🧬 Could Antibody-Driven Dissolving of Charcot–Leyden Crystals Help ABPA?
Researchers have recently discovered that Charcot–Leyden crystals (CLCs) — the needle-shaped structures formed from the eosinophil protein galectin-10 — are not just debris.
In laboratory studies, specially designed antibodies can dissolve these crystals.
This has raised two important questions:
-
Could dissolving the crystals reduce airway inflammation?
-
Could dissolving them make mucus plugs easier to clear?
Here is what we currently know.
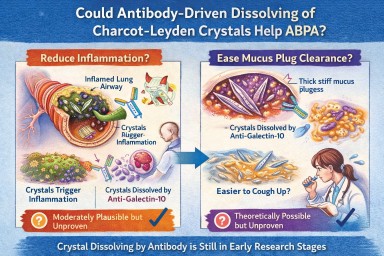
1️⃣ Could dissolving crystals reduce airway inflammation?
What we know
Laboratory and animal studies have shown:
-
Charcot–Leyden crystals can activate immune cells (especially macrophages).
-
They can stimulate inflammatory pathways (including inflammasome signalling).
-
In mouse models, antibodies targeting galectin-10 dissolved the crystals.
-
When crystals were dissolved, airway inflammation decreased.
This suggests that the crystals themselves may amplify inflammation, rather than simply mark it.
What this means biologically
In ABPA and eosinophilic asthma:
-
Eosinophils release galectin-10.
-
Galectin-10 crystallises.
-
Crystals may trigger further immune activation.
-
That leads to more inflammation → more eosinophils → more crystals.
Dissolving the crystals could theoretically interrupt this feedback loop.
How likely is this to help inflammation in humans?
Moderately plausible, but not yet proven.
The biological mechanism is strong.
The animal data are encouraging.
But no human clinical trials have yet shown reduced inflammation through crystal dissolution.
If developed successfully, this approach could:
-
Reduce airway immune activation
-
Lower exacerbation risk
-
Potentially reduce steroid dependence
But at present, it remains investigational.
2️⃣ Could dissolving crystals make mucus plugs easier to cough up?
This is more speculative — but still biologically reasonable.
Why mucus plugs are so thick in ABPA
ABPA mucus plugs contain:
-
Gel-forming mucins
-
DNA from inflammatory cells
-
Dead cells
-
Fungal fragments
-
Eosinophil proteins
-
Charcot–Leyden crystals
The crystals are:
-
Rigid
-
Needle-shaped
-
Structurally stable
When embedded in mucus, they likely increase:
-
Mechanical stiffness
-
Plug density
-
Resistance to deformation
From a physics perspective:
Removing rigid crystalline structures from a gel should reduce stiffness and improve flow.
Do we have direct evidence?
No.
There are currently:
-
No human studies measuring mucus clearance after crystal dissolution
-
No trials showing improved plug expectoration from crystal-targeting therapy
So while it is plausible that dissolving crystals could soften plugs, this has not yet been demonstrated in patients.
3️⃣ How strong is the overall case?
| Outcome | Evidence strength | Likelihood |
|---|---|---|
| Reduced inflammation | Strong biological rationale + animal data | Moderately promising |
| Easier mucus clearance | Biophysical plausibility only | Possible but unproven |
Inflammation reduction is the more evidence-supported target.
Improved plug clearance is plausible but currently theoretical.
4️⃣ How does this compare to existing treatments?
Current therapies (e.g., anti-IL-5 biologics) reduce eosinophils upstream.
That leads to:
-
Less galectin-10 release
-
Fewer crystals forming
-
Reduced inflammation
-
Often improved mucus plugging
So biologics already indirectly reduce crystal burden.
A crystal-dissolving antibody would act downstream, targeting the structural product directly.
This could theoretically:
-
Accelerate resolution of existing plugs
-
Reduce residual inflammatory signalling
But again, this remains in early research stages.
5️⃣ Practical take-home message
At present:
-
Dissolving Charcot–Leyden crystals reduces inflammation in animal models.
-
It is biologically plausible that this could also soften mucus plugs.
-
There is no human clinical proof yet.
-
No approved therapy currently targets the crystals directly.
The concept is scientifically credible — but still under development.
🔭 The Bigger Picture
ABPA is increasingly understood as a condition driven by:
-
Eosinophils
-
Allergic immune signalling
-
Abnormal mucus biology
-
Structural plug formation
Crystal-targeting therapies may eventually become part of a more precise approach to treating eosinophilic airway disease.
But for now, they remain a promising research direction rather than a clinical option.
🔬 Charcot–Leyden Crystals in ABPA and Asthma
What are they? Why do they form? Do they matter?
If you live with Allergic Bronchopulmonary Aspergillosis (ABPA) or severe asthma, you may see the term Charcot–Leyden crystals in a sputum or pathology report.
They can sound worrying.
They are:
-
Not fungus
-
Not infection
-
Not cancer
They are a sign of a particular type of allergic inflammation in the airways.
🧬 What Are Charcot–Leyden Crystals?
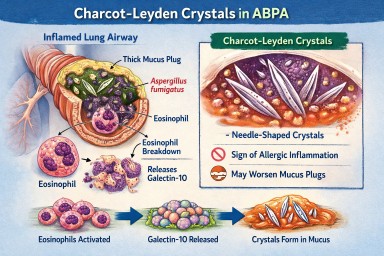
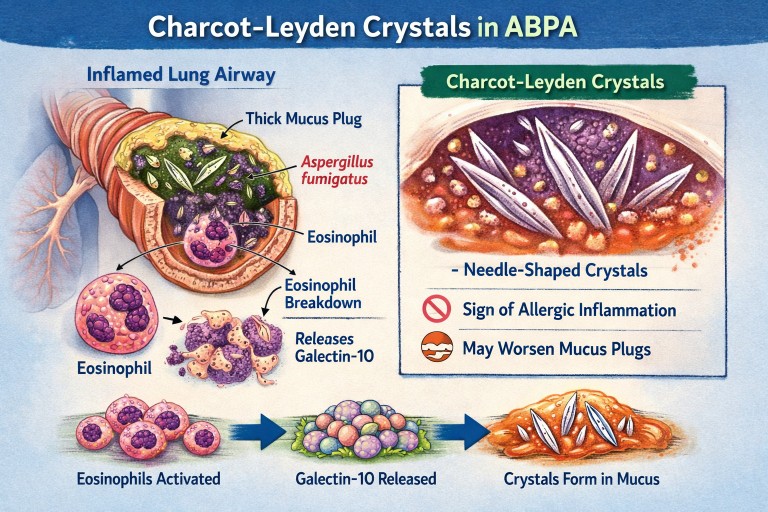
Charcot–Leyden crystals are microscopic, needle-shaped structures found in mucus.
They are made from a protein called galectin-10, which is stored inside a type of white blood cell called an eosinophil.
Eosinophils are immune cells involved in:
-
Allergic asthma
-
ABPA
-
Severe asthma with fungal sensitisation
-
Parasitic infections
When eosinophils are activated and break down, they release galectin-10.
If enough of this protein accumulates in thick airway mucus, it crystallises into visible crystals.
So the crystals are made from your immune cells, not from Aspergillus.
🫁 Why Do They Appear in ABPA?
In ABPA:
-
The immune system overreacts to Aspergillus fumigatus.
-
This triggers a strong allergic (Type 2) immune response.
-
Large numbers of eosinophils move into the airways.
-
Eosinophils break down and release galectin-10.
-
The protein crystallises inside mucus plugs.
The crystals are therefore a footprint of intense allergic inflammation, not fungal invasion.
🌡 Is Most ABPA Eosinophilic?
Yes — almost all classical ABPA is eosinophilic.
ABPA is fundamentally a Type 2 allergic condition, driven by immune pathways involving:
-
IL-4
-
IL-5
-
IL-13
-
IgE
-
Eosinophils
IL-5 in particular stimulates eosinophil production and survival.
Because of this, eosinophils are central to the disease process.
Historically, raised blood eosinophils have been part of diagnostic criteria.
However:
-
Eosinophil counts can fluctuate
-
Steroids can suppress blood levels
-
Eosinophils may still be present in airway mucus even if blood counts appear normal
So ABPA is biologically eosinophilic — even if a single blood test does not show a high count.
True non-eosinophilic ABPA would be unusual and would prompt clinicians to reconsider the diagnosis.
❓ Are Crystals Caused by Aspergillus Infection?
No.
They are caused by the immune reaction to Aspergillus — not by the fungus itself.
They can also be seen in:
-
Severe eosinophilic asthma
-
Parasitic infections
-
Other allergic lung conditions
They reflect eosinophil activity, not fungal growth.
🧠 Why Don’t All People with Asthma Develop These Crystals?
Asthma is not one single disease. It has different inflammatory patterns.
Type 2 (Eosinophilic) Asthma
This involves high eosinophils and allergic pathways.
Common in:
-
Allergic asthma
-
ABPA
-
Severe eosinophilic asthma
These patients can develop Charcot–Leyden crystals.
Non–Type 2 (Non-Eosinophilic) Asthma
This includes:
Neutrophilic asthma
Driven by neutrophils rather than eosinophils.
Paucigranulocytic asthma
Very few inflammatory cells present.
In these forms:
-
Eosinophils are low
-
Galectin-10 is not released in large amounts
-
Crystals are unlikely to form
🧱 Do Charcot–Leyden Crystals Make Mucus Plugs Worse?
Possibly.
Research suggests they may:
-
Increase mucus thickness
-
Contribute mechanically to airway blockage
-
Stimulate further inflammation
For many years they were thought to be harmless debris.
Modern studies suggest they may actively amplify inflammation when present in large amounts.
🎯 Do They Have a Purpose?
Eosinophils evolved mainly to help fight parasitic infections.
Galectin-10 probably has immune signalling roles inside cells.
However, when large amounts are released into thick airway mucus, crystallisation appears to be a by-product of excessive immune activity rather than a useful defence.
In ABPA and allergic asthma, they are more likely part of the problem than part of the solution.
💧 Can Their Formation Be Reduced?
Hydration alone does not stop them forming.
Drinking fluids helps:
-
Keep mucus less sticky
-
Support airway clearance
But it does not prevent eosinophils releasing galectin-10.
What reduces crystal formation?
Reducing eosinophilic inflammation:
-
Corticosteroids
-
Anti-IL-5 biologics
-
Anti-IL-4/IL-13 biologics
When eosinophil numbers fall:
→ Less galectin-10 is released
→ Fewer crystals form
Antifungal treatment in ABPA may indirectly help by reducing allergic stimulation, but the main driver is the immune response.
📊 Do They Change Treatment?
Not directly.
Doctors base treatment on:
-
Symptoms
-
Blood eosinophils
-
Total IgE
-
Imaging
-
Lung function
-
Exacerbation history
Crystals support the diagnosis of eosinophilic inflammation but do not determine treatment alone.
🔎 What Do They Tell Us?
Charcot–Leyden crystals tell us:
-
The airway inflammation is eosinophilic.
-
The immune response is strongly allergic.
-
Mucus plugging risk may be higher.
They are a marker of immune overreaction, not infection severity.
🧠 Key Points to Remember
-
They are made from proteins released by eosinophils.
-
They are not Aspergillus.
-
They do not mean invasive fungal infection.
-
Most classical ABPA is eosinophilic.
-
They are unlikely in non-eosinophilic asthma.
-
Reducing eosinophils reduces their formation.
-
Hydration helps clearance but does not prevent formation.
In simple terms:
Charcot–Leyden crystals are microscopic signs that the immune system is working too hard in the airways.
Event: 📸 Science is Open: A Photo Journey of Research Lab Samples
Have you ever wondered what happens to samples used in research?
The KHP Centre for Translational Medicine is inviting people living with a lung condition to take part in a unique behind-the-scenes experience:
Science is Open: A Photo Journey of Research Lab Samples
This is a rare opportunity to:
-
Go inside a working research laboratory
-
Learn how lung samples are processed and studied
-
Capture the experience through photography
-
Help tell the story of research from a patient perspective
📍 When and Where?
Date: Monday 16th February
Time: Afternoon (exact timing provided after registration)
Location: Central London – Lab tour at Guy's Hospital (London Bridge)
🔬 On the Day You Will:
-
Tour a research lab at Guy’s Hospital
-
Hear directly from researchers about how lung tissue samples are handled and studied
-
Take photographs during the visit (you will be provided with a simple, easy-to-use camera)
-
Receive guidance from a professional photographer
-
Help create a visual story to share with other lung health communities
No previous research or photography experience is needed — just bring yourself.
💷 Reimbursement
-
Travel expenses will be covered
-
Payment for your time: £27.50 per hour (NIHR standard rate)
✉ How to Express Interest
To register your interest, please email Emily and include:
-
A few sentences about your experience with lung health
-
Why you would like to be involved
-
Why you feel your perspective is important
Email Emily to express your interest.
Places are limited and events like this do not come around often.
🌍 Other Opportunities
The team are also running similar events for people living with:
-
Cancer
-
Arthritis
-
Cardiovascular conditions
-
Child and maternal health conditions
If any of these areas are of interest to you, your friends, or family members, please contact Emily for further details.
Event organised by the KHP Centre for Translational Medicine
Shared with thanks to Catherine, Senior Research Impact Officer, Asthma + Lung UK.
How to Join Our Microsoft Teams Meetings - Troubleshooting

(For patients, carers and external guests)
You do not need a Microsoft account to join.
Most people can join easily using their internet browser.
✅ The Easiest Way to Join (Laptop or Desktop)
-
Click the meeting link we sent you.
-
When prompted, choose:
“Continue in this browser”
(You do NOT need to download Teams.) -
Type your name.
-
Click Join now.
-
If asked, allow access to your microphone and camera.
Please wait in the lobby until we admit you.
📱 Joining on a Phone or Tablet
-
Tap the meeting link.
-
If you already have the Microsoft Teams app, it will open automatically.
-
If not, you can download the free Teams app from your app store.
-
Enter your name and join.
Phones often work even if laptops have problems.
⚠️ If You Are Asked to Sign In
You do not need to sign into Microsoft.
If you see a sign-in screen:
-
Look for “Join as guest”
-
Or close the page and reopen the link
-
Or choose “Continue in this browser”
Avoid signing in with a work or NHS account unless you are sure it allows external meetings.
🔧 If It Doesn’t Work on Your Laptop
Try one of these:
-
Open the link in a Private / Incognito window
-
Try a different browser (Chrome or Edge usually work best)
-
Make sure you are not already signed into multiple Microsoft accounts
If you are using a work or NHS laptop, security settings may block external meetings. In that case:
👉 Try your personal laptop or your phone.
🎤 Audio & Camera Tips
-
If your microphone does not work, leave the meeting and rejoin.
-
If you prefer, you can turn your camera off.
-
You can also use the chat box to type questions.
📞 Still Having Trouble?
If you cannot join:
-
Try using your phone instead.
-
Or contact us before the meeting and we will help where we can.
Health effects: why aspergillosis patients are higher-risk
Home › Knowledge Hub › Damp, mould and aspergillosis › Health effects
This page explains what damp and mould can do to the lungs and how to recognise patterns that suggest your home is contributing to symptoms.
How damp buildings can affect the lungs
Damp homes can increase exposure to airborne particles including fungal spores and fragments. For many people this causes irritation; for people with chronic lung disease it can trigger significant exacerbations.
- Worsening cough, wheeze, breathlessness
- Increased mucus and reduced airway clearance
- More frequent chest infections / antibiotic courses
- Allergic-type reactions in sensitised individuals
Why aspergillosis and severe airways disease are different
If you have Aspergillus-related disease or severe asthma/bronchiectasis, your airways may react strongly to fungal material and irritants. Some patients also require systemic or inhaled corticosteroids, which can increase susceptibility to infections and complicate symptom control.
Important nuance: a damp home does not automatically “cause” aspergillosis, but it can be a powerful driver of persistent symptoms and repeated flares.
Patterns that support a housing contribution
- Time-and-place pattern: symptoms worsen at home and improve when away (even partially).
- Multi-person effects: more than one household member develops respiratory/allergic symptoms.
- Post-disturbance worsening: symptoms worsen after “repairs” or “remediation”.
- Escalating medication use: increased reliever inhaler/nebuliser use, repeated steroid bursts, more antibiotics.
Simple symptom timeline template (copy/paste)
Keeping a short, factual timeline helps clinicians and councils understand risk.
Date(s): Where damp/mould is present: What changed (rain event, leak, repair work, return after decant): Symptoms (breathlessness/wheeze/cough/fever/skin/eyes/nose): Medication change (inhalers, steroids, antibiotics): Healthcare use (GP/A&E/hospital): Improves when away from home? (yes/no, how quickly):
What to ask your clinician to document
- Diagnosis (e.g., Chronic pulmonary aspergillosis, allergic bronchopulmonary aspergillosis, asthma, bronchiectasis)
- That symptoms are consistent with environmental triggers or worsened by damp/mould exposure (they do not need to prove causation)
- Any vulnerability factors (steroid use, immunosuppression, reduced lung function)
Damp, mould and aspergillosis in rented homes (UK)
Home › Knowledge Hub › Damp, mould and aspergillosis (UK rented homes)
A comprehensive guide for patients and carers. If you rent your home and worry that damp or mould may be worsening symptoms, these pages explain how to recognise risk, what to do next, and how to escalate safely.
Who this guide is for
- People living with Chronic pulmonary aspergillosis and other long-term lung disease.
- People living with Allergic bronchopulmonary aspergillosis or Severe asthma with fungal sensitisation.
- Carers, family members, and support workers.
- Clinicians and housing professionals seeking a patient-centred overview.
The key message
A damp home does not automatically cause aspergillosis. However, damp and mould can:
- worsen airway inflammation and symptoms
- trigger exacerbations in asthma/bronchiectasis
- increase allergic-type reactions in sensitised people
- make it harder to stabilise symptoms even with optimal treatment
This hub focuses on practical steps: recognising risk early, communicating effectively, understanding remediation quality, and using UK escalation routes.
How to use this hub
- Start with Recognising a damp home to build an evidence base.
- Read Health effects to understand patterns that support an environmental contribution.
- Use Landlord communications to push for a safe plan, not cosmetic fixes.
- Check Remediation & refusal to move if you’re being pressured to return.
- Use Law & support for UK rights and escalation routes.
Important safety note
If you have severe breathlessness, chest tightness, wheeze, or features of anaphylaxis (for example lip/tongue swelling, throat tightness, collapse), seek urgent medical help. If you are repeatedly attending A&E with symptoms that seem worse at home, tell clinicians you are concerned about damp/mould exposure.
Recognising a damp or mouldy home
Home › Knowledge Hub › Damp, mould and aspergillosis › Recognising a damp home
Many high-risk exposures occur before mould is obvious. This page helps you identify early signs and start documenting evidence.
Early warning signs (often missed)
- Condensation on windows most mornings
- Cold, clammy walls or cupboards; wardrobes that feel “damp”
- Persistent musty odour (especially when returning home)
- Peeling wallpaper, bubbling paint, cracking plaster
- Recurring black staining on silicone/grout
- Swollen skirting boards, warped flooring, rusting fittings
High-risk hidden locations
- Behind wardrobes/sofas on external walls
- Inside cupboards on outside walls
- Under sinks, behind washing machines, around toilets/baths
- Window reveals, behind curtains/blinds
- Loft hatches and boxed-in pipework
Common causes (useful when speaking to landlords)
- Water ingress (doors/windows, defective seals, roof, gutters, downpipes)
- Plumbing leaks (slow leaks behind walls or under floors)
- Ventilation failures (broken/weak extract fans, blocked vents)
- Cold bridging and persistent condensation in poorly insulated areas
- Previous flooding/leaks with inadequate drying
Quick evidence checklist (10 minutes)
- Take dated photos of any mould, staining, peeling paint, wet patches.
- Photograph likely sources: door thresholds, window seals, gutters if visible, extractor fans, vents.
- Write down where the smell is strongest and when it’s worst (after rain, in winter, after showers).
- Start a brief symptom note (see Page 3) and keep everything in one folder.
- Report the issue in writing to your landlord/agent and keep screenshots/confirmation.
What not to do (for safety)
- Do not scrape or disturb mouldy plasterboard or insulation yourself.
- Do not rely on bleach-only cleaning as a “solution” (it may not address underlying moisture or embedded contamination).
- Do not accept repeated “paint over and close the ticket” approaches without a cause-and-fix plan.
Your rights, the law, and UK support organisations
Key legal frameworks (plain English)
- Fitness for human habitation: rented homes must be safe and fit to live in. Damp and mould can make a home unfit.
- Local council enforcement: councils can inspect and require action where hazards exist (including damp and mould).
- Social housing (England): stronger timeframes and duties apply for significant damp/mould hazards.
Note: housing law differs across England, Scotland, Wales, and Northern Ireland. Health risks are consistent UK-wide, but escalation routes can vary by nation.
Escalation pathway (practical)
- Landlord/agent (in writing): report damp/mould + request an evidence-based plan (Page 4 templates).
- Formal complaint: ask for escalation to stage 2 / senior review.
- Environmental Health (local council): request inspection for damp/mould hazards if unresolved.
- Ombudsman/regulator route: for social housing complaints after internal process.
- Independent housing advice: Shelter or Citizens Advice can help with wording and next steps.
UK support organisations (start here)
- Shelter (England) — housing rights and escalation support.
- Shelter Scotland
- Shelter Cymru (Wales)
- Housing Rights (Northern Ireland)
- Citizens Advice — practical support and signposting.
- Housing Ombudsman — complaints for social housing providers (after the landlord complaints process).
What you do not need to prove
- You do not need a blood test “proving mould exposure”.
- You do not need to name a specific fungal species.
- You do not need the landlord’s contractor to agree with you.
What matters is credible evidence of a hazard plus a plausible link to health deterioration (especially with clinically vulnerable occupants).
Suggested “resources” box for this hub
These are authoritative starting points you can list at the end of each page (optional):
- UK Government: Damp and mould — health risks and guidance
- UK Government: Awaab’s Law guidance (England, social rented sector)
- UK Government: Fitness for Human Habitation — tenant guide
- UK Parliament: Damp and mould — tenant briefing (England)
- Housing Ombudsman: Damp and mould spotlight report
- Asthma + Lung UK (health advice and support)
Remediation, verification, and refusal to move you
Home › Knowledge Hub › Damp, mould and aspergillosis › Remediation & refusal to move
If remediation is done but symptoms persist or worsen, the key question becomes: has the home been demonstrated to be safe to occupy?
What “good remediation” should include
- Cause fixed: leak/ingress/defect repaired, not just cleaned.
- Drying: adequate drying time and moisture checks.
- Material decisions: water-damaged porous materials removed where needed.
- Safe work: dust/spore spread controlled (important for medically vulnerable households).
- Verification: documented checks that work is complete and the home is safe.
Red flags (“bad remediation”)
- Paint over staining or “mould spray” only
- No drying plan, no moisture measurements
- No documentation of what was removed/replaced
- Work that creates dust without protection/containment
- Refusal to provide any meaningful post-remediation checks
If symptoms worsen after remediation
Worsening symptoms can occur if contaminated materials were disturbed or if damp persists behind walls/floors. This is a strong indicator that the hazard may not be resolved.
Actions:
- Document symptoms and healthcare visits (Page 3 template).
- Ask landlord for written evidence of safety and remediation details.
- Request escalation to Environmental Health if unresolved.
If the landlord refuses to move you (decant)
Use this framing:
- The issue is not “repairs completed” — it is safety and health risk.
- Ask: “What evidence shows the home is safe to occupy?”
Template request for temporary alternative accommodation
Subject: Request for temporary alternative accommodation (health risk / damp and mould) Hello [Name/Team], Despite remediation work, we are experiencing ongoing damp/mould concerns and worsening health symptoms in a clinically vulnerable household. Please provide written evidence that the home is safe to occupy, including: - confirmation the moisture source has been resolved, - evidence of drying/moisture checks, - what materials were removed/replaced, - what post-remediation checks were completed. Given the uncertainty around safety and the health impacts, we are requesting temporary alternative accommodation until the property can be demonstrated to be safe to occupy. Kind regards, [Name]
If you return “under protest”
If you have no alternative but to return, keep it in writing:
We are returning to the property due to lack of alternative accommodation. We do not accept that the damp/mould hazard has been resolved and will continue to document health impacts and seek independent assessment.
How to raise damp and mould with your landlord (and get action)
Home › Knowledge Hub › Damp, mould and aspergillosis › Raising with your landlord
Your aim is to secure a safe, evidence-based plan: fix the cause, dry properly, remediate safely, and confirm the home is safe to occupy.
Principles that prevent “cosmetic fixes”
- Source control: stop the leak/ingress/defect.
- Drying: dry building fabric, not just the air.
- Safe remediation: remove/clean contaminated materials appropriately.
- Verification: show the home is safe to occupy.
How to report effectively
- Report in writing (email/portal) and keep everything.
- Attach dated photos and a brief symptom timeline.
- Use health-focused language: “damp and mould hazard”, “medical vulnerability”, “safe to occupy”.
- Ask for timescales, named contact, and written findings.
Template email (copy/paste)
Subject: Damp and mould hazard – urgent investigation and repair plan (health impact) Hello [Landlord/Housing Officer/Letting Agent], I am reporting ongoing damp and/or mould at [address], affecting [rooms/locations]. This has been present since [date] and appears linked to [water ingress/leak/condensation/ventilation failure]. We have clinically vulnerable occupants in the household, including [brief: chronic lung disease / aspergillosis / severe asthma / bronchiectasis], and symptoms are worsening. Please confirm in writing: 1) the inspection date and who will attend, 2) the findings (including likely cause), 3) the repair and drying plan (including timeframes), 4) what remediation will be undertaken (not just surface cleaning/painting), 5) how you will confirm the property is safe to occupy once works are completed. Thank you, [Name] [Phone]
If you are told it is “lifestyle” or “just condensation”
You can reply calmly:
Thank you. We are doing reasonable ventilation and heating measures. However, the pattern and location suggest a building/ventilation defect that requires investigation and repair. Given medical vulnerability in the household, we need a written plan that addresses the underlying cause and confirms the home is safe to occupy.
When to escalate early
- Repeated failed repairs, or mould returns quickly
- Visible mould plus long-term damp patches
- Health deterioration, repeated GP/A&E attendances
- Pressure to accept repainting/bleach-only cleaning
See Page 6 for UK escalation routes and support organisations.