?
Fatigue associated with aspergillosis, allergic fungal airway disease, or severe asthma can sometimes be improved when the underlying causes are treated. Because several different processes contribute to fatigue, treatment usually focuses on improving multiple factors rather than a single cure.
Treating airway inflammation
Inflammation in the airways is one of the major contributors to fatigue. When the airways are inflamed:
- breathing requires more effort
- mucus production increases
- oxygen exchange becomes less efficient
Treatments aimed at reducing airway inflammation may include:
- Inhaled corticosteroids – commonly used in asthma to reduce inflammation directly in the airways.
- Antifungal therapy – in some patients, reducing fungal growth can reduce immune activation and inflammation.
- Biologic therapies – newer treatments that target specific immune pathways involved in allergic and inflammatory lung disease.
Biologic treatments
Biologics are one of the most promising areas of treatment for severe asthma and allergic airway disease. These medications target specific parts of the immune system that drive inflammation.
| Biologic | Target | Effect |
|---|---|---|
| Omalizumab | IgE | Reduces allergic inflammation |
| Mepolizumab / Benralizumab | IL-5 pathway | Reduces eosinophilic inflammation |
| Dupilumab | IL-4 / IL-13 | Reduces type-2 inflammation |
| Tezepelumab | TSLP | Blocks upstream inflammatory signalling |
Some patients with conditions such as allergic bronchopulmonary aspergillosis (ABPA) or severe asthma report improvements in breathlessness, symptoms and overall energy levels when inflammation is better controlled.
Improving mucus clearance
Mucus in the airways increases the work of breathing and can contribute to fatigue. Strategies that may help include:
- airway clearance techniques
- physiotherapy
- maintaining good hydration
- using prescribed inhalers or nebulisers correctly
Treating other contributing factors
Fatigue can also be worsened by other health issues that are common in chronic lung disease, such as:
- iron deficiency
- vitamin deficiencies
- poor sleep
- adrenal insufficiency
- muscle deconditioning
Addressing these factors can sometimes improve overall energy levels.
Pulmonary rehabilitation
Pulmonary rehabilitation programmes combine exercise training, breathing techniques and education about pacing activities. These programmes can improve muscle efficiency and exercise tolerance, and many patients report reduced fatigue and improved quality of life.
Future treatments
Research into inflammatory lung diseases is advancing rapidly. New biologic drugs and other targeted therapies are being developed that may improve control of airway inflammation.
Researchers are also studying how the lung microbiome (bacteria and fungi living in the airways) influences inflammation. In the future, this may lead to more personalised treatments for patients with fungal-related lung disease.
Inflammatory fatigue
Researchers are increasingly recognising that chronic inflammatory diseases can cause a form of fatigue sometimes called “inflammatory fatigue.”
In these conditions, immune signalling chemicals released during inflammation can affect the brain and energy metabolism. Similar patterns of fatigue are seen in diseases such as rheumatoid arthritis and inflammatory bowel disease.
This may help explain why some treatments that reduce inflammation — including biologic therapies — can improve fatigue even when lung function measurements change only modestly.
Interrupting the fatigue cycle
Treatment aims to interrupt the cycle that can develop in chronic lung disease:
Airway inflammation
↓
Breathing requires more effort
↓
More energy used by respiratory muscles
↓
Immune system activity
↓
Reduced overall energy and fatigue
By improving airway inflammation, mucus clearance, muscle strength and overall health, many patients find their energy levels become more manageable.
When to talk to your doctor
Seek medical advice if:
- fatigue becomes progressively worse
- breathlessness increases
- new symptoms appear such as chest pain or coughing up blood
- fatigue prevents normal daily activities
A reassuring message
Many people with aspergillosis or allergic airway disease worry that exhaustion means their condition is worsening.
In most cases it reflects the extra energy required for breathing, inflammation and immune activity. Learning to pace activity can help people live more comfortably with chronic lung disease.
Author: National Aspergillosis Centre information team
Review: Clinical review recommended
Last reviewed: 2026
Nontuberculous Mycobacteria (NTM–MAC) and Aspergillosis
Why these infections sometimes occur together
Audience: Aspergillosis patients, carers, GPs and non-specialist clinicians
Some patients with Allergic Bronchopulmonary Aspergillosis (ABPA) may be investigated for nontuberculous mycobacteria (NTM), because airway damage from ABPA can increase susceptibility to other lung infections.
Key points
- Nontuberculous mycobacteria (NTM) are environmental bacteria that sometimes infect damaged lungs.
- The most common NTM causing lung disease is the Mycobacterium avium complex (MAC).
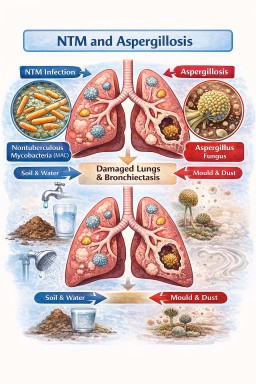
- NTM infection and aspergillosis often occur in the same patients because both thrive in damaged airways such as bronchiectasis or lung cavities.
- Some patients with ABPA are investigated for NTM because ABPA can lead to bronchiectasis and impaired mucus clearance.
- NTM infections usually grow very slowly, so treatment is sometimes monitored rather than started immediately.
- Treating NTM and aspergillosis together can be difficult because some NTM antibiotics interfere with antifungal medicines.
- Doctors usually treat the infection causing the most harm first while monitoring the other carefully.
Table of contents
- What are NTM?
- What is Mycobacterium avium complex (MAC)?
- Why NTM infections occur in some people
- What is bronchiectasis?
- Why patients with ABPA may be asked about NTM
- Why NTM and Aspergillus infections often occur together
- The lung infection cycle
- Chronic lung disease as a microbial ecosystem
- Why treatment can be complicated
- When treatment for NTM may be delayed
- How doctors balance treatment decisions
- NTM vs Aspergillosis – comparison table
- Common questions patients ask about NTM and Aspergillus
- When should patients seek medical advice?
- Reducing exposure to NTM in the environment
What are nontuberculous mycobacteria (NTM)?
Nontuberculous mycobacteria (NTM) are bacteria found naturally in the environment.
They live in:
- soil
- water
- dust
- plumbing systems
- shower heads and taps
Unlike tuberculosis, these bacteria are not normally spread between people.
Most people inhale them regularly without becoming ill. However, in some people with damaged lungs, these bacteria can establish a long-term lung infection.
What is Mycobacterium avium complex (MAC)?
The Mycobacterium avium complex (MAC) is the most common cause of NTM lung disease.
This group includes:
- Mycobacterium avium
- Mycobacterium intracellulare
MAC lung disease usually develops slowly over months or years.
Symptoms may include:
- chronic cough
- sputum production
- breathlessness
- fatigue
- weight loss
Because symptoms develop gradually, diagnosis can sometimes take time.
Why NTM infections occur in some people
NTM infections usually develop in people who already have structural lung disease.
Examples include:
- bronchiectasis
- chronic obstructive pulmonary disease (COPD)
- cystic fibrosis
- previous tuberculosis
- severe asthma
- aspergillosis
In these conditions, the lungs have damaged or widened airways, making it harder to clear mucus and microbes.
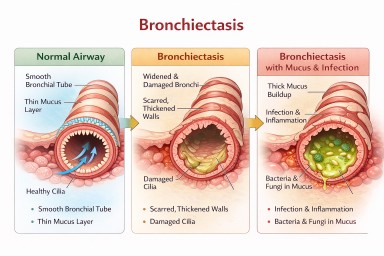
What is bronchiectasis?
Bronchiectasis is a condition where the airways become permanently widened and distorted.
In healthy lungs, mucus is cleared using:
- mucus movement
- tiny hair-like structures called cilia
- coughing
In bronchiectasis:
- mucus collects in the airways
- microbes become trapped
- infections become more likely
Bronchiectasis is common in patients with Allergic Bronchopulmonary Aspergillosis (ABPA) and other chronic lung diseases.
Why patients with ABPA may be asked about NTM
Some patients with Allergic Bronchopulmonary Aspergillosis (ABPA) are surprised when their doctors start investigating nontuberculous mycobacteria (NTM).
This usually happens because ABPA can lead to bronchiectasis, which increases the risk of other lung infections.
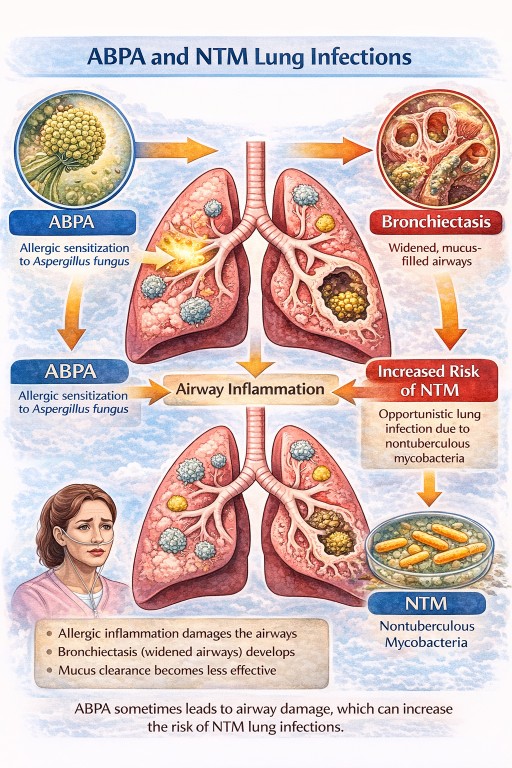
In ABPA:
- inflammation caused by allergic reactions to Aspergillus can damage the airways
- over time the airways may become widened and distorted, causing bronchiectasis
- mucus clearance becomes less effective
When mucus accumulates in the airways, microbes that are normally cleared from the lungs can sometimes persist. These may include:
- nontuberculous mycobacteria (NTM)
- Pseudomonas bacteria
- other organisms that affect bronchiectasis patients
For this reason, doctors sometimes test patients with ABPA for NTM if:
- CT scans show bronchiectasis or nodules
- sputum cultures repeatedly grow unusual organisms
- symptoms worsen without a clear explanation
Importantly, having ABPA does not mean you will develop NTM infection. Most patients with ABPA never develop NTM disease.
However, because the conditions share similar risk factors, doctors sometimes check for both.
Why NTM and Aspergillus infections often occur together
NTM bacteria and Aspergillus fungi both thrive in damaged lungs.
Three factors explain the overlap.
1. Damaged airways trap microbes
When airways are widened or distorted:
- mucus collects
- microbes are not cleared effectively
This allows organisms such as NTM and Aspergillus to persist.
2. Chronic infection causes further lung damage
NTM infection can lead to:
- inflammation
- worsening bronchiectasis
- lung nodules
- sometimes lung cavities
These cavities can then be colonised by Aspergillus, which may lead to chronic pulmonary aspergillosis (CPA).
3. Aspergillus can worsen structural damage
Once Aspergillus becomes established it can cause:
- inflammation
- enlargement of lung cavities
- worsening bronchiectasis
This further damage makes the lungs even more susceptible to infection.
The lung infection cycle
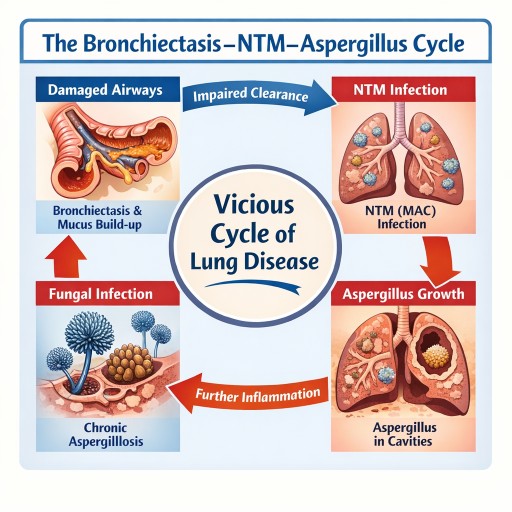
In many patients the relationship between bronchiectasis, NTM and Aspergillus becomes a cycle:
- Lung disease develops
- Bronchiectasis forms
- NTM infection establishes
- Lung damage worsens
- Aspergillus colonises damaged tissue
- Chronic aspergillosis develops
- Lung damage continues
At this stage the lungs may contain multiple organisms simultaneously.
Chronic lung disease as a microbial ecosystem
Doctors increasingly recognise that damaged lungs may contain several interacting microbes rather than a single infection.
Common organisms include:
- Mycobacterium avium complex (MAC)
- Aspergillus species
- Pseudomonas bacteria
- other organisms
For this reason clinicians sometimes describe chronic lung disease as a disturbed lung microbial ecosystem.
Why treatment can be complicated
NTM and aspergillosis treatments can interact.
Typical MAC treatment includes:
- azithromycin or clarithromycin
- ethambutol
- rifampicin
However rifampicin strongly reduces levels of antifungal drugs, including:
- itraconazole
- voriconazole
- posaconazole
These antifungals are commonly used to treat chronic pulmonary aspergillosis.
Because of this interaction, treating both infections at the same time can be challenging.
Other medicines that may interact with rifampicin
Rifampicin affects how the liver processes many medicines. This means it can reduce the effectiveness of several commonly used drugs, including some treatments for heart conditions, blood thinners, hormonal medicines, and certain antidepressants.
Because of this, doctors and pharmacists always review a patient’s medication list before starting rifampicin. Patients should tell their healthcare team about all medicines they take, including over-the-counter medicines, inhalers, and herbal supplements. In most cases, safe alternatives or dose adjustments can be used if needed.
When treatment for NTM may be delayed
Unlike many bacterial infections, MAC often progresses slowly.
Doctors sometimes monitor the infection before starting treatment. This approach is called active monitoring or watchful waiting.
Monitoring may include:
- CT scans
- sputum cultures
- lung function tests
- symptom assessment
Treatment may be delayed if:
- symptoms are mild
- CT scans are stable
- another condition requires more urgent treatment
For example, aspergillosis may be treated first if it is causing the main symptoms or lung damage.
How doctors balance treatment decisions
When both infections are present, clinicians try to identify which infection is currently causing the most harm.
Doctors consider:
Symptoms
- worsening cough
- breathlessness
- fatigue
- weight loss
- haemoptysis (coughing blood)
CT scan findings
- enlarging cavities
- fungal balls
- nodules typical of NTM disease
- worsening bronchiectasis
Laboratory results
- sputum cultures for NTM
- Aspergillus blood tests, such as Aspergillus IgG
If one infection clearly explains the patient’s symptoms, that infection usually becomes the treatment priority.
Treatment plans may then change over time as the balance of disease changes.
NTM vs Aspergillosis – What’s the difference?
| Feature | NTM (MAC) Lung Disease | Aspergillosis |
|---|---|---|
| Type of organism | Bacteria | Fungus |
| Source | Soil, water, plumbing | Airborne fungal spores |
| Spread between people | Rare | Does not spread |
| Typical speed | Slow, chronic infection | Variable |
| Typical CT findings | Nodules, bronchiectasis, cavities | Cavities, fungal balls, airway inflammation |
| Treatment | Long antibiotic courses, often 12–18 months | Antifungal medicines |
| Drug interaction issues | Rifampicin interferes with antifungals | Antifungal levels can be reduced by rifampicin |
Common questions patients ask about NTM and Aspergillus
If MAC grows slowly, why treat it?
Although MAC grows slowly, it can still cause progressive lung damage over time.
Treatment is usually recommended if there is:
- worsening symptoms
- declining lung function
- progressive CT scan changes
Can NTM be present without causing disease?
Yes. Some people have NTM colonisation without active infection.
Doctors diagnose NTM lung disease only when symptoms, imaging findings and repeated cultures all support the diagnosis.
Why do NTM and Aspergillus often occur together?
Both organisms tend to grow in damaged airways, especially where bronchiectasis is present and mucus clearance is poor.
Will both infections always be treated?
Not necessarily. Doctors often treat the infection causing the most immediate problem while monitoring the other.
Does NTM mean my aspergillosis is worsening?
Not necessarily. Both infections occur in damaged lungs, so they may simply share the same environment.
Can NTM lead to aspergillosis?
Sometimes. If NTM infection causes lung cavities or worsening bronchiectasis, these damaged areas may later become colonised by Aspergillus.
Should I worry if my doctor decides not to treat NTM immediately?
Not necessarily. Because MAC often progresses slowly, doctors sometimes choose active monitoring rather than immediate treatment.
When should patients seek medical advice?
People living with aspergillosis, bronchiectasis or NTM infection often have ongoing symptoms such as cough and sputum production. These symptoms may fluctuate and do not always mean the disease is worsening.
However, certain changes should prompt medical review.
Seek medical advice if you notice worsening breathing symptoms
- increasing breathlessness
- a significant increase in cough
- a noticeable increase in sputum production
- sputum becoming thicker, darker or foul-smelling
These symptoms may indicate:
- bacterial infection
- worsening bronchiectasis
- progression of NTM infection
- worsening aspergillosis
Coughing up blood (haemoptysis)
Haemoptysis can occur in both bronchiectasis and aspergillosis.
Seek medical advice if:
- bleeding increases
- blood appears repeatedly
- there is more than a small amount of blood
- bleeding occurs suddenly with breathlessness
Large amounts of blood should be treated as a medical emergency.
Unexplained weight loss or increasing fatigue
Persistent or worsening:
- weight loss
- fatigue
- loss of appetite
may indicate:
- progressive infection
- increasing inflammation
- advancing NTM disease
Fever or feeling unwell
New symptoms such as:
- fever
- chills
- chest discomfort
- feeling generally unwell
may suggest a new infection, such as a bacterial chest infection, which may require treatment.
Rapid change in symptoms
Seek medical advice if you experience:
- sudden worsening breathlessness
- significant chest pain
- new wheezing
- severe fatigue developing quickly
Symptoms that may remain stable
Many people with chronic lung disease experience symptoms that remain relatively stable for long periods, including:
- a chronic cough
- daily sputum production
- mild breathlessness
- intermittent fatigue
Doctors monitor these symptoms over time using:
- CT scans
- sputum cultures
- lung function tests
These investigations help clinicians determine whether infections such as NTM or Aspergillus are stable or progressing.
Reducing exposure to NTM in the environment
Patients with bronchiectasis, ABPA, or other chronic lung diseases sometimes ask whether they should try to avoid environmental exposure to nontuberculous mycobacteria (NTM).
NTM are very common in the natural environment and cannot be completely avoided. They are found in:
- soil and compost
- garden dust
- natural water sources
- tap water and plumbing systems
- showerheads
- hot tubs and spa pools
For most people, the goal is sensible risk reduction rather than strict avoidance. Major lifestyle restrictions are usually not necessary.
Water exposure
NTM can grow in biofilms inside plumbing systems, including showerheads. Small amounts of bacteria may become airborne when water is aerosolised.
Some simple precautions may help reduce exposure:
- avoid frequent use of hot tubs or spa pools
- allow taps or showers to run briefly if they have not been used for several days
- clean showerheads periodically to remove biofilm and limescale
Normal showering and bathing are considered safe for most patients.
NTM infection occurs when bacteria are inhaled into the lungs rather than swallowed. Drinking ordinary tap water is therefore considered safe for most people, and patients are not usually advised to avoid tap water for drinking.
Gardening and soil exposure
NTM bacteria are often present in soil and compost. Gardening can still be enjoyed safely with a few sensible precautions.
- wear gloves when gardening
- avoid inhaling dust from dry compost or soil
- dampen compost before handling to reduce dust
- wash hands after gardening
For people with bronchiectasis or NTM disease, wearing a mask during dusty gardening activities may help reduce inhalation of soil particles.
Reducing dust exposure
Activities that generate dust can increase inhalation of environmental microbes.
Helpful precautions include:
- avoiding sweeping very dusty areas indoors
- ventilating indoor spaces
- wearing a mask during dusty tasks such as handling compost or dry soil
Cleaning showerheads
Cleaning showerheads periodically can help remove limescale and biofilms where microbes may grow.
A simple method is:
- Remove the showerhead if possible.
- Soak it in white vinegar for about 30–60 minutes.
- Gently scrub the spray holes with a small brush.
- Rinse thoroughly.
- Run hot water for 30–60 seconds before use.
If the showerhead cannot be removed, a plastic bag filled with vinegar can be tied around the head so that it soaks.
Cleaning every 1–3 months is usually sufficient.
What is usually not necessary
Experts generally do not recommend major lifestyle changes to avoid NTM exposure. In most cases it is not necessary to:
- avoid showers
- avoid gardening completely
- install specialised water filtration systems
These activities are important for quality of life and general health, and evidence that strict avoidance prevents NTM disease is limited.
The most important protection
For patients with ABPA, bronchiectasis or aspergillosis, the most important protective measures remain:
- good airway clearance
- regular medical monitoring
- prompt treatment of infections
- maintaining overall lung health
Reducing environmental exposure may help slightly, but good management of lung disease remains the most important factor.
Key message
When NTM and Aspergillus infections occur together, treatment decisions focus on which infection is currently causing the most damage, while avoiding harmful drug interactions.
For patients with ABPA, one reason NTM may be discussed is that ABPA can lead to bronchiectasis and impaired mucus clearance, which can make other infections more likely.
Many patients live with these conditions for years with careful monitoring and specialist management.
Author: National Aspergillosis Centre Patient Information Team
Last reviewed: March 2026
Asthma and Aspergillosis
How fungal spores interact with asthma and other lung diseases
Every day we inhale thousands of microscopic fungal spores from the environment. One of the most common fungi in the air is Aspergillus fumigatus. In healthy lungs these spores are removed quickly by the lungs’ natural defence systems and cause no illness.
However, in people with asthma—particularly severe asthma—the interaction between the lungs and Aspergillus can be very different. The fungus may trigger allergic inflammation, grow in mucus within the airways, or occasionally contribute to chronic lung disease.
Understanding this relationship helps explain several important conditions including:
-
Aspergillus sensitisation
-
Severe Asthma with Fungal Sensitisation (SAFS)
-
Allergic Bronchopulmonary Aspergillosis (ABPA)
-
Aspergillus bronchitis
-
Chronic Pulmonary Aspergillosis (CPA)
Although asthma is the most common condition linked to Aspergillus allergy, other lung diseases such as bronchiectasis, Chronic Obstructive Pulmonary Disease (COPD), and tuberculosis-related lung damage can also create environments where the fungus becomes important.
Why Asthma Creates a Favourable Environment for Aspergillus
Asthma is a disease of airway inflammation and hyper-reactivity. The bronchi narrow during attacks because the airway wall becomes swollen and the surrounding smooth muscle contracts.
Several features of asthma make it easier for Aspergillus spores to remain in the lungs.
Mucus production
Asthma often causes increased production of thick airway mucus.
Normally mucus traps inhaled particles and moves them upward toward the throat via the mucociliary escalator.
In asthma:
-
mucus becomes thicker
-
clearance becomes less efficient
-
spores remain trapped
This trapped environment allows fungal spores to persist in the airway mucus.
Allergic immune responses
Many asthma patients have Type-2 (T2) inflammation (50-70%), involving immune pathways driven by:
-
Immunoglobulin E (IgE)
-
Interleukin-4
-
Interleukin-5
-
Interleukin-13
-
eosinophils
These pathways respond strongly to fungal allergens. When the immune system recognises Aspergillus proteins it may trigger allergic inflammation in the airways.
Fungal sensitisation is increasingly recognised as an important contributor to severe asthma (PMID: 24735832).
Aspergillus Sensitisation
Many people with asthma develop allergic sensitisation to Aspergillus.
Sensitisation means the immune system produces antibodies against fungal proteins.
Features include:
-
positive Aspergillus skin test or IgE blood test
-
worsening asthma symptoms
-
increased exacerbations
Studies suggest 10–25% of patients attending severe asthma clinics show Aspergillus sensitisation (PMID: 24735832).
However, sensitisation alone does not necessarily cause lung damage.
Severe Asthma with Fungal Sensitisation (SAFS)
Some patients with severe asthma have fungal sensitisation but do not meet the criteria for ABPA.
This condition is known as Severe Asthma with Fungal Sensitisation (SAFS).
Typical features include:
-
severe or poorly controlled asthma
-
fungal allergy
-
moderate IgE elevation
A randomised controlled trial demonstrated that antifungal therapy may improve symptoms in some SAFS patients (PMID: 18948425).
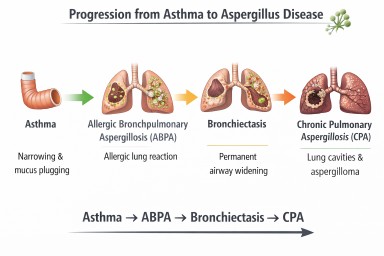
Allergic Bronchopulmonary Aspergillosis (ABPA)
Allergic Bronchopulmonary Aspergillosis is the most important Aspergillus-related disease associated with asthma.
ABPA occurs when Aspergillus grows within airway mucus and triggers a strong allergic immune response.
Typical findings include:
-
very high total IgE levels
-
Aspergillus-specific IgE and IgG antibodies
-
eosinophilia
-
mucus plugs containing fungal hyphae
-
central bronchiectasis
ABPA occurs in approximately:
-
1–2% of all asthma patients
-
up to 10–15% of severe asthma patients
These figures come from global prevalence estimates of ABPA in asthma populations (PMID: 23210682/.
Modern diagnostic criteria for ABPA were updated by the International Society for Human and Animal Mycology (ISHAM) in 2024 (PMID: 38423624).
Asthma and Aspergillus Disease Pathway
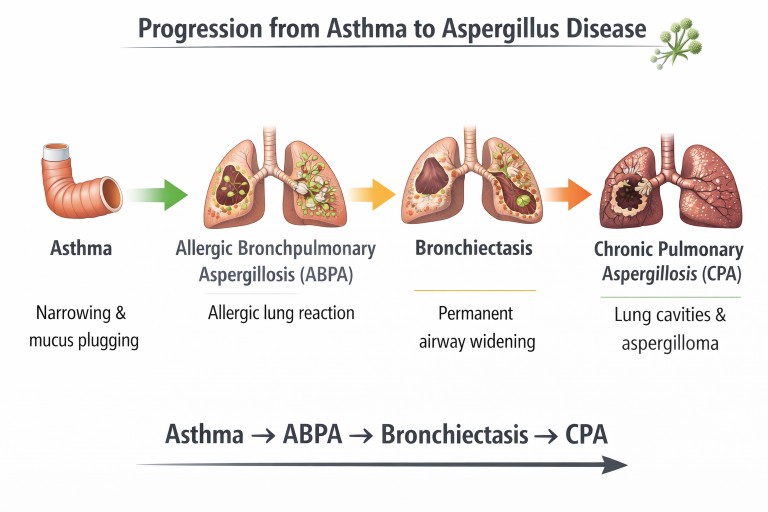
Possible interactions between asthma and Aspergillus. Some patients develop allergic disease (ABPA) which may lead to airway damage such as bronchiectasis (NB Progression to CPA is very rare).
When ABPA Causes Bronchiectasis
Repeated inflammation from ABPA may damage airway walls and lead to bronchiectasis.
Bronchiectasis occurs when airways become:
-
permanently widened
-
distorted
-
unable to clear mucus effectively
Instead of being cleared from the lungs, mucus pools in the airways.
This retained mucus creates an environment where microorganisms—including fungi—can grow.
Aspergillus Bronchitis
In some patients with bronchiectasis or chronic lung disease, Aspergillus may persist in airway mucus and cause chronic airway infection rather than allergy.
Symptoms may include:
-
chronic cough
-
sputum production
-
repeated positive Aspergillus cultures
IgE levels are usually lower than in ABPA.
Chronic Pulmonary Aspergillosis (CPA)
Chronic Pulmonary Aspergillosis is a slowly progressive fungal infection of damaged lung tissue.
CPA usually develops in lungs containing:
-
cavities
-
severe structural damage
Common underlying diseases include:
-
tuberculosis
-
sarcoidosis
-
severe COPD
Globally, the most common cause of CPA is previous tuberculosis infection (PMID: 22271943).
Asthma alone rarely causes CPA, but severe bronchiectasis or ABPA-related lung damage may occasionally lead to it.
Aspergillosis and Immune Competence
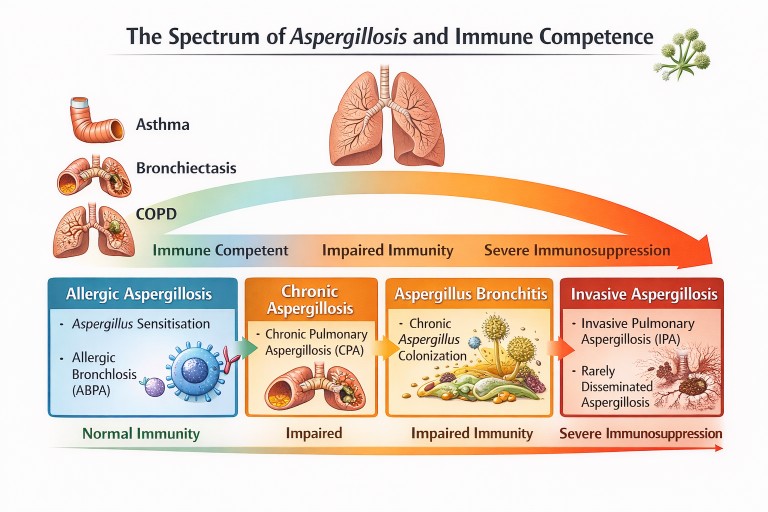
Different forms of aspergillosis occur depending on lung damage and immune function.
Other Lung Diseases Linked to Aspergillus
Although asthma is the most common condition associated with Aspergillus allergy, several other lung diseases can predispose to fungal disease.
Bronchiectasis
Dilated airways trap mucus, allowing fungi and bacteria to persist.
COPD
Chronic airway inflammation may lead to Aspergillus bronchitis or chronic pulmonary aspergillosis.
Tuberculosis
Post-tuberculosis lung cavities are the most common global cause of chronic pulmonary aspergillosis (PMID: 22271943).
Key Messages
-
Asthma is one of the most important diseases associated with Aspergillus-related lung conditions.
-
Many asthma patients develop fungal sensitisation.
-
A smaller proportion develop Allergic Bronchopulmonary Aspergillosis (ABPA).
-
Repeated inflammation from ABPA can lead to bronchiectasis.
-
Chronic pulmonary aspergillosis is rare in asthma alone but may occur if significant lung damage develops.
Understanding these interactions helps guide diagnosis and treatment for people living with asthma and Aspergillus-related disease.
Further reading
Agarwal R, Chakrabarti A, Shah A, Gupta D, Meis JF, Guleria R, Moss R, Denning DW; ABPA complicating asthma ISHAM working group. Allergic bronchopulmonary aspergillosis: review of literature and proposal of new diagnostic and classification criteria. Clin Exp Allergy. 2013 Aug;43(8):850-73. doi: 10.1111/cea.12141. PMID: 23889240.
Denning DW, Pleuvry A, Cole DC. Global burden of chronic pulmonary aspergillosis as a sequel to pulmonary tuberculosis. Bull World Health Organ. 2011 Dec 1;89(12):864-72. doi: 10.2471/BLT.11.089441. Epub 2011 Sep 27. PMID: 22271943; PMCID: PMC3260898.
Weekly Aspergillosis Research Update – Week 10, 2026
Focus: chronic aspergillosis, allergic Aspergillus disease, and long-term lung damage
This week’s papers are especially relevant to people living with Allergic Bronchopulmonary Aspergillosis (ABPA), allergic bronchopulmonary mycosis, and Chronic Pulmonary Aspergillosis (CPA). The strongest themes are the potential value of Immunoglobulin E (IgE) as a marker of future lung decline, the growing role of biologic therapies in steroid-sparing care, and improved tools for diagnosing CPA in people with previous tuberculosis.
Acute invasive aspergillosis papers are included lower down for context, but this update prioritises chronic and longer-term disease.
Chronic and allergic Aspergillus disease
High total serum IgE level at diagnosis was associated with a progressive decline in lung function in asthmatic patients with allergic bronchopulmonary mycosis
Kodama Y, Takaoka S, Nakashima T, Matsunaga K, Terada K, Yamashita Y, Masumitsu H, Miyasaka A, Muraoka T, Masumoto N, Kaneko T, Watanabe M, Tsurikisawa N.
Allergy Asthma Clin Immunol. 2026 Mar 8. doi: 10.1186/s13223-026-01024-2.
PMID: https://pubmed.ncbi.nlm.nih.gov/41796390/
Why this matters
This is one of the most important chronic-disease papers in this batch. It suggests that very high total IgE at diagnosis may not just reflect current disease activity, but may also predict future lung damage.
Key points
Patients with allergic bronchopulmonary mycosis (ABPM), including many with Allergic Bronchopulmonary Aspergillosis (ABPA), who had higher IgE levels at diagnosis showed a more progressive decline in lung function over time.
This raises the possibility that baseline IgE could help identify patients at higher risk of long-term airway damage.
It supports the idea that some patients may need closer monitoring and earlier treatment escalation rather than waiting for repeated flare-ups.
Relevance
For patients and clinicians, this paper reinforces that IgE is not just a number to follow during treatment. A very high starting IgE may signal the need for more careful long-term planning, especially in people with asthma, mucus plugging, recurrent exacerbations or bronchiectasis.
Biologics Use in Eosinophilic Lung Disease: Controversies and Consensus
Pérez de Llano L, Rivas DD, Pavord I, Aslam MMS, Lugogo N.
J Allergy Clin Immunol Pract. 2026 Mar;14(3):583-596.e12. doi: 10.1016/j.jaip.2026.01.022.
PMID: https://pubmed.ncbi.nlm.nih.gov/41786384/
Why this matters
This review is highly relevant to current ABPA care because biologics are increasingly being used to reduce reliance on oral corticosteroids, especially in people with severe asthma and recurrent eosinophilic inflammation.
Key points
The review discusses biologics including omalizumab, mepolizumab, benralizumab, dupilumab and tezepelumab.
It highlights growing evidence that biologics may help some patients with ABPA by reducing steroid burden, improving asthma control and lowering exacerbation frequency.
The authors also stress that evidence in ABPA is still developing and remains less robust than in severe eosinophilic asthma.
Relevance
This is a useful overview of where the field is heading. For many patients with ABPA, the major clinical problem is not only fungal sensitisation but the long-term harm caused by repeated steroid courses. Biologics are becoming an increasingly important part of steroid-sparing strategy, though patient selection remains crucial.
Differential Diagnosis of Eosinophilic Lung Diseases
Emmi G, Bass J, Baratella E, Akuthota P, Loscocco GG.
J Allergy Clin Immunol Pract. 2026 Mar;14(3):542-557. doi: 10.1016/j.jaip.2026.01.027.
PMID: https://pubmed.ncbi.nlm.nih.gov/41786383/
Why this matters
ABPA is still often missed, mislabelled or diagnosed late. This review is useful because it places ABPA in the wider context of eosinophilic lung disease, where several conditions can look similar.
Key points
The paper compares ABPA with other eosinophilic lung diseases such as chronic eosinophilic pneumonia, eosinophilic granulomatosis with polyangiitis, and drug-related eosinophilic lung disease.
It emphasises the importance of combining history, imaging, blood eosinophils, total IgE, fungal sensitisation and radiology.
The review underlines how easily overlap can occur, especially in people with severe asthma.
Relevance
For patients, this matters because getting the diagnosis right affects treatment. Not every eosinophilic lung disease is ABPA, and not every worsening in an asthma patient with high eosinophils is due to fungus. For clinicians, it is a helpful reminder to keep a broad differential diagnosis.
Chronic Pulmonary Aspergillosis
Performance of the LDBio Aspergillus ICT lateral flow assay and western blot for diagnosing chronic pulmonary aspergillosis in post-tuberculosis patients: a prospective study from South India
Samaddar A, Pramanik P, Voleti H, Akshata JS, Nagarathna S, Thennarasu K, Nagraja C.
Microbiol Spectr. 2026 Mar 6:e0384725. doi: 10.1128/spectrum.03847-25.
PMID: https://pubmed.ncbi.nlm.nih.gov/41789940/
Why this matters
This is the key CPA paper in this week’s list. It focuses on a major real-world problem: how to diagnose CPA more effectively in people left with lung damage after tuberculosis.
Key points
The study found that the LDBio Aspergillus immunochromatographic test (ICT) performed well in diagnosing CPA in post-tuberculosis patients.
Western blot also performed strongly, and combining the tests improved diagnostic confidence.
The results support the use of simpler, more accessible serology in settings where advanced imaging or specialist fungal laboratories may be limited.
Relevance
CPA after tuberculosis remains underdiagnosed worldwide. This paper is especially important because it supports the use of practical, lower-complexity diagnostics that may help identify patients earlier. That has implications far beyond India, particularly in regions where post-tuberculosis lung disease is common.
Host susceptibility and chronic disease risk
Oncostatin M receptor deficiency as a novel candidate genetic cause of autosomal recessive hyper-IgE syndrome
Andersen S, Assing K, Jensen J, Rasmussen LD, Laursen CB, Dellgren CD, Hinke DM, Degn SE, Mogensen TH.
J Hum Immun. 2026 Mar 3;2(3):e20250119. doi: 10.70962/jhi.20250119.
PMID: https://pubmed.ncbi.nlm.nih.gov/41783139/
Why this matters
Some patients develop chronic or severe Aspergillus disease because of an underlying immune problem that may not be obvious at first. This paper adds a possible new genetic explanation.
Key points
The authors describe a patient with very high IgE, eczema, bone fractures and Chronic Pulmonary Aspergillosis (CPA).
They identified a rare variant in the oncostatin M receptor (OSMR) gene.
The paper proposes OSMR deficiency as a possible new cause of autosomal recessive hyper-IgE syndrome.
Relevance
Although rare, studies like this help explain why a small number of people develop unusual susceptibility to chronic fungal disease. Over time, this kind of work may improve genetic diagnosis, immune work-up and personalised management in patients with recurrent or unexplained Aspergillus disease.
Important diagnostic lesson
Peripheral T-cell lymphoma-NOS presenting with cavitary lung lesions mimicking invasive aspergillosis
Lopez Ventosa J, Rodriguez A, Garcia N, Tirado M, Nieves Rivera J.
BMJ Case Rep. 2026 Mar 4;19(3):e268805. doi: 10.1136/bcr-2025-268805.
PMID: https://pubmed.ncbi.nlm.nih.gov/41781006/
Why this matters
Although this is not a chronic aspergillosis paper, it is worth noting because it highlights a key problem in lung medicine: cavities and positive biomarkers do not always equal Aspergillus infection.
Key points
A patient with cavitary lung lesions and a positive serum galactomannan was initially treated for presumed aspergillosis.
Tissue biopsy did not support fungal infection.
The final diagnosis was peripheral T-cell lymphoma.
Relevance
This is a valuable reminder that malignancy, tuberculosis and other diseases can mimic CPA or invasive aspergillosis, and that tissue diagnosis remains important when the picture does not fit cleanly.
Acute invasive aspergillosis: important context papers
How to safely discontinue antifungal treatment in invasive pulmonary aspergillosis? - Clinical considerations in haematology
Stemler J, Sprute R, Koehler P, Cornely OA.
Clin Microbiol Infect. 2026 Mar 6:S1198-743X(26)00106-0. doi: 10.1016/j.cmi.2026.03.001.
PMID: https://pubmed.ncbi.nlm.nih.gov/41796963/
25 years of improvement in mortality in invasive aspergillosis in haematology patients: will it be sustained or is it under threat?
Maertens JA, Vanbiervliet Y, Mercier T, Aerts R, Lagrou K, Slavin MA.
J Antimicrob Chemother. 2026 Mar 4;81(4):dkag077. doi: 10.1093/jac/dkag077.
PMID: https://pubmed.ncbi.nlm.nih.gov/41790511/
Invasive aspergillosis in liver transplant recipients in France (2007-21): a nationwide, retrospective, matched case-control study
Le Hyaric C, Melenotte C, Lefebvre F, Saliba F, Botterel F, El-Domiaty N, Dumortier J, Persat F, Do R, Pasquier G, Camus C, Gangneux JP, Kamar N, Iriart X, Monsel A, Fekkar A, Conti F, Vuotto F, Loridant S, Durand F, Bonnal C, Barbaz M, Chesnay A, Vignals C, Lefranc M, Guerin R, Moniot M, Weil D, Bellanger AP, Decaens T, Maubon D, Lebossé F, Artzner T, Morel G, Letscher-Bru V, Herbrecht R, Ader F, Lortholary O, Lefort A, Guichon C, Danion F.
Lancet Microbe. 2026 Mar 2:101272. doi: 10.1016/j.lanmic.2025.101272.
PMID: https://pubmed.ncbi.nlm.nih.gov/41785881/
Treatment Monitoring and Outcome Prediction in Invasive Aspergillosis using Immunologic Markers
Pereira A, Scott J, Sarlea A, Sprute R, Aerts R, Lass-Flörl C, Mikulska M, Sedik S, Garcia-Vidal C, Gangneux JP, Giacobbe DR, Prattes J, Grothe J, Biswas S, Monzo-Gallo P, Bassetti M, Maertens J, Kumar V, Koehler P, Cunha C, Netea MG, Carvalho A, Hoenigl M.
J Infect Dis. 2026 Mar 4:jiag140. doi: 10.1093/infdis/jiag140.
PMID: https://pubmed.ncbi.nlm.nih.gov/41778487/
Bronchiectasis in Aspergillosis Patients
Many people with aspergillosis also develop bronchiectasis, a condition in which some of the airways in the lungs become permanently widened and damaged. Understanding bronchiectasis can help explain many symptoms experienced by patients with Allergic Bronchopulmonary Aspergillosis (ABPA – Allergic Bronchopulmonary Aspergillosis) and Chronic Pulmonary Aspergillosis (CPA – Chronic Pulmonary Aspergillosis).
Although bronchiectasis cannot usually be reversed, it can often be managed effectively, and understanding how it works helps patients recognise symptoms and flare-ups early.
The airways of the lungs
Your lungs contain a branching network of tubes called bronchi and bronchioles that carry air in and out of the lungs.
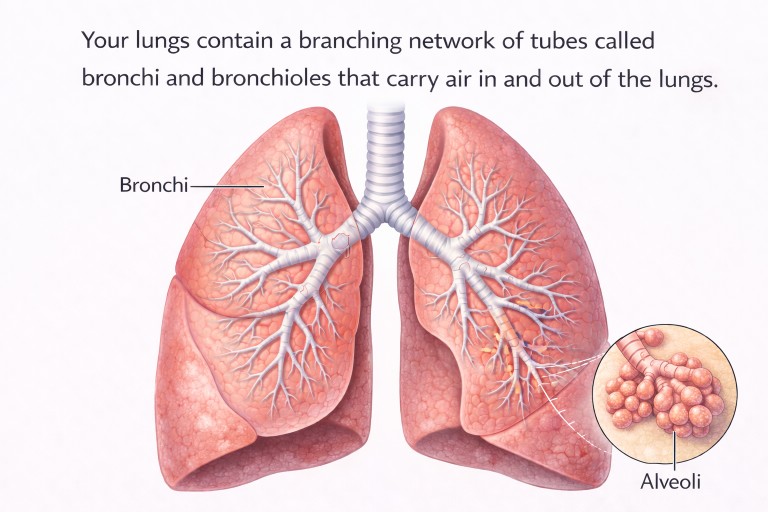
Air travels through the trachea (windpipe) into the bronchi, which then divide repeatedly into smaller and smaller tubes called bronchioles. At the ends of the bronchioles are millions of tiny air sacs called alveoli, where oxygen moves into the bloodstream.
The lining of the airways produces a thin layer of mucus that traps dust, bacteria and fungal spores that we breathe in every day.
Tiny hair-like structures called cilia move this mucus upward toward the throat, where it can be swallowed or coughed out. This system acts like a self-cleaning escalator, helping keep the lungs clear.
What is bronchiectasis?
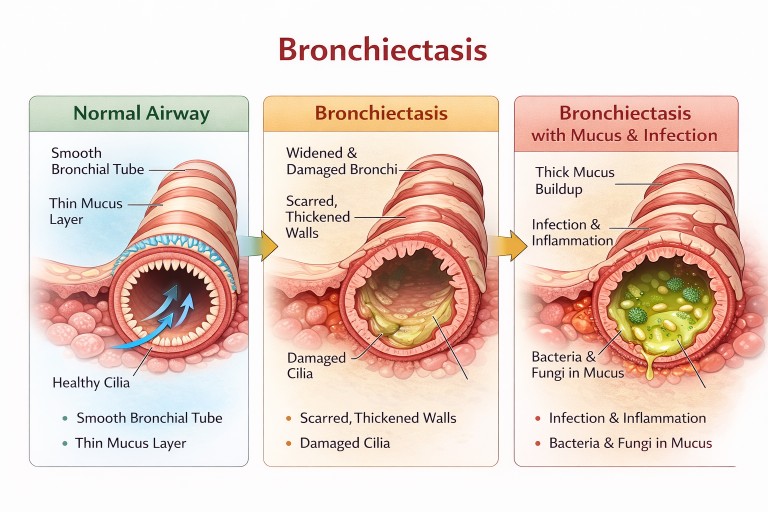
In bronchiectasis, some of the airways become permanently widened and damaged.
When this happens:
-
the airway walls become inflamed and weakened
-
the tubes widen and lose their normal shape
-
mucus becomes harder to clear
-
bacteria and fungi can grow in trapped mucus
Over time, this leads to repeated infections and inflammation.
Doctors often describe bronchiectasis as a vicious cycle:
-
Infection or inflammation damages the airway
-
The airway widens and mucus clearance becomes poor
-
Mucus builds up in the airway
-
Bacteria and fungi grow in the mucus
-
Infection and inflammation occur again
Without treatment, this cycle can gradually worsen airway damage.
Why bronchiectasis is common in aspergillosis
Bronchiectasis is particularly common in patients with aspergillosis, especially in ABPA.
In ABPA, the immune system reacts strongly to Aspergillus spores in the airways. This causes:
-
allergic inflammation in the bronchi
-
thick mucus plugs
-
repeated airway irritation
Over time, this inflammation can damage the airway walls and lead to bronchiectasis, often affecting the central airways of the lungs.
Once bronchiectasis develops, mucus becomes harder to clear, which can allow bacteria and fungi such as Aspergillus to persist in the lungs.
Symptoms of bronchiectasis
Many symptoms of bronchiectasis overlap with those of aspergillosis.
Common symptoms include:
-
persistent cough
-
regular sputum (phlegm) production
-
breathlessness
-
fatigue
-
frequent chest infections
Sputum may be:
-
clear
-
yellow or green
-
occasionally blood-streaked
In people with ABPA, patients sometimes cough up thick mucus plugs, which may appear brown or rubbery.
How bronchiectasis is diagnosed
Bronchiectasis is usually diagnosed using a High Resolution CT (HRCT) scan of the lungs.
On a CT scan, doctors may see:
-
widened airways
-
thickened airway walls
-
mucus plugs
-
airways extending closer to the edge of the lung than normal
Radiologists sometimes describe a typical appearance called the “signet ring sign”, where the widened airway appears larger than the nearby blood vessel.
Bronchiectasis and aspergillosis flare-ups
Because bronchiectasis and aspergillosis affect the same airways, it can sometimes be difficult for patients to recognise whether worsening symptoms are caused by:
-
a bronchiectasis infection, or
-
an aspergillosis flare-up.
Understanding the differences can help patients recognise when to seek medical advice.
Bronchiectasis exacerbations
Bronchiectasis flare-ups are usually caused by bacterial infection in trapped mucus.
Patients may notice:
-
increased sputum production
-
sputum becoming yellow or green
-
increased coughing
-
fever or feeling unwell
-
breathlessness
Many patients describe bronchiectasis exacerbations as feeling like a chest infection.
Treatment usually involves:
-
antibiotics
-
airway clearance physiotherapy
-
increased mucus clearance
Aspergillosis flare-ups
Aspergillosis flare-ups are usually caused by fungal activity or immune reactions to Aspergillus.
Patients may notice:
-
worsening wheezing
-
chest tightness
-
increased breathlessness
-
thick mucus plugs
Some patients cough up:
-
brown mucus
-
rubbery mucus plugs
-
mucus shaped like small airway casts
In Chronic Pulmonary Aspergillosis, patients may also experience:
-
persistent cough
-
fatigue
-
weight loss
-
occasionally coughing blood
Treatment may involve:
-
steroid treatment
-
antifungal medication
-
biologic therapies in ABPA
Key differences patients often notice
| Feature | Bronchiectasis flare-up | Aspergillosis flare-up |
|---|---|---|
| Main cause | Bacterial infection in trapped mucus | Fungal activity or immune reaction to Aspergillus |
| Sputum colour | Yellow or green | Brown mucus plugs or thick sticky mucus |
| Fever | More common | Less common |
| Wheezing | Sometimes present | Often worse |
| Feeling like a chest infection | Common | Less typical |
| Response to antibiotics | Usually improves | Usually little improvement |
| Mucus plugs | Less common | More common in ABPA |
| Blood tests | Usually unchanged | IgE may rise in ABPA |
Both conditions can occur together
In reality, bronchiectasis and aspergillosis often interact with each other.
For example:
-
ABPA can cause bronchiectasis
-
bronchiectasis allows fungi and bacteria to remain in mucus
-
infection and fungal inflammation can occur at the same time
Doctors may investigate flare-ups using:
-
sputum cultures
-
blood tests (for example IgE levels in ABPA)
-
CT scans
-
inflammatory markers
Why airway clearance is important
Because bronchiectasis makes mucus harder to clear, airway clearance physiotherapy becomes a key part of treatment.
Common techniques include:
-
Active Cycle of Breathing Technique (ACBT)
-
Autogenic drainage
-
oscillating devices such as Flutter or Acapella
Regular airway clearance helps:
-
remove mucus from the lungs
-
reduce infections
-
improve breathing
-
reduce cough
For patients with aspergillosis, clearing mucus may also help remove fungal material from the airways.
When patients should seek medical advice
Patients should contact their healthcare team if they notice:
-
rapidly increasing sputum
-
fever or feeling unwell
-
coughing blood
-
severe breathlessness
-
large mucus plugs
Early treatment can often prevent a mild flare-up from becoming a more serious infection.
The key message
Bronchiectasis means that some airways in the lungs have become permanently widened, making mucus harder to clear.
However, many people with aspergillosis and bronchiectasis live active lives with stable lung function.
With good treatment, airway clearance, and early management of infections, bronchiectasis can often be well controlled for many years.
Damp & Mould Health Evidence Monitor: 4 March 2026
Date of check
4 March 2026
🆕 New papers since last check
Early-life indoor mould exposure and lung function
Journal: Environmental Research: Health
Published: Feb 2026
Summary
Children exposed to serious indoor mould in early life showed:
-
reduced lung function in adolescence
-
increased risk of asthma during childhood
This study used long-term cohort data and objective lung function testing, strengthening the evidence that early mould exposure can have lasting respiratory consequences.
PMID: 39162373
PubMed link:
https://pubmed.ncbi.nlm.nih.gov/39162373/
Damp and mouldy homes: impact on lung health in childhood
Authors: Moorcroft C, Whitehouse A, Grigg J
Journal: Archives of Disease in Childhood (2025)
Summary
This clinical review explains how damp housing contributes to:
-
childhood asthma
-
recurrent respiratory infections
-
allergic disease
It emphasises that paediatric clinicians frequently encounter children whose symptoms are linked to poor housing conditions.
PMID: 39814530
PubMed link:
https://pubmed.ncbi.nlm.nih.gov/39814530/
Damp housing and mental health effects
Journal: Environmental Health Perspectives
Summary
A major review examining whether damp and mould exposure affects mental health.
Evidence suggests associations with:
-
anxiety
-
depression
-
psychological stress
Mechanisms may include chronic illness, housing insecurity, and inflammatory responses to mould exposure.
PMID: 39162373
PubMed link:
https://pubmed.ncbi.nlm.nih.gov/39162373/
📊 Summary
New or important items identified
• Evidence continues to strengthen the link between damp housing and respiratory disease
• Long-term cohort data show effects on lung development
• Emerging research also suggests mental health impacts
Weeks 6–7 Aspergillosis Literature Update
22 February – 3 March 2026
1️⃣ Chronic Pulmonary Aspergillosis (CPA) & Structural Lung Disease
Clinical impact of chronic pulmonary aspergillosis in patients with pulmonary nontuberculous mycobacterial disease
Annals of Medicine
Lee MR et al., 24 Feb 2026
PMID: 41736260
🔗 https://pubmed.ncbi.nlm.nih.gov/41736260/
Key Findings
-
CPA subtypes identified:
-
Chronic cavitary pulmonary aspergillosis (CCPA)
-
Chronic fibrosing pulmonary aspergillosis (CFPA)
-
Subacute invasive pulmonary aspergillosis (SAIA)
-
-
CPA significantly worsened:
-
Mortality
-
Lung function trajectory
-
Treatment burden
-
Relevance
-
Reinforces strong NTM–CPA interaction.
-
Supports routine Aspergillus IgG screening in deteriorating NTM patients.
-
Highly relevant for structured longitudinal services such as NAC.
Post-tuberculosis lung disease and pulmonary aspergillosis management
Expert Review of Anti-infective Therapy
Sehgal IS et al., 22 Feb 2026
PMID: 41674445
🔗 https://pubmed.ncbi.nlm.nih.gov/41674445/
Highlights
-
CPA is the most frequent fungal sequela of treated tuberculosis.
-
Diagnostic delay remains common.
-
Imaging + Aspergillus IgG remain central tools.
Strategic Implication
Post-TB surveillance pathways should incorporate fungal screening protocols.
2️⃣ Invasive Aspergillosis (IA) – ICU & CNS
Why do we urgently need a new treatment for cerebral aspergillosis?
Expert Review of Anti-infective Therapy
Soman R et al., 27 Feb 2026
PMID: 41758247
🔗 https://pubmed.ncbi.nlm.nih.gov/41758247/
Core Issues
-
Extremely high mortality.
-
Poor CNS penetration of many antifungals.
-
Delayed diagnosis remains common.
Direction of Travel
-
CNS-penetrant azoles
-
Host-directed adjunctive therapy
-
Earlier molecular diagnostics
Clinical characteristics of probable invasive pulmonary aspergillosis in the ICU
(Research Square – preprint; not yet indexed in PubMed)
Key Themes
-
Rising IPA incidence in ICU.
-
Increasing recognition in non-classical immunocompromised hosts.
-
Diagnostic uncertainty persists.
Invasive Aspergillus Tracheobronchitis Presenting as Subglottic Stenosis
Respirology Case Reports
Sato T et al., 1 Mar 2026
(Indexing pending — searchable in PubMed by title)
Significance
-
Airway-dominant invasive disease.
-
Reminds clinicians that IA is not purely parenchymal.
3️⃣ Diagnostics – AI, Biomarkers & Rapid Testing
Identification of Aspergillus at section and species levels by AI-based microscopic morphology recognition
Journal of Clinical Microbiology
Tan M et al., 27 Feb 2026
PMID: 41757926
🔗 https://pubmed.ncbi.nlm.nih.gov/41757926/
Why It Matters
-
Species-level ID influences resistance prediction.
-
AI microscopy may support antifungal stewardship.
-
Potential synergy with resistance-genotyping services.
Performance evaluation of chemiluminescence immunoassay for quantitative (1,3)-β-D-glucan
Medical Mycology
Yuan K et al., 24 Feb 2026
PMID: 41733444
🔗 https://pubmed.ncbi.nlm.nih.gov/41733444/
Implication
Improved BDG quantification could refine:
-
Diagnostic confidence
-
Antifungal escalation decisions
-
AMS compliance
Rapid and reliable diagnosis of mucormycosis using colorimetric LAMP
Journal of Clinical Microbiology
Gu Y et al., 26 Feb 2026
PMID: 41746213
🔗 https://pubmed.ncbi.nlm.nih.gov/41746213/
Broader Context
Improving differentiation between mould pathogens is increasingly critical in ICU and transplant settings.
Evaluation of the Aspergillus Lateral Flow Assay
(Preprint; not indexed in PubMed)
Movement toward rapid, bedside semi-quantitative testing continues.
4️⃣ Immunology & Host Response
Ferroptosis-related biomarkers and subtypes in invasive aspergillosis
Toxicology Research
Tang L et al., 23 Feb 2026
PMID: 41756099
🔗 https://pubmed.ncbi.nlm.nih.gov/41756099/
Emerging Theme
-
Ferroptosis signatures in IA.
-
Potential for biomarker-guided host-directed therapy.
Immune Exhaustion in Chronic Infection and Cancer
MedComm
Song Y et al., 26 Feb 2026
PMID: 41768369
🔗 https://pubmed.ncbi.nlm.nih.gov/41768369/
Relevance
-
T-cell exhaustion pathways implicated in invasive pulmonary aspergillosis models.
-
Checkpoint biology may influence future antifungal immunotherapy.
Helminth Immune Modulation and Invasive Fungal Infections in Sub-Saharan Africa
Journal of Fungi
Fonte L et al., 23 Feb 2026
PMID: 41745302
🔗 https://pubmed.ncbi.nlm.nih.gov/41745302/
Implication
Immune skewing in endemic regions may influence:
-
IA susceptibility
-
Allergic bronchopulmonary aspergillosis (ABPA) patterns
Proposition for a New Classification of Hypersensitivity Reactions
Clinical Reviews in Allergy & Immunology
Szegedi A et al., 26 Feb 2026
PMID: 41746569
🔗 https://pubmed.ncbi.nlm.nih.gov/41746569/
Relevance to ABPA
Supports more nuanced immunophenotyping in complex hypersensitivity states.
5️⃣ Therapeutics & Antifungal Strategy
Influence of Extended Itraconazole Antifungal Prophylaxis After Lung Transplant
Journal of Transplantation
Fischer S et al., 26 Feb 2026
PMID: 41769149
🔗 https://pubmed.ncbi.nlm.nih.gov/41769149/
Clinical Angle
Balancing prolonged prophylaxis with resistance development and toxicity.
Auranofin and iodoquinol as repurposing drugs against filamentous fungi
Microbiology Spectrum
Xisto MIDdS et al., 27 Feb 2026
PMID: 41757906
🔗 https://pubmed.ncbi.nlm.nih.gov/41757906/
Takeaway
Repurposed agents remain essential in the slow antifungal pipeline landscape.
Arp9 modulates drug resistance and aflatoxin biosynthesis in Aspergillus flavus
PLoS Pathogens
Ma D et al., 2 Mar 2026
PMID: 41770810
🔗 https://pubmed.ncbi.nlm.nih.gov/41770810/
Strategic Significance
Links:
-
Chromatin regulation
-
Temperature adaptation
-
Drug resistance
-
Environmental pathogenic evolution
6️⃣ Radiology & Case-Based Insights
Radiologic Characterization of Invasive Fungal Infections of the Paranasal Sinuses and Skull Base
Cureus
S S et al., 23 Feb 2026
PMID: 41743147
🔗 https://pubmed.ncbi.nlm.nih.gov/41743147/
Clinical Utility
Supports ENT + radiology diagnostic differentiation.
Atypical Manifestations of Aspergillosis
Respirology Case Reports
Zahiri L et al., 1 Mar 2026
(Indexing pending — searchable by title)
Message
Aspergillosis remains a spectrum disease influenced heavily by host immunity.
📊 2-Week Synthesis
Emerging Patterns
-
Increasing ICU and CNS complexity
-
Strong CPA overlap with structural lung disease (NTM, TB)
-
Rapid diagnostic evolution (AI, BDG quantification, LAMP, LFA)
-
Growing focus on host biology (ferroptosis, immune exhaustion)
-
Continued therapeutic gap — particularly cerebral disease
National Aspergillosis Centre, Antifungal Therapeutic Drug Monitoring (TDM), Molecular Resistance Testing & Antimicrobial Stewardship
How the National Aspergillosis Centre Supports UK Clinicians
Long-term antifungal therapy in aspergillosis presents a distinct antimicrobial stewardship (AMS) challenge. Treatment is often prolonged, drug exposure is highly variable, and resistance may emerge during therapy.
The National Aspergillosis Centre (NAC), working closely with the Mycology Reference Centre Manchester (Manchester UK"], provides national expertise through:
-
Therapeutic drug monitoring (TDM)
-
Molecular resistance testing
-
Specialist Advice & Guidance
-
Remote multidisciplinary team (MDT) review
-
Standardised laboratory processes
Together, these services enable UK clinicians to optimise antifungal therapy while aligning with national AMS strategy and antimicrobial resistance (AMR) policy.
The National AMS Framework: Why This Matters
Antifungal stewardship sits within the wider UK antimicrobial resistance strategy.
Key national resources include:
1️⃣ NHS England – Digital Vision for Antimicrobial Stewardship
https://www.england.nhs.uk/long-read/digital-vision-for-antimicrobial-stewardship-in-england/
Emphasises:
-
Data-driven optimisation
-
Decision support
-
Clear documentation
-
Measurable stewardship interventions
2️⃣ Antimicrobial Prescribing & Stewardship Competency Framework
https://www.gov.uk/government/publications/antimicrobial-prescribing-and-stewardship-competencies
Defines clinician responsibilities including:
-
Right drug
-
Right dose
-
Right duration
-
Monitoring for toxicity
-
Review and stop decisions
3️⃣ English Surveillance Programme for Antimicrobial Utilisation and Resistance (ESPAUR)
Supports:
-
National resistance monitoring
-
Stewardship benchmarking
-
Reduction of inappropriate antimicrobial exposure
4️⃣ Chronic Pulmonary Aspergillosis (CPA) Service Specification
https://www.england.nhs.uk/publication/chronic-pulmonary-aspergillosis-service-adults/
This specialised service model explicitly includes:
-
Optimisation of antifungal therapy
-
Toxicity monitoring
-
Therapeutic drug monitoring
Antifungal stewardship is embedded within the commissioned service design.
Why Aspergillosis Requires Enhanced Stewardship
Unlike short-course antibacterial therapy, aspergillosis often involves:
-
Long-term triazole therapy
-
Structural lung disease
-
High interaction burden
-
Emerging environmental resistance
-
Potential for treatment failure despite adequate adherence
Effective stewardship therefore requires both:
-
Assurance of adequate drug exposure (TDM)
-
Assurance of organism susceptibility (molecular testing)
1️⃣ Therapeutic Drug Monitoring (TDM)
Triazole antifungals demonstrate:
-
High pharmacokinetic variability
-
Concentration-dependent toxicity
-
Reduced efficacy if under-dosed
TDM enables:
✔ Early detection of subtherapeutic exposure
✔ Prevention of toxicity
✔ Dose optimisation
✔ Reduction of avoidable escalation
This directly fulfils AMS competency expectations.
2️⃣ Molecular Resistance Testing
Azole resistance in Aspergillus fumigatus is increasingly recognised in the UK.
Through MRCM, NAC supports:
CYP51A Mutation Analysis
Common mutations include:
-
TR34/L98H
-
TR46/Y121F/T289A
These may arise:
-
Environmentally (azole fungicide pressure)
-
During long-term therapy
Phenotypic Susceptibility Testing
Where viable isolates are available:
-
Minimum inhibitory concentration (MIC) testing
-
Clinical interpretation to guide therapy
Why Resistance Testing Is Essential for AMS
If a patient deteriorates despite adequate serum levels:
-
Continuing the same azole is not stewardship
-
Escalating empirically without evidence increases antimicrobial pressure
Molecular confirmation ensures:
✔ Rational switching
✔ Avoidance of ineffective therapy
✔ Contribution to national resistance surveillance
This aligns with ESPAUR and national AMR objectives.
3️⃣ Remote Advice & Guidance & MDT Review
The NAC provides structured national clinician support.
This strengthens stewardship by:
✔ Refining diagnosis
✔ Preventing indication drift
✔ Setting defined review points
✔ Supporting stop decisions
✔ Reducing empirical prolonged therapy
Early specialist review is one of the most effective stewardship interventions.
Integrated Stewardship Model
| Clinical Situation | TDM | Molecular Testing |
|---|---|---|
| Initiation of azole | Yes | Not routine |
| Poor response + low level | Adjust dose | Not primary |
| Poor response + adequate level | Confirm exposure | Essential |
| Long-term therapy | Periodic monitoring | Consider if progression |
| Relapse on therapy | Check level | Strongly consider |
Exposure optimisation + susceptibility confirmation = complete antifungal stewardship.
Practical Workflow for UK Teams
Step 1 – Define Indication
-
Syndrome
-
Treatment objective
-
Planned review date
Step 2 – Baseline Safety Checks
-
Interaction review
-
Liver function tests
-
ECG where appropriate
Step 3 – Perform TDM
Include:
-
Drug
-
Dose
-
Time of last dose
-
Time of sampling
Step 4 – If Clinical Failure Occurs
-
Confirm adequate drug exposure
-
Consider molecular resistance testing
Step 5 – Define Stop/Review Criteria
Avoid open-ended therapy without documented reassessment.
Demonstrating AMS Compliance in Practice
Using NAC-supported services allows Trusts to evidence:
✔ Documented indication
✔ Dose optimisation
✔ Toxicity mitigation
✔ Rational escalation
✔ Defined review intervals
✔ Resistance surveillance contribution
✔ Specialist consultation
This is measurable, defensible antimicrobial stewardship.
Conclusion
Antifungal stewardship in aspergillosis cannot rely on restriction alone.
It requires:
-
Precision dosing
-
Genetic resistance detection
-
Structured specialist review
-
Alignment with national AMS frameworks
Through integrated therapeutic drug monitoring, molecular resistance testing, and national clinical support, the National Aspergillosis Centre provides a UK model for precision antifungal stewardship aligned with national antimicrobial resistance strategy.
🏥 Good News: New AI “Digital Scribe” Helping Doctors Spend More Time With Patients
We’re pleased to share some exciting developments from Manchester University NHS Foundation Trust that could directly improve your experience at clinic appointments.
A new technology called Ambient Voice Technology (AVT) is gradually being introduced across parts of the Trust. Think of it as a secure “digital scribe” that supports your clinician during your consultation.
What does it do?
With your permission, the system listens to the natural conversation between you and your doctor or nurse. It then:
-
Creates the clinical notes automatically
-
Drafts follow-up actions
-
Updates the electronic patient record (*i.e. another reason to use myMFT)
This means your clinician doesn’t need to spend as much time typing or looking at a screen — and can focus more fully on you.
📊 What Have the Early Results Shown?
Colleagues from Manchester University NHS Foundation Trust recently presented results from the Dragon Copilot trial at the Microsoft AI Tour in London.
The findings are encouraging:
✅ 88% of clinicians report saving around 2 minutes per appointment on documentation
✅ 88% say it improves quality and increases face-to-face time with patients
✅ Reduced mental workload for clinicians
✅ Significant reduction in after-clinic administrative work
Two minutes may not sound like much — but across a full clinic list, it adds up. Over time, this could help improve efficiency, reduce waiting times, and improve the overall clinic experience.
💻 How Is It Being Used?
The Dragon Copilot system connects directly into the Trust’s Hive Electronic Patient Record system. It is currently being used in:
-
Outpatient clinics
-
Manchester Royal Infirmary’s Emergency Department
Further expansion is planned in the coming weeks.
❤️ Why This Matters for NAC Patients
For patients with chronic conditions such as aspergillosis, consultations are often detailed and complex. Anything that:
-
Frees up clinician time
-
Improves note accuracy
-
Reduces administrative burden
-
Supports more focused, human interaction
…is a positive step forward.
The aim is not to replace clinicians — but to support them, so your appointment time is spent on what matters most: listening, explaining, planning, and answering your questions.
We’ll continue to keep you updated as this technology develops. It’s encouraging to see innovation being used to strengthen patient-centred care.
If you’d like to learn more, a short video featuring Trust leaders and clinicians was showcased at the Microsoft AI Tour and is available via Trust communications channels.