Antifungal Medicines: Dosing, Monitoring, and the Role of Specialist Care
A detailed reference for patients and non-specialist clinicians
1. Why antifungal treatment is different from most medicines
Oral antifungal medicines—especially azole antifungals—are essential for treating long-term fungal diseases such as chronic pulmonary aspergillosis and allergic bronchopulmonary aspergillosis.
They differ from many common medicines because they:
-
Have a narrow margin between effectiveness and toxicity
-
Behave very differently between individuals
-
Are often taken for months or years, not days
-
Interact with many commonly prescribed drugs
For these reasons, antifungal treatment requires individualised dosing, monitoring, and specialist input, rather than a standard fixed dose.
2. What “pharmacokinetics” means (plain language)
Pharmacokinetics describes what the body does to a drug:
-
Absorption – how well the drug enters the bloodstream from the gut
-
Distribution – how effectively it reaches tissues such as the lungs
-
Metabolism – how quickly the liver breaks it down
-
Elimination – how the drug leaves the body
Differences at any of these stages explain why the same dose can be ineffective for one person and toxic for another.
3. Different generations of azole antifungals behave differently
Each generation of azole antifungal was designed to improve effectiveness, but chemical changes also altered how the body handles the drug.
First-generation azoles (older drugs)
Examples
-
Ketoconazole
-
Fluconazole (limited activity against Aspergillus)
Key features
-
Variable absorption
-
Shorter half-life
-
Less reliable lung penetration
Clinical relevance
-
Rarely used now for chronic aspergillosis
Second-generation azoles (mainstay treatment)
Examples
-
Itraconazole
-
Voriconazole
-
Posaconazole
Key features
-
Excellent lung and tissue penetration
-
Highly variable metabolism between people
-
Strong interaction with liver enzymes
Clinical relevance
-
Very effective
-
Blood levels vary widely
-
Dose adjustment and monitoring are often essential
Newer azoles
Example
-
Isavuconazole
Key features
-
More predictable absorption
-
Long, stable half-life
-
Fewer extreme peaks and troughs
Clinical relevance
-
Often better tolerated long-term
-
Monitoring still important, but dosing may be more stable
4. Why the “right dose” matters so much
Too little antifungal
-
Infection not adequately controlled
-
Symptoms persist or worsen
-
Risk of antifungal resistance
-
Fewer future treatment options
Too much antifungal
-
Liver irritation or damage
-
Nausea, appetite loss
-
Neurological or visual side effects
-
Drug accumulation, especially with long-term use
The aim is always the lowest dose that effectively controls the fungus.

5. How clinicians know whether the dose is right
No single test determines this. The correct dose is identified when three elements align:
1️⃣ Blood level testing (therapeutic drug monitoring)
-
Measures how much drug is actually in the bloodstream
-
Helps identify:
-
Under-dosing
-
Target-range dosing
-
Toxic levels
-
2️⃣ Clinical response
-
Symptoms stabilise or improve
-
Fewer flare-ups or complications
-
Better day-to-day function
3️⃣ Safety monitoring
-
Liver and kidney blood tests
-
Review of side effects
-
Ongoing assessment of drug interactions
Only when effectiveness and safety are both acceptable is the dose considered “right”.
6. Why the right dose can change over time
A dose that was correct initially may later need adjustment because of:
-
Weight or body-composition changes
-
Age-related metabolic changes
-
New medications (including antibiotics or steroids)
-
Changes in liver or kidney function
-
Gradual drug accumulation during long-term therapy
Regular review is therefore expected and appropriate.
7. Is it sometimes impossible to find a stable dose?
Yes. For a minority of patients, a perfectly balanced dose cannot be found.
Reasons include:
-
Extremely fast or slow drug metabolism
-
A very narrow safety window
-
Long-term toxicity despite “acceptable” blood levels
-
Unavoidable interacting medications
-
Liver, kidney, or neurological vulnerability
-
Partial or full antifungal resistance
In these cases, the dose that controls the fungus and the dose that causes side effects may overlap.
This reflects biological limits, not treatment failure.
8. What clinicians do when a stable dose cannot be achieved
Options may include:
-
Switching to a different azole with different pharmacokinetics
-
Using modified dosing schedules (split dosing, slower titration)
-
Accepting a lower suppressive dose rather than full eradication
-
Considering non-azole antifungals where appropriate
-
Prioritising symptom control and quality of life
All are intentional, safety-focused decisions.
9. The central role of the specialist pharmacist
Specialist pharmacists are key to safe antifungal care, particularly for long-term azole therapy.
They play a critical role in:
Interpreting drug levels
-
Assessing whether a level is truly low or high
-
Accounting for dose timing and formulation
-
Preventing unnecessary or unsafe dose changes
Managing drug–drug interactions
Azoles interact with many common medicines, including:
-
Steroids and inhalers
-
Heart rhythm drugs
-
Blood thinners
-
Anti-epileptics
-
Pain medications
The specialist pharmacist:
-
Reviews the full medication list
-
Anticipates interactions before harm occurs
-
Advises on adjusting both interacting drugs
Individualising dosing
When standard doses do not work, they help design:
-
Non-standard doses
-
Split dosing schedules
-
Slow titration plans
-
Alternative azoles with different pharmacokinetics
Protecting patients during long-term treatment
They monitor:
-
Trends in liver and kidney tests
-
Signs of cumulative toxicity
-
Whether symptoms may be drug-related rather than disease-related
Coordinating care
They act as a bridge between:
-
Laboratory results
-
Clinical decision-making
-
Patient experience

Their involvement often changes management, not just fine-tunes it.
10. Where antifungal drug level testing is done in the UK
In the UK, antifungal drug level testing is centralised.
-
Blood samples are taken locally
-
Samples are sent to specialist reference laboratories, most commonly the
Mycology Reference Centre Manchester -
Results are returned to the local clinical team for interpretation
Patients managed through specialist services such as the
National Aspergillosis Centre
benefit from integrated expertise in antifungal pharmacology, imaging, and long-term monitoring.
This process is routine and standard for antifungal care.
11. Key reassurance for patients
-
Dose changes are normal and expected
-
Side effects are often biology-driven, not your fault
-
Blood tests make treatment safer, not riskier
-
Switching drugs is a planned strategy, not giving up
12. One-paragraph summary
Antifungal medicines—particularly azole antifungals—have complex and highly variable behaviour in the body, with a narrow balance between effectiveness and toxicity. Safe use requires individualised dosing, therapeutic drug monitoring, symptom review, and long-term safety checks. Specialist pharmacists play a central role in interpreting drug levels, managing interactions, and tailoring treatment. For some patients, a perfectly balanced dose cannot be achieved, and alternative strategies are required. This reflects biological complexity, not failure, and the overarching aim is always effective fungal control with the best possible long-term safety and quality of life.
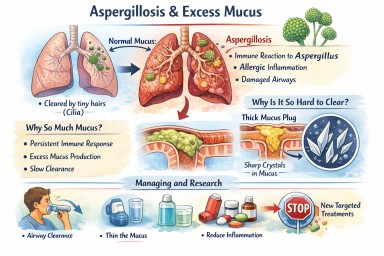
Airways mucus and aspergillosis
A clear, patient-friendly explainer
People living with aspergillosis often say that mucus is one of the hardest symptoms to manage — thick sputum, coughing fits, plugs that feel “stuck”, and flare-ups that seem to come out of nowhere. This explainer brings everything together in one place: what mucus is for, why aspergillosis causes so much of it, why it becomes abnormal, and what current and future treatments aim to do.
1. What is airway mucus and why do we need it?
Mucus is normal, healthy, and essential. Everyone produces it all the time.
Its main roles are to:
-
Trap inhaled particles (dust, spores, bacteria, pollution)
-
Protect the airway lining from drying and irritation
-
Support the immune system
-
Clear the lungs, using tiny moving hairs (cilia) that sweep mucus upwards so it can be swallowed or coughed out
(this clearance system is called the mucociliary escalator)
In healthy lungs:
-
Mucus is thin
-
Produced in small amounts
-
Cleared without you noticing it
2. Why aspergillosis causes excessive mucus
In aspergillosis, the lungs are under ongoing stress. Several factors combine:
Persistent immune activation
The immune system keeps reacting to Aspergillus material in the airways. Even when the fungus is controlled, inflammation can persist.
Allergic-type inflammation (especially in ABPA)
Allergic immune responses strongly stimulate mucus-producing cells, leading to:
-
Large volumes of mucus
-
Very sticky or rubbery sputum
Airway damage
Conditions commonly associated with aspergillosis (such as bronchiectasis or long-standing asthma) cause:
-
Widened or damaged airways
-
Poor mucus clearance
-
Pools of mucus that are hard to shift
Slowed clearance
Inflammation and infection impair cilia, so mucus:
-
Moves more slowly
-
Sits in the lungs longer
-
Becomes thicker and harder to clear
➡️ What starts as a protective response becomes a self-perpetuating problem.
3. Why thick mucus causes symptoms
Excess or abnormal mucus can:
-
Block airways → breathlessness and wheeze
-
Trigger coughing → especially overnight or on waking
-
Trap infection → repeated flare-ups
-
Reduce oxygen exchange
-
Increase fatigue and chest discomfort
Many patients describe it as:
“Glue-like”, “stringy”, “rubbery”, or “impossible to move”
4. Mucus plugs and crystals – why some mucus is so hard to clear
Mucus plugs
When mucus becomes very thick, it can:
-
Form plugs that partially or completely block airways
-
Show up on CT scans
-
Worsen breathlessness suddenly
Charcot–Leyden crystals
In allergic and eosinophilic airway disease (including allergic bronchopulmonary aspergillosis):
-
Breakdown products of allergic immune cells can form microscopic crystals
-
These crystals make mucus:
-
Stiffer
-
More irritating
-
Harder to clear
-
Their presence is a sign of ongoing allergic inflammation, not infection alone.
5. Why managing mucus really matters
Mucus is not just an inconvenience. Poor mucus control can:
-
Increase infection risk
-
Drive repeated exacerbations
-
Worsen lung damage over time
-
Reduce quality of life and sleep
-
Increase hospital admissions
For aspergillosis, mucus management is core treatment, not optional.
6. What helps now (current approaches)
A. Thin the mucus
-
Good hydration
-
Nebulised saline (normal or hypertonic)
-
Selected mucolytic medicines (used carefully)
B. Move it out
-
Regular airway clearance physiotherapy
-
Breathing techniques (e.g. active cycle breathing)
-
Oscillating devices (flutter, Acapella, Aerobika)
-
Gentle, regular physical activity where possible
C. Reduce inflammation
-
Inhaled corticosteroids (when appropriate)
-
Oral steroids (used cautiously)
-
Biologic therapies for selected allergic or eosinophilic disease
-
Antifungal treatment when fungal burden is contributing
D. Treat infections early
-
Bacterial infections thicken mucus further
-
Prompt treatment reduces long-term damage
7. What research is doing differently (emerging therapies)
Research is moving beyond simply “loosening mucus”.
1. Reducing mucus production at source
Scientists are developing drugs that aim to:
-
Switch off excessive mucus secretion
-
Preserve normal protective mucus
This targets the mucus-producing cells directly.
2. Blocking the signals that drive over-production
Inflammation sends chemical signals telling airways to make more mucus. New treatments aim to:
-
Calm allergic and immune pathways
-
Prevent expansion of mucus-producing cells
Some current biologic therapies already reduce mucus indirectly; future drugs will be more precise.
3. Changing mucus structure
Instead of thinning everything, researchers are studying ways to:
-
Loosen the internal “mesh” of mucus
-
Prevent dense plugs from forming
-
Restore normal movement by cilia
4. Targeting mucus crystals
In allergic aspergillosis, research is exploring how to:
-
Reduce crystal formation
-
Calm the specific immune responses that create them
5. New inhaled and physical approaches
Early trials are testing:
-
Inhaled therapies designed to mobilise secretions
-
Treatments that improve airflow behind mucus plugs
6. Precision medicine
Future mucus treatments are likely to be:
-
Personalised
-
Based on inflammation type, fungal involvement, airway damage, and immune markers
Two people with aspergillosis may have very different mucus drivers — and need different solutions.
8. What this means for patients today
-
There is no single “anti-mucus cure” yet
-
Promising therapies are in research and early trials
-
Safety and long-term effects must be proven first
For now:
-
Regular airway clearance remains essential
-
Treating inflammation and infection promptly is crucial
-
Understanding why your mucus behaves as it does helps guide treatment
Key messages to remember
-
Mucus is normally protective
-
Aspergillosis turns a helpful system into a problem
-
Thick, sticky mucus reflects ongoing inflammation and airway damage
-
Crystals signal allergic involvement, not just infection
-
Research is moving toward preventing abnormal mucus formation, not just thinning it
Latest Aspergillosis & Related Research Updates (Week 3).
January–February 2026
Search term is 'aspergillosis'.
This update highlights recent publications relevant to aspergillosis, allergic bronchopulmonary aspergillosis, nontuberculous mycobacterial lung disease, antifungal stewardship, diagnostics, and environmental fungal exposure. Papers are grouped by clinical theme, with key findings and clinical relevance highlighted.
1. Diagnostics, Molecular Methods & Imaging Innovation
Clinical Characteristics, Molecular Diagnosis, and Drug Resistance Profiles of Nontuberculous Mycobacteria Infections
Wang K, Xu D, Gao Y, Zhao W, Ma K
Clinical and Translational Science, 19(2):e70479, Feb 2026
Key highlights
-
Retrospective analysis using polymerase chain reaction melting curve technology to identify nontuberculous mycobacterial species.
-
Demonstrates rapid differentiation of clinically relevant species, with integrated resistance profiling.
-
Highlights marked heterogeneity in clinical presentation and antimicrobial resistance patterns.
Why this matters
-
Increasing relevance for patients with bronchiectasis, chronic obstructive pulmonary disease, and aspergillosis, where nontuberculous mycobacteria co-infection complicates diagnosis and treatment.
-
Supports the shift away from prolonged culture-only pathways toward faster molecular diagnostics.
Amplicon-based sequencing as a diagnostic tool for severe pneumonia in the ICU
Michel C, Imamura H, Yin N, et al.
Scientific Reports, 16(1):2845, Jan 2026
Key highlights
-
Amplicon-based sequencing applied directly to respiratory samples in intensive care.
-
Detects invasive aspergillosis alongside bacterial and viral pathogens.
-
Highlights limitations of current definitions of “proven invasive aspergillosis” when relying solely on histopathology.
Why this matters
-
Reinforces the diagnostic gap in critical care–associated pulmonary aspergillosis.
-
Supports broader adoption of molecular and microbiome-informed diagnostics in high-risk settings.
Deep learning detection and classification of fungal and non-fungal calcifications on paranasal sinus CT imaging
Yang Z, Choi I, Yun H, et al.
PLOS One, 21(1):e0340832, Jan 2026
Key highlights
-
Deep learning model distinguishes fungal ball (commonly aspergillosis) from non-fungal calcifications.
-
High diagnostic accuracy on routine sinus computed tomography scans.
-
Addresses a frequent diagnostic uncertainty in chronic rhinosinusitis.
Why this matters
-
Potential to reduce diagnostic delay and unnecessary surgery.
-
Particularly relevant for centres without ready access to specialist radiology expertise.
2. Invasive Aspergillosis: Expanding Risk Profiles & Clinical Phenotypes
Unmasking Invasive Pulmonary Aspergillosis: Insights From a Case Series at a Tertiary Care Center
Munasinghe K, Nanayakkara A, De Zoysa W, et al.
Cureus, 17(12), Jan 2026
Key highlights
-
Case series illustrating heterogeneous clinical presentations.
-
Emphasises delayed recognition outside classic immunocompromised populations.
-
Reinforces global incidence estimates of approximately 250,000 cases annually.
Why this matters
-
Supports growing recognition that invasive pulmonary aspergillosis occurs in broader patient groups, including those with chronic lung disease and critical illness.
Disseminated Invasive Aspergillosis in a Young Patient With Chronic Alcohol Use and Seemingly Preserved Immunocompetence
Khandwala K, Sawliha Syed H, Anwar S, et al.
Clinical Case Reports, 14(2), Jan 2026
Key highlights
-
Disseminated disease involving multiple organs.
-
Chronic alcohol use identified as a functional immunosuppressive state.
-
Challenges traditional “immunocompetent vs immunocompromised” dichotomy.
Why this matters
-
Reinforces the need for high clinical suspicion even when standard immune markers appear preserved.
-
Relevant for emergency, acute medical, and respiratory teams.
Intensification of Treosulfan–Fludarabine Conditioning With Thiotepa in Allogeneic Hematopoietic Stem Cell Transplantation
Tosoni L, Facchin G, Plos R, et al.
Transplant Direct, 12(2):e1896, Jan 2026
Key highlights
-
Real-world study in older or comorbid transplant recipients.
-
Reports four cases of invasive aspergillosis (three pulmonary, one cerebral).
-
Conditioning regimen was otherwise effective and tolerable.
Why this matters
-
Reinforces persistent invasive fungal infection risk despite modern conditioning approaches.
-
Supports ongoing need for antifungal prophylaxis and surveillance.
Infective Endocarditis Caused by Pan-Azole-Resistant Aspergillus fumigatus in a Lung Transplant Recipient
Ukai K, Kawashima M, Ikeuchi K
Transplant Infectious Disease, Jan 2026
Key highlights
-
Rare but severe manifestation: fungal endocarditis.
-
Pan-azole resistance significantly limited treatment options.
-
Occurred in a lung transplant recipient.
Why this matters
-
Adds to evidence of clinically catastrophic azole resistance.
-
Reinforces importance of resistance testing and antifungal stewardship.
3. Antifungal Toxicity & Stewardship
Voriconazole-associated peripheral polyneuropathy: A case report
González BJ, Ivarola P, Miranda M, et al.
Archivos Argentinos de Pediatría, 124(1), Feb 2026
Key highlights
-
Documents peripheral neuropathy linked to prolonged voriconazole exposure.
-
Emphasises reversibility only after early recognition and drug withdrawal.
Why this matters
-
Highly relevant for patients on long-term antifungal therapy for chronic pulmonary aspergillosis.
-
Supports routine neurological symptom surveillance.
Antifungal Stewardship: Time to Reappraise the Priorities toward Increasing Invasive Fungal Infections
Singh S
Annals of African Medicine, Jan 2026
Key highlights
-
Reviews stewardship challenges across aspergillosis, candidemia, and mucormycosis.
-
Highlights overuse, under-diagnosis, and limited access to diagnostics.
-
Calls for stewardship frameworks equivalent to antibacterial programmes.
Why this matters
-
Directly relevant to azole resistance, drug toxicity, and resource-limited settings.
-
Aligns with national and international fungal disease priorities.
4. Allergy, Mycotoxins & Inflammatory Pathways
Common inflammatory markers predict risk of ABPA development in children with cystic fibrosis
Crabtree HED, Malajczuk CJ, Ho HY, et al.
Journal of Cystic Fibrosis, Jan 2026
Key highlights
-
Identifies routinely measured inflammatory markers predictive of allergic bronchopulmonary aspergillosis.
-
Potential for earlier identification and intervention.
Why this matters
-
May support risk stratification in paediatric cystic fibrosis clinics.
-
Relevant for future screening and monitoring protocols.
Potential mechanisms and effects of AFB1-induced asthma
Yu Z, Gao M, Wu X, et al.
PLOS One, 21(1):e0341172, Jan 2026
Key highlights
-
Network toxicology and molecular docking suggest links between aflatoxin B1 exposure and:
-
Asthma
-
Allergic bronchial pulmonary aspergillosis
-
Lung malignancy in severe cases
-
Why this matters
-
Strengthens environmental and occupational health links to fungal allergy and chronic lung disease.
-
Supports broader discussion of mould exposure beyond infection alone.
Mycotoxins – Determination of aflatoxins, ochratoxin A, gliotoxin, and others in urine by LC–MS/MS
Berger M, Deharde M, Neuhoff J, et al.
MAK Collection for Occupational Health and Safety, 10(2), Jan 2026
Key highlights
-
Validated biomonitoring method for gliotoxin, aflatoxins, and ochratoxins.
-
Discusses potential use of urine biomarkers for early detection of invasive aspergillosis.
Why this matters
-
Provides methodological groundwork for future biomarker-driven diagnostics.
-
Particularly relevant for occupational and environmental exposure assessment.
Money and Microbes: A Global Systematic Review and Meta-Analysis of Currency Contamination
Appiah PO, Odoom A, Tetteh-Quarcoo PB, Donkor ES
Environmental Health Insights, Jan 2026
Key highlights
-
Identifies paper currency as a reservoir for microbial and fungal contamination.
-
Notes links to serious infections, including pulmonary aspergillosis.
Why this matters
-
Highlights overlooked environmental reservoirs of fungal exposure.
-
Relevant for public health messaging and infection control.
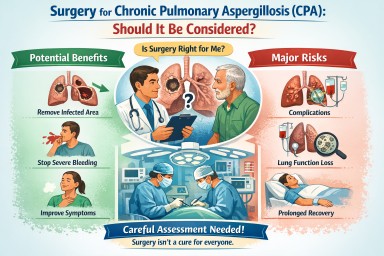
Surgery for Chronic Pulmonary Aspergillosis (CPA): why it is sometimes considered – and often not
For people living with chronic pulmonary aspergillosis (CPA), the idea of surgery can raise difficult questions. Some patients are told surgery might offer a chance of cure; others are advised very firmly against it. Both positions can be correct, depending on the individual situation.
This article explains when surgery may be considered, why it is often avoided, and what “success” or “cure” really means in CPA.
Why is surgery even considered in CPA
CPA usually develops in lungs that are already damaged (for example, by tuberculosis, chronic obstructive pulmonary disease, bronchiectasis, sarcoidosis, or prior infections). Antifungal medicines are therefore the mainstay of treatment.
However, surgery may be considered in a small and carefully selected group of patients, most commonly when:
1. Disease is localised to one area of the lung
If the aspergillus infection is confined to a single cavity or one lobe, and the rest of the lungs are relatively healthy, it may be technically possible to remove the affected area.
2. Recurrent or life-threatening haemoptysis (coughing up blood)
Large-volume or repeated bleeding is one of the strongest reasons surgery is considered. In some cases, surgery is viewed as a way to prevent catastrophic bleeding, rather than to eradicate infection.
3. A simple aspergilloma
Patients with a simple aspergilloma (a single fungal ball in a cavity, minimal surrounding disease, and preserved lung function) are the group most likely to benefit.
4. Failure or intolerance of antifungal therapy
If antifungal drugs cannot be taken long term due to side effects, drug resistance, or lack of response—and the disease remains localised—surgery may be discussed.
Why surgery is often not recommended
Although surgery can sound appealing, CPA surgery is high-risk and not suitable for most patients.
1. CPA is often widespread
Many patients have a disease affecting both lungs or multiple lobes. Removing one area does not treat the remaining infection.
2. Underlying lung reserve is limited
CPA commonly occurs in people with reduced lung function. Removing lung tissue can lead to:
-
Long-term breathlessness
-
Oxygen dependence
-
Reduced quality of life
Even if the operation itself is technically successful.
3. Surgery carries significant risks
Compared with many other lung operations, CPA surgery has higher complication rates, including:
-
Prolonged air leaks
-
Serious infections
-
Bleeding
-
Bronchopleural fistula (abnormal airway–pleural connection)
-
Need for prolonged hospitalisation or intensive care
4. Surgery does not address the underlying vulnerability
CPA reflects an ongoing susceptibility of the lung environment. Removing one fungal focus does not remove the underlying reason aspergillus was able to grow in the first place.
What is the “success rate” of surgery?
Success depends heavily on patient selection and surgical expertise.
In specialist centres:
-
Operative mortality (risk of death around the time of surgery):
Typically reported between 1–5%, but higher in complex diseases. -
Major complication rates:
Often 15–40%, depending on disease extent and lung health. -
Symptom improvement:
Many patients selected for surgery experience reduced haemoptysis and improved local control of disease.
These figures are why surgery is only offered after careful multidisciplinary discussion, usually involving respiratory physicians, infectious disease specialists, thoracic surgeons, and radiologists.
Is surgery a “cure” for CPA?
This is one of the most misunderstood points.
Short answer: sometimes, but often not in the long term
-
In a simple aspergilloma, surgery can be genuinely curative if:
-
The disease is completely removed
-
There is no other active CPA elsewhere
-
The patient’s lungs remain stable
-
-
In chronic cavitary or fibrosing CPA, surgery is rarely a true cure. Instead, it may:
-
Control bleeding
-
Remove a particularly problematic area
-
Reduce fungal burden
-
Even after apparently successful surgery, some patients still require:
-
Long-term antifungal therapy
-
Ongoing monitoring with scans and blood tests
Recurrence of aspergillus infection elsewhere in the lungs can occur months or years later.
Why are many patients managed medically instead
For most people with CPA, long-term antifungal therapy offers:
-
Disease stabilisation
-
Symptom control
-
Lower risk than surgery
While antifungals do not usually “cure” CPA either, they can:
-
Slow or halt progression
-
Reduce inflammation and symptoms
-
Improve quality of life
This is why surgery is best seen as a highly selective tool, not a standard treatment.
How decisions about surgery are made
If surgery is discussed, your team will usually consider:
-
Extent and pattern of CPA on imaging
-
Lung function tests
-
General fitness and other medical conditions
-
History of haemoptysis
-
Response and tolerance to antifungal treatment
-
Your own priorities and acceptable trade-offs
Importantly, being told surgery is not advised does not mean your care is being limited—it usually reflects a judgement that risks outweigh benefits in your specific case.
Key messages for patients
-
Surgery for CPA is uncommon and highly selective
-
It is most useful in localised disease or severe bleeding
-
Complication rates are significant
-
A guaranteed or permanent “cure” is not typical, except in carefully chosen cases
-
Long-term medical management remains the safest and most effective option for most patients
If surgery has been mentioned—or ruled out—in your case, it is reasonable to ask your team:
-
What specific problem would surgery aim to solve for me?
-
What risks apply to my lungs and overall health?
-
Would antifungal treatment still be needed afterwards?
These discussions are an important part of shared decision-making in CPA care.
Connecting patients, carers, clinicians and scientists to improve life with aspergillosis
World Aspergillosis Day (WAD) is an annual global event that brings together people who live with, care for, treat, and research long-term forms of aspergillosis — particularly chronic pulmonary aspergillosis (CPA) and allergic bronchopulmonary aspergillosis (ABPA).
Each year, WAD creates a shared space where:
-
patients and carers can hear directly from specialists,
-
clinicians and scientists can learn from patient experience,
-
and everyone can explore how new research translates into better care.
🎥 Missed previous events?
Recordings from earlier World Aspergillosis Day meetings are available on our YouTube channel.
📅 NAC World Aspergillosis Day Meeting 2026
The National Aspergillosis Centre (NAC) will once again host a free online meeting:
🗓 Tuesday 3 February 2026
💻 Online via Microsoft Teams
👥 Open to patients, carers, clinicians, scientists, and anyone who lives or works with aspergillosis
🧬 This year’s theme:
“How can the genomics revolution help patients with chronic aspergillosis?”
Why genomics — and why now?
Modern molecular tests such as PCR and DNA sequencing are becoming faster, cheaper and more accurate. Because of this, the NHS is increasingly exploring how genomic technologies can be used to improve diagnosis, monitoring and treatment across many diseases — including aspergillosis.
This year’s WAD meeting will start an open discussion between patients and professionals about which genomic and molecular tests are likely to matter most for people with aspergillosis in the years ahead.
Topics will include:
-
🧠 Is there a “gene for aspergillosis”?
Should people be tested for genetic susceptibility? -
💊 Genes and voriconazole dosing
Can testing the CYP2C19 gene help personalise antifungal treatment? -
🦠 Tracking antifungal resistance
How molecular testing of Aspergillus strains can help hospitals monitor resistance. -
🔬 Aspergillus PCR at NAC
How PCR is already used to diagnose and monitor chronic aspergillosis.
🗣️ Patient voices at the heart of the meeting
As always, patient experience will be central to the day.
This year will include new patient stories, including Alison, who will talk about how her aspergillosis treatment led to the development of adrenal insufficiency, and what that has meant for her care and daily life.
“I don’t know anything about genetics — is this for me?”
Absolutely yes.
You don’t need any background in genetics to take part. Everything will be explained clearly, step by step, with minimal jargon.
Planned discussion topics include:
-
What do my Aspergillus PCR test results actually mean?
-
Is there really a “gene for CPA”?
-
Why do genes matter for antifungal dosing?
In fact, the more questions you ask — especially the “silly” ones — the better. The discussion from the day will be used to create a new patient leaflet, designed to help people better understand their diagnosis and test results.
✅ Registration is now open
🎟 Book your free place via Eventbrite:
👉 www.eventbrite.co.uk/e/world-aspergillosis-day-tickets-1980707139373
💻 Joining via Microsoft Teams
The meeting will be held online using Microsoft Teams, which you can download here:
👉 www.microsoft.com/en-gb/microsoft-teams/group-chat-software
If you haven’t used Teams before, we recommend doing a test call in advance. If you run into any problems setting things up, we’re very happy to help.
We hope you can join us for World Aspergillosis Day 2026 — to learn, to ask questions, and to help shape the future of aspergillosis care together.
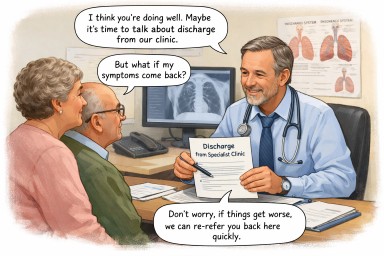
When discharge from a specialist service is being discussed
A reassuring explanation for people with long-standing Aspergillus bronchitis
This page is for people who have lived for many years with a diagnosis of Aspergillus bronchitis, and who are now hearing that discharge from a specialist or tertiary service may be discussed, or is being gently considered.
Many patients tell us this brings up worries such as:
-
“Does this mean I’m less safe?”
-
“Does this mean they’re not sure anymore?”
-
“What if things get worse later?”
These feelings are very common, and they make sense.
First — nothing has been decided yet
If discharge is being discussed, it usually means:
-
Your team is reviewing your care carefully
-
They are looking at whether regular specialist follow-up is still helping right now
-
They are not withdrawing care, and not closing doors
Discussion is part of good medicine — especially with conditions that can change slowly over time.
Why might discharge even come up after many years?
This can feel surprising, but it is usually because:
-
Your condition has been stable for a long time
-
There has been no clear progression
-
Specialist treatments are not currently being changed
-
Ongoing follow-up may not be adding extra benefit at this stage
This is often a sign of relative stability, not doubt or disbelief.
Does this mean they think you never had Aspergillus bronchitis?
No — not at all.
What it usually means is:
-
Aspergillus bronchitis was a reasonable and helpful way to understand your symptoms at the time
-
Over time, the balance has shifted
-
Aspergillus may now be less active or less central to how you are feeling
Medical understanding evolves, and long-term conditions often change their shape rather than disappear or suddenly become “wrong”.
Does discharge mean Aspergillus is no longer important?
Not exactly.
It usually means:
“We don’t think Aspergillus is the main thing driving your symptoms right now.”
It does not mean:
“Aspergillus will never matter again.”
Your specialists know that Aspergillus-related problems can:
-
Fluctuate
-
Become more relevant during periods of illness or change
-
Need revisiting later on
That possibility is built into discharge planning, even if it is not always said clearly.
Why does this still feel unsettling?
Because specialist care often feels like a safety net.
You may have felt:
-
Known and understood by the team
-
Reassured by specialist oversight
-
Protected by regular review
Thinking about discharge can feel like losing that protection — even when nothing is actually changing day to day.
That emotional response is completely understandable.
What discharge from a specialist service usually does mean
If discharge does happen, it usually means:
-
Your care continues with your GP or respiratory team
-
Your history does not disappear
-
You are not starting from scratch
-
Re-referral is expected if things change
Specialist teams rarely intend discharge to be permanent or final.
What about being re-referred if things worsen?
This is one of the most important points — and a reassuring one.
In most cases:
-
Re-referral is anticipated
-
Patients previously known to the service are often reviewed more quickly
-
You do not need to “prove” everything again
Discharge usually comes with an open door, even if that door is not labelled as such.
What helps patients feel safer at this stage
It is reasonable to want:
-
A clear explanation of why discharge is being discussed
-
Reassurance that this is about now, not forever
-
Clarity about what would prompt a return
-
Confidence that your GP knows your history
These are normal needs — not demands.
What you might gently ask your team
You could ask:
-
“If my symptoms change, would re-referral be straightforward?”
-
“What sort of changes should prompt a review?”
-
“Will my GP have clear guidance from you?”
-
“Is discharge something we can review over time?”
These questions often help turn uncertainty into reassurance.
Key things to hold onto
-
Discussion of discharge usually reflects stability, not dismissal
-
It does not mean your past diagnosis was wrong
-
It does not mean you are being left unsupported
-
Re-referral is part of good planning, not failure
-
Feeling unsure or vulnerable at this point is very common
In gentle terms
Talking about discharge usually means “you are doing well enough not to need us right now” — not “you never needed us” and not “you’re on your own”.
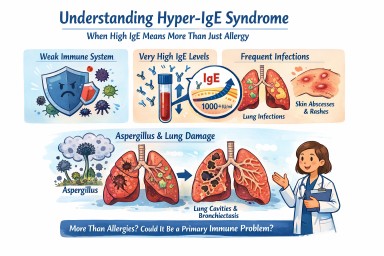
Hyper-IgE syndrome
A patient-friendly guide (and why it matters if you have aspergillosis)
It is not the same as having lots of allergies, even though it can look very similar at first.
What is IgE, and why does it matter?
IgE is usually involved in allergies and asthma.
In Hyper-IgE syndrome:
-
IgE levels are extremely high (often many thousands)
-
But the immune system is unbalanced
-
This makes infections—especially in the lungs and skin—harder to control
So IgE is high, but protection is weak.
How might Hyper-IgE syndrome affect everyday life?
Not everyone has the same symptoms, but common features include:
Lung and chest problems
-
Repeated chest infections (often from a young age)
-
Ongoing cough, breathlessness and mucus
-
Lung damage such as bronchiectasis
-
Lung cavities that can later become infected by moulds such as Aspergillus
Skin and infection problems
-
Long-standing eczema or very sensitive skin
-
Recurrent skin infections or boils
-
Infections that keep coming back or take a long time to clear
Other clues (in some people)
-
Frequent infections in childhood
-
Bone or joint problems
-
Dental issues (for example baby teeth not falling out on time)
Why is this important for people with aspergillosis?
For many people, Aspergillus causes allergy or irritation.
In Hyper-IgE syndrome:
-
The immune system struggles to control moulds
-
Aspergillus can behave more like a true infection, not just an allergy
-
Lung damage can happen more easily and progress faster
This means doctors may need to:
-
Monitor lungs more closely
-
Treat fungal disease earlier and for longer
-
Be cautious with repeated or long-term steroid use
Specialist centres such as the National Aspergillosis Centre are often involved when aspergillosis and immune problems overlap.
Isn’t this just severe allergy or ABPA?
Hyper-IgE syndrome can look similar to:
-
Severe allergic asthma
-
Allergic Bronchopulmonary Aspergillosis (ABPA)
The key difference is that in Hyper-IgE syndrome:
-
The immune system itself is faulty
-
High IgE is part of a wider immune problem
-
Treating allergy alone may not be enough
Some people are treated for asthma or ABPA for years before this possibility is considered.
How is Hyper-IgE syndrome treated?
There is no single cure, but good treatment can make a big difference. The aim is to prevent infections, protect the lungs, and reduce symptoms.
1. Preventing infections (most important)
Because the immune system does not fight germs normally:
-
Some people take regular low-dose antibiotics
-
Others use antibiotics early and promptly when infections start
For people with aspergillosis:
-
Antifungal medicines may be needed
-
Monitoring is usually closer and longer-term
2. Protecting the lungs
Many people develop bronchiectasis or lung damage, so care often includes:
-
Airway clearance physiotherapy
-
Saline nebulisers to help clear mucus
-
Regular sputum tests
-
Early treatment of flare-ups
The goal is to stop the cycle of:
infection → inflammation → permanent lung damage
3. Managing inflammation and allergy (carefully)
People may also have asthma-like symptoms, eczema and multiple allergies.
-
Steroids can help symptoms, but long-term or frequent use can increase infection risk
-
Doctors usually try to keep steroid doses as low as possible
Biologic treatments (such as anti-IgE medicines):
-
May help some people
-
Do not fix the immune problem
-
Are considered on an individual basis, usually in specialist centres
4. Skin care
-
Regular moisturising
-
Prompt treatment of infected eczema
-
Good skin care helps reduce infection risk
How is Hyper-IgE syndrome diagnosed?
Diagnosis usually involves:
-
A detailed review of your medical history (often including childhood infections)
-
Blood tests of immune function
-
Referral to an immunology specialist
-
Sometimes genetic testing
Does having high IgE mean I definitely have this?
No.
Hyper-IgE syndrome is rare.
But it may be worth asking about if:
-
Your IgE has always been extremely high
-
You’ve had repeated infections for many years
-
You have bronchiectasis without a clear cause
-
Aspergillosis seems unusually persistent or severe
-
Standard asthma or allergy treatments don’t fully explain your symptoms
Key message
Very high IgE does not always mean “just allergy.”
In a small number of people, it reflects a deeper immune problem that changes how aspergillosis behaves and how it should be treated.
If your illness doesn’t quite fit the usual labels, it is reasonable to ask whether an immunology review would help.
January–February 2026 Aspergillosis Papers (week 3)
Grouped by relevance and impact
🟥 HIGH IMPACT / PRACTICE-RELEVANT
(Most important for patients, clinicians, and services)
1. Chronic Pulmonary Aspergillosis (CPA): outcomes and mortality
Clinical Features and Mortality of Chronic Pulmonary Aspergillosis in Brazil
Open Forum Infectious Diseases, Jan 2026
Why this is important
-
Large multicentre cohort
-
Real-world data from TB-endemic, resource-limited settings
-
Directly relevant to global CPA burden, including post-TB disease
Key messages
-
CPA carries substantial long-term mortality
-
Tuberculosis is a major driver of CPA worldwide
-
Delayed diagnosis and limited antifungal access worsen outcomes
➡ This is one of the most important papers in the list for public health, service planning, and advocacy.
2. Invasive Aspergillosis in Intensive Care (including COVID-19)
Clinical spectrum of ICU-acquired invasive pulmonary aspergillosis according to SARS-CoV-2 infection
Eur J Clin Microbiol Infect Dis, Jan 2026
Why this is important
-
Large prospective multicentre ICU cohort
-
Builds on lessons from COVID-19 Associated Pulmonary Aspergillosis (CAPA)
Key messages
-
ICU-acquired aspergillosis remains common and deadly
-
COVID-19 patients are typically older and more severely ill
-
Early fungal testing in ICU is critical
➡ High relevance for intensivists, respiratory teams, and hospital policy.
3. Drug interactions in invasive aspergillosis
Concurrent administration of triazoles with chemotherapeutic and/or immunosuppressant agents
Mycopathologia, Jan 2026
Why this is important
-
Addresses real-world prescribing risk
-
Highly relevant to cancer, transplant, and haematology patients
Key messages
-
Triazole antifungals cause clinically dangerous drug–drug interactions
-
Requires specialist pharmacy oversight and monitoring
-
Not theoretical – directly affects patient safety
➡ High importance for clinicians and pharmacists, less so for patients directly, but critical for safe care.
🟧 MODERATE IMPACT / CLINICALLY INFORMATIVE
(Important, but narrower scope or smaller evidence base)
4. Aspergillosis beyond the “immunocompromised”
Pulmonary fungal infections in the immunocompetent host
Chest, Jan 2026 – Review
Why this matters
-
Challenges outdated assumptions
-
Useful for GPs and general physicians
Key messages
-
Serious fungal lung disease can occur without classic immune suppression
-
Chronic lung disease, viral infection, or exposure can be sufficient
-
Supports earlier fungal consideration when antibiotics fail
➡ Good educational review, especially for non-specialists.
5. Aspergillus species diversity and resistance
Beyond Fumigatus: a molecular portrait of clinical Aspergillus diversity
Antimicrobial Agents and Chemotherapy, Jan 2026
Why this matters
-
Advances understanding of non-fumigatus Aspergillus
-
Relevant to antifungal resistance
Key messages
-
Aspergillosis is caused by multiple species
-
Species identification may influence treatment success
-
Supports move toward precision mycology
➡ Important scientifically, indirect impact for patients (for now).
6. Minimally invasive treatment of aspergilloma
Minimally invasive management of a centrally located pulmonary aspergilloma
MMCTS, Jan 2026
Why this matters
-
Demonstrates evolving surgical approaches
-
Relevant to selected patients only
Key messages
-
Less invasive procedures may reduce surgical risk
-
Careful patient selection is crucial
➡ Clinically interesting, but case-based and niche.
🟨 LOW IMPACT / EARLY-STAGE / NICHE
(Useful context or future potential, limited immediate impact)
7. ABPA immunology and diagnostics (early-stage science)
Pathogen-specific IgE-reactive cytosolic allergenic epitopes of Aspergillus fumigatus
Ann Clin Microbiol Antimicrob, Jan 2026
Why this matters
-
Laboratory-based discovery research
Key messages
-
May improve future ABPA diagnostics
-
Potential foundation for targeted immunotherapy
➡ Promising but not practice-changing yet.
8. Voriconazole neurotoxicity (single case)
Voriconazole-associated peripheral polyneuropathy: A case report
Arch Argent Pediatr, Feb 2026
Why this matters
-
Highlights a rare but serious adverse effect
Key messages
-
Neurological symptoms on antifungals should not be ignored
-
Reinforces importance of monitoring during long-term therapy
➡ Low evidence level, but high awareness value.
9. Invasive aspergillosis in complex transplant oncology case
An Unforeseen Diagnosis After Liver Transplantation for Acute Liver Failure
Case Reports in Hepatology, Jan 2026
Why this matters
-
Illustrates diagnostic complexity in extreme immunosuppression
Key messages
-
Invasive aspergillosis can be rapidly fatal
-
Symptoms may be masked by other conditions
➡ Educational case, not generalisable.
10. Food enzyme safety (non-clinical)
Safety evaluation of the food enzyme aspergillopepsin I
EFSA Journal, Jan 2026
Why this matters
-
Addresses public concern rather than clinical disease
Key messages
-
Aspergillus-derived food enzymes are safe when regulated
-
Dietary exposure ≠ inhaled fungal spores
➡ Reassuring, but peripheral to aspergillosis care.
🔑 Overall “Most Important” Papers (Quick List)
Top tier
-
CPA outcomes and mortality (Brazil cohort)
-
ICU / COVID-19 associated invasive aspergillosis
-
Triazole drug–drug interactions
Second tier
4. Fungal infection in immunocompetent hosts
5. Aspergillus species diversity & resistance
January–February 2026 Aspergillosis Papers – Source Links
🟥 High-impact / practice-relevant
-
Clinical Features and Mortality of Chronic Pulmonary Aspergillosis in Brazil: a Multicenter Cohort Study
de Oliveira VF et al., Open Forum Infectious Diseases, Jan 2026
🔗 https://pubmed.ncbi.nlm.nih.gov/41536616/ -
Clinical spectrum of ICU-acquired invasive pulmonary aspergillosis according to SARS-CoV-2 infection: a multicenter prospective cohort study
Reizine F et al., European Journal of Clinical Microbiology & Infectious Diseases, Jan 2026
🔗 https://pubmed.ncbi.nlm.nih.gov/41526761/ -
Concurrent Administration of Triazoles with Chemotherapeutic and/or Immunosuppressant Agents Known to Have Moderate-to-Severe Drug-Drug Interactions in Patients with Hematologic Malignancies Hospitalized for Invasive Aspergillosis
Walsh TJ et al., Mycopathologia, Jan 2026
🔗 https://pubmed.ncbi.nlm.nih.gov/41528615/
🟧 Moderate-impact / clinically informative
-
Pulmonary fungal infections in the immunocompetent host (Review)
Lieu A et al., Chest, Jan 2026
🔗 https://pubmed.ncbi.nlm.nih.gov/41544957/ -
Beyond Fumigatus: a molecular portrait of clinical Aspergillus diversity, pathogenicity, and antifungal resistance
Aneke CI et al., Antimicrobial Agents and Chemotherapy, Jan 2026
🔗 https://pubmed.ncbi.nlm.nih.gov/41528247/ -
Minimally invasive management of a centrally located pulmonary aspergilloma in an adolescent patient
Mikilps-Mikgelbs R et al., Multimedia Manual of Cardiothoracic Surgery, Jan 2026
🔗 https://pubmed.ncbi.nlm.nih.gov/41537646/
🟨 Lower-impact / niche / early-stage
-
Pathogen-specific IgE-reactive cytosolic allergenic epitopes of Aspergillus fumigatus for immunodiagnostic and immunotherapeutic applications against allergic aspergillosis
Koundal P et al., Annals of Clinical Microbiology and Antimicrobials, Jan 2026
🔗 https://pubmed.ncbi.nlm.nih.gov/41540426/ -
Voriconazole-associated peripheral polyneuropathy: A case report
González BJ et al., Archivos Argentinos de Pediatría, Feb 2026
🔗 https://pubmed.ncbi.nlm.nih.gov/40728252/ -
An Unforeseen Diagnosis After Liver Transplantation for Acute Liver Failure: Extranodal NK/T-Cell Lymphoma (includes invasive aspergillosis)
Soares GL et al., Case Reports in Hepatology, Jan 2026
🔗 https://pubmed.ncbi.nlm.nih.gov/41542139/ -
Safety evaluation of the food enzyme aspergillopepsin I from the genetically modified Trichoderma reesei strain DP-Nzq40
EFSA Panel on Food Enzymes, EFSA Journal, Jan 2026
🔗 https://pubmed.ncbi.nlm.nih.gov/41531469/
What’s New in Aspergillosis Clinical Trials (Last ~4 Months)
An overview for patients and non-specialist readers — 19 January 2026
Over the past four months, research into aspergillosis — including chronic, allergic, and invasive forms — has continued across a range of clinical trials. These studies include treatments, diagnostics, and better ways to understand who gets sick and how best to manage it.
Below is a summary of the most relevant trials now active, recruiting, or updated recently. Whenever possible, we link to the official ClinicalTrials.gov record so you can see the details, eligibility criteria, locations, and contact information.
📋 Clinical Trials of Interest
1. Phase III Olorofim Trial for Invasive Aspergillosis
Study title: Olorofim Aspergillus Infection Study
Condition: Invasive aspergillosis (IA)
What it’s testing: A new antifungal drug called olorofim compared with liposomal amphotericin B followed by standard care.
Status: Active — not currently recruiting new patients but ongoing through 2026.
Official record: Olorofim Aspergillus Infection Study on ClinicalTrials.gov
Last updated: January 4, 2026
Why this matters: Olorofim is a completely new class of antifungal designed for patients whose infection is difficult to treat with standard drugs. It may offer an alternative for those with drug-resistant or treatment-intolerant infections.
2. Rezafungin in Chronic Pulmonary Aspergillosis (CPA)
Study title: Rezafungin for Treatment of Chronic Pulmonary Aspergillosis
Condition: Chronic pulmonary aspergillosis
What it’s testing: A long-acting echinocandin antifungal (rezafungin) that might reduce dosing frequency.
Status: Recruiting / active
Official record: Rezafungin CPA Trial on ClinicalTrials.gov
Why this matters: Current CPA treatments can require daily medication and prolonged therapy. Rezafungin’s once-weekly dosing could help reduce burden and hospital visits.
3. Combination Trial: Ibrexafungerp + Voriconazole (SCYNERGIA)
Study title: Evaluate Safety and Efficacy of Ibrexafungerp With Voriconazole in Invasive Pulmonary Aspergillosis
Condition: Invasive pulmonary aspergillosis
What it’s testing: Whether combining two antifungals works better than standard therapy alone.
Status: Active (ongoing)
Official record: SCYNERGIA Combination Trial on ClinicalTrials.gov
Why this matters: Some patients don’t respond well to single-agent treatment. Combination therapy may help in severe cases, especially where resistance is a concern.
4. PCR Diagnostic Study for Aspergillus fumigatus
Study title: PCR for Aspergillus Fumigatus in Blood and Bronchoalveolar Lavage Fluid
Condition: Aspergillosis (diagnostic focus)
What it’s testing: A blood and lung fluid PCR test to improve early detection of aspergillosis.
Status: Recruiting
Official record: PCR Aspergillus fumigatus Diagnostic Trial on ClinicalTrials.gov
First posted: 2 January 2026
Why this matters: Early diagnosis increases the chance of successful treatment. A reliable PCR test could allow clinicians to start antifungal therapy sooner.
🔎 What Else Is Ongoing?
There are other studies that include aspergillosis patients or Aspergillus exposure as part of broader research, such as:
-
All-of-Us Research Program fungal infection analysis — large observational work looking at fungal disease patterns in hundreds of thousands of people in the U.S., including aspergillosis. (Not a clinical trial per se but relevant to understanding how aspergillosis affects populations.)
-
Historic or related trials — e.g., older isavuconazole comparisons (e.g., NCT00412893) exist but are not newly updated.
🧠 What This Means for Patients
-
New antifungal drugs like olorofim and rezafungin are being tested in late-stage studies — these could expand treatment options in the future.
-
Combination therapies (e.g., ibrexafungerp + voriconazole) are being assessed to tackle difficult or resistant infections.
-
Improved diagnostics (e.g., PCR tests for Aspergillus fumigatus) are now being studied to help clinicians diagnose infections earlier and more accurately.
-
Not all trials are about treatment — some focus on better ways to detect infection or understand disease patterns, which are important for prevention and clinical practice.
🗓 How to Use These Links
Clicking a trial link takes you to the official ClinicalTrials.gov page, where you can often see:
-
Who can participate
-
Locations and contact information
-
Detailed eligibility criteria
-
Sponsor and trial timelines
If you have questions about joining a trial or how it applies to you specifically, always discuss this with your healthcare team.
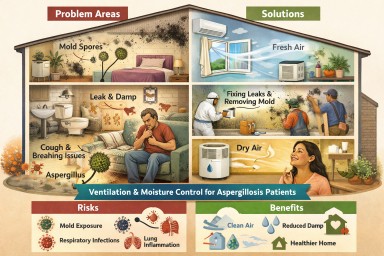
Indoor Damp, Ventilation & Aspergillosis
What a Major UK Evidence Review Means for Patients and Professionals
This large UK Health and Safety Executive (HSE) review examined whether microorganisms inside buildings (homes, offices, workplaces) can harm health — and what actually helps reduce risk.
Although it does not focus on a single disease, its findings are highly relevant to people living with aspergillosis, asthma, bronchiectasis, and other chronic lung conditions, as well as the professionals who support them.
The short answer (for everyone)
Yes — indoor environments can significantly affect lung health.
And ventilation and moisture control are central to reducing risk, especially for people vulnerable to fungal exposure.
What the review confirms (in plain language)
1. Indoor fungi are common — and not harmless
High confidence evidence
Many buildings contain airborne and surface fungi, especially when dampness is present.
The fungi most often found indoors include:
-
Aspergillus
-
Penicillium
-
Cladosporium
-
Alternaria
For aspergillosis patients, this matters because:
-
Aspergillus is not just an “outdoor mould”
-
Ongoing exposure can worsen symptoms, trigger inflammation, or complicate recovery
-
Even low levels may be problematic for sensitised or immunocompromised people
2. Dampness is a major driver of fungal exposure
High confidence
Damp buildings — whether due to leaks, condensation, or poor airflow — consistently show:
-
Higher mould growth
-
More fungal spores in the air
-
Stronger links to respiratory symptoms
Important point for patients:
You do not need to see black mould for damp to be affecting your lungs.
Mould smell (“musty odour”) is one of the strongest warning signs.
3. Ventilation is the most important protective factor
High confidence
Ventilation:
-
Dilutes fungal spores, bacteria, and viruses
-
Reduces moisture build-up
-
Lowers exposure for occupants
This applies to:
-
Homes
-
Flats
-
Offices
-
Other non-industrial indoor spaces
⚠️ The review highlights a key modern problem:
Energy-efficient, airtight buildings can unintentionally trap damp and fungi if ventilation is inadequate.
For aspergillosis patients, this means:
-
A “warm” home is not always a “healthy” home
-
Reduced airflow can increase fungal exposure even without visible mould
4. Indoor air also spreads infections
High confidence
Respiratory viruses (e.g. influenza, COVID-19) spread mainly through indoor air, especially when ventilation is poor.
This is relevant for aspergillosis patients because:
-
Viral infections can destabilise lung disease
-
Recovery may be slower
-
Secondary infections are more likely
Ventilation therefore protects against both fungal and viral risks.
5. Surfaces matter too — but air matters more
Medium–high confidence
-
Fungal material and microbes accumulate in dust, carpets, soft furnishings, and damp surfaces
-
Toilets and bathrooms can generate contaminated aerosols
-
Good hygiene helps, but cannot compensate for poor ventilation
For patients:
Cleaning alone will not solve a damp or ventilation problem.
What actually helps (evidence-based)
Strongest evidence
✔️ Adequate ventilation (natural or mechanical)
✔️ Fixing leaks and moisture sources
✔️ Removing mould-damaged materials
✔️ Preventing condensation on cold surfaces
Moderate evidence
✔️ HEPA air filtration (helpful but not a substitute for ventilation)
✔️ UV air disinfection (context-specific)
✔️ Touch-free fittings in shared buildings
⚠️ No single measure works on its own — combined approaches are needed.
Why this matters specifically for aspergillosis patients
This review strongly supports what many patients already experience:
-
Symptoms may persist despite treatment if exposure continues
-
Indoor environments can drive inflammation and relapse
-
“Just take your medication” is not enough if housing conditions are harmful
Importantly, the review recognises that:
-
Health effects vary by individual vulnerability
-
Those with asthma, bronchiectasis, aspergillosis, or immune suppression are more sensitive
-
There are no universally safe mould levels for everyone
What non-specialists should take from this
For GPs and clinicians
-
Damp and poor ventilation are legitimate medical risk factors
-
Persistent respiratory symptoms may be environment-driven
-
Asking about housing conditions is clinically relevant
For housing, environmental health & social care
-
Mould and damp are health hazards, not cosmetic defects
-
Ventilation failures can directly affect chronic disease
-
Energy efficiency must be balanced with respiratory health
For patients and carers
-
You are not “overreacting” if your home affects your breathing
-
Ventilation and moisture control are part of disease management
-
Evidence supports advocating for safer living conditions
Bottom line
This major UK review confirms that indoor dampness and poor ventilation increase exposure to fungi — including Aspergillus — and worsen respiratory health.
For people living with aspergillosis, building conditions are not secondary issues: they are part of the disease environment.