How to Ask Fewer, Better Questions in Appointments
Focusing on what matters most to you—without feeling you’re wasting time
Many patients and carers worry about “asking too much” in clinic. Appointments are short, clinicians are busy, and you may already have a long list of questions in your head. The aim isn’t to stop asking questions—it’s to ask the right ones, at the right time, in the right way.
Here are practical strategies that help you stay focused, feel heard, and make the most of limited time.
1. Decide your Top 3 priorities before you go
Before the appointment, write down everything you’re thinking about. Then circle just three things that matter most right now.
Good priorities are usually:
-
A symptom that is new, worsening, or frightening
-
A treatment issue that affects daily life (side-effects, adherence, cost, function)
-
A decision you need to make soon
If it doesn’t change what happens in the next few weeks, it may not need airtime today.
If you remember only one thing: appointments are for decisions, not encyclopaedias.
2. Separate “need to know” from “nice to know”
It’s easy to mix curiosity with urgency.
Need to know (ask now):
-
Is this symptom important?
-
Is this treatment still right for me?
-
What should I do if X happens?
-
Are we monitoring the right things?
Nice to know (park for later):
-
Mechanisms, pathways, emerging research
-
Rare side-effects without symptoms
-
“What if” scenarios far in the future
Keep a “parking list” for later reading or discussion.
3. Frame questions around impact, not theory
Clinicians work best when questions are grounded in real life.
Instead of:
-
“I read a paper saying X might affect Y…”
Try:
-
“I’m noticing X in daily life—does that change what we do?”
-
“Is this symptom something you’d want to investigate?”
This signals relevance and helps clinicians triage quickly.
4. Ask one question at a time
Long, multi-part questions feel overwhelming and are easy to partially answer.
Break them down:
-
First: Is this important?
-
Then (if yes): What do we do about it?
-
Then (if needed): What should I watch for?
You’ll often find later questions become unnecessary once the first is answered.
5. Use the “Is this something we should…” test
This single phrase keeps questions concise and respectful of time:
-
“Is this something we should investigate?”
-
“Is this something that changes treatment?”
-
“Is this something I should worry about?”
A clear yes/no (or not yet) is often all you need.
6. Accept that not everything fits in one appointment
It’s okay—and normal—to say:
-
“I know we may not have time today—what should I prioritise?”
-
“Which of these matters most from your point of view?”
This shows partnership, not passivity.
If something needs more time, ask how best to handle it:
-
Another appointment
-
A nurse specialist
-
Written advice
-
Monitoring and review later
7. Bring written notes (but don’t read them all out)
A short list helps you stay focused under pressure.
Tip:
-
Highlight your top 3
-
Tick them off as they’re addressed
-
If time runs out, you still covered what mattered most
8. For carers: ask on behalf, not over
Carers often worry about dominating the conversation.
Helpful approaches:
-
Ask the patient first: “What do you most want answered today?”
-
Step in only if something important is being missed
-
Offer to follow up questions outside the appointment if possible
9. Reassure yourself: clinicians don’t expect perfection
You are not expected to:
-
Understand everything
-
Ask the “right” questions every time
-
Cover your entire condition in one visit
Good clinicians prefer:
a focused conversation
over
a rushed, overloaded one
10. A simple closing question that saves time
If time is tight, end with:
-
“Is there anything you think I should have asked but didn’t?”
This often surfaces the most important point of all.
The takeaway
You are not wasting time by asking questions—you’re wasting time by asking too many unfocused ones.
Clarity, prioritisation, and relevance help everyone:
-
You leave with answers that matter
-
Clinicians can make better decisions
-
Anxiety is reduced, not fuelled
Learning About Aspergillosis (and Related Treatments)
How to stay curious, informed, and safe — without overload
Many people living with aspergillosis, or caring for someone who is, become highly motivated learners. You may read scientific papers, books, online articles, social media posts, AI summaries, and news stories about antifungal treatments, steroids, biologics, side-effects, immunity, mould exposure, diet, exercise, and wellbeing.
That curiosity is a strength. It helps you ask better questions, notice changes early, and feel more involved in your care.
At the same time, not all information is reliable, relevant, or helpful, and even good information can become harmful if it is over-interpreted or taken out of context. This article is about finding the balance: learning with confidence, without increasing anxiety or risk.
Why learning helps — and why it can sometimes backfire
The positives
-
Empowerment: Understanding your condition improves confidence.
-
Better conversations: Appointments are more productive when you share a common language.
-
Early awareness: You may recognise symptoms or side-effects sooner.
-
Reassurance: Knowledge can reduce uncertainty and fear.
The risks
-
Over-interpretation: A single paper or post can feel more important than it is.
-
Variable quality: Some research is weak, outdated, biased, or misapplied.
-
Loss of context: Lab studies or rare case reports may not apply to you.
-
Rising anxiety: Constant searching can amplify worry rather than reduce it.
-
Information overload: Too much input can make decisions harder, not easier.
A healthier approach to learning
1. Think in weight of evidence, not single findings
One article, story, or AI answer almost never changes medical care on its own.
When you read something new, ask:
-
Is this supported by more than one study?
-
Does it appear in guidelines or specialist practice?
-
Is it discussed cautiously, or presented as a breakthrough?
A useful rule of thumb:
The more dramatic the claim, the stronger the evidence needs to be.
2. Separate biological possibility from clinical reality
Many things are biologically plausible — immune pathways, hormones, inflammation, the microbiome — but that doesn’t mean they are proven or clinically relevant.
Helpful questions include:
-
Was this studied in people, or only in the lab?
-
Were the patients similar to me?
-
Did it improve symptoms or outcomes, not just blood tests?
Choosing trusted health information: practical guidance
Learning safely isn’t about reading less. It’s about choosing better sources and knowing how much weight to give them.
3. Start with sources that anchor practice
Your most reliable foundations are sources that:
-
Reflect clinical consensus, not speculation
-
Are written or reviewed by specialist teams
-
Change slowly because they are evidence-based
Examples include:
-
Specialist centre or hospital websites
-
National or international guidelines
-
Established patient organisations linked to clinical services
Examples:
-
-
NHS website
A good starting point for clear, balanced information on symptoms, tests, treatments, and general health advice.
Useful for understanding what is considered standard care in the UK. -
British National Formulary (BNF)
The main UK reference for medicines.
Particularly helpful for:-
Medication side-effects
-
Drug interactions (including antifungals, steroids, and inhalers)
-
Practical prescribing information
Side-effects are listed cautiously, so not everything applies to every person.
-
-
aspergillosis.org
A specialist resource focused specifically on aspergillosis, written for patients, carers, and professionals.
Helpful for understanding different forms of aspergillosis, investigations, treatments, and living with the condition. -
European Lung Foundation – Aspergillosis resources
Patient-focused information developed with respiratory specialists and patient representatives across Europe.
Particularly useful for:-
Plain-language explanations
-
Patient priorities and lived experience
-
Shared decision-making and questions to ask in clinic
-
-
Asthma + Lung UK (BLF)
A trusted source for asthma and other lung diseases.
Helpful for inhaler use, breathlessness, flare-ups, lifestyle advice, and living well with chronic lung conditions. - Aspergillosis Trust
This website was created by patients who suffer from Aspergillosis. Please navigate around the website to read more about this disease, also the impact it has upon patients and their carers.
-
These sources may feel less exciting — but they set the safe boundaries of what is known.
4. Learn to spot interpretation versus evidence
Two people can read the same paper and draw very different conclusions.
Ask yourself:
-
Is this source presenting evidence, or interpreting it strongly?
-
Are limitations and uncertainty acknowledged?
-
Is the language careful or absolute?
Trusted sources often say:
“Evidence suggests…” or “We don’t yet know…”
Less reliable ones often say:
“This proves…” or “This explains everything.”
5. Use a simple credibility checklist
You don’t need to be a scientist to judge quality.
When reading anything, consider:
Who wrote it?
Clinical specialists, recognised organisations, or anonymous individuals?
Why was it written?
To inform and support — or to sell, persuade, or provoke?
What evidence is used?
Multiple studies and guidelines — or a single paper or personal story?
What tone is used?
Balanced and cautious — or dramatic and fear-based?
Several warning signs together should lower confidence.
6. Be cautious with “hidden” or “overlooked” explanations
Phrases that should trigger caution include:
-
“Doctors don’t tell you this…”
-
“The hidden cause…”
-
“The real reason…”
-
“One simple explanation…”
Conditions like aspergillosis are complex. Simple, universal explanations are rarely accurate.
7. Understand where research sits on the evidence ladder
Not all research carries the same weight.
Very roughly:
-
Clinical guidelines and consensus statements
-
Large clinical trials and systematic reviews
-
Observational studies
-
Case reports
-
Laboratory or animal studies
-
Opinions and anecdotes
Lower down the ladder does not mean “worthless” — but it does mean less certain and less likely to change care on its own.
8. Treat patient stories and forums as experience, not prediction
Patient experiences are invaluable for:
-
Feeling less alone
-
Understanding day-to-day challenges
-
Sharing coping strategies
They are not reliable predictors of:
-
What will happen to you
-
How common a problem is
-
Whether a treatment will help or harm you
A helpful distinction:
Stories help you feel understood. Evidence helps guide decisions.
9. Use AI tools wisely
AI can be excellent for:
-
Explaining terminology
-
Summarising broad topics
-
Helping you generate questions
AI cannot:
-
Replace specialist judgement
-
Fully understand your medical history
-
Balance risk in the way clinicians do
Treat AI as:
“A map to the topic,”
not
“An answer about me.”
10. Limit your sources — and give yourself permission to stop
Many people feel calmer once they:
-
Choose two to four trusted sources
-
Revisit those instead of endlessly searching
-
Accept that not every new paper needs action
Stopping is not giving up — it is protecting your wellbeing.
Bringing what you’ve learned into clinic
A good sign you’ve chosen reliable information:
-
You feel comfortable sharing it with clinicians
-
It leads to discussion, not confusion
-
It helps prioritise decisions
You might say:
-
“I’ve been reading from a specialist source — how relevant is this to me?”
-
“This helped me understand X, but I’m not sure how much weight to give it.”
When to pause or rebalance your learning
Consider stepping back if:
-
Searching increases anxiety every time
-
You feel pressure to solve everything yourself
-
Conflicting information leaves you stuck
-
Illness becomes the only thing you think about
Taking breaks from research is not disengagement — it is self-care.
The key message
Learning is a powerful tool. Used well, it supports confidence, partnership, and resilience. Used without guardrails, it can undermine peace of mind.
Aim for:
-
Curiosity with caution
-
Knowledge with context
-
Questions with balance
You don’t need to know everything.
You need to know what helps you live well and safely.
This article pairs with:
“Making the Most of Appointments: Asking Fewer, Better Questions” — a practical guide to deciding what to raise in clinic and how to use limited time effectively.
Recent Aspergillosis Research – January 2026 (week 2)
A curated update with direct links to the evidence
Allergic Bronchopulmonary Aspergillosis (ABPA) and Aspergillus Airway Disease
Several recent papers continue to refine how ABPA is diagnosed and managed, particularly outside classic asthma and cystic fibrosis settings.
A practical treatment overview for ABPA in cystic fibrosis is provided by Thimmesch et al., focusing on steroid strategies and antifungal use
👉 https://pubmed.ncbi.nlm.nih.gov/41537324/
A major narrative review of ABPA and Aspergillus-related airway disease in bronchiectasis reframes ABPA as part of a wider spectrum of Aspergillus-driven lung disease, with implications for earlier recognition
👉 https://pubmed.ncbi.nlm.nih.gov/41522133/
Diagnostic uncertainty remains a major challenge. A comparative study of IgG/IgE ELISA versus serum precipitins highlights strengths and limitations of commonly used tests
👉 https://pubmed.ncbi.nlm.nih.gov/41514182/
Treatment optimisation is addressed in a retrospective cohort study examining glucocorticoid dose, duration, and antifungal combinations in ABPA, showing that regimen choice meaningfully affects outcomes
👉 https://pubmed.ncbi.nlm.nih.gov/41492771/
ABPA is increasingly recognised beyond asthma and cystic fibrosis. A short but influential paper proposes Aspergillus sensitisation and ABPA in COPD as a “treatable trait”
👉 https://pubmed.ncbi.nlm.nih.gov/41520264/
Rare but clinically important overlap is illustrated by a case report describing invasive pulmonary aspergillosis overlapping with ABPA
👉 https://pubmed.ncbi.nlm.nih.gov/41496036/
Chronic Pulmonary Aspergillosis (CPA) and Aspergilloma
A large multicentre cohort study from Brazil provides valuable data on CPA in high tuberculosis-burden settings, documenting delayed diagnosis and significant mortality
👉 https://pubmed.ncbi.nlm.nih.gov/41536616/
Surgical innovation is highlighted in a report describing minimally invasive management of a centrally located pulmonary aspergilloma in an adolescent, showing evolving interventional approaches
👉 https://pubmed.ncbi.nlm.nih.gov/41537646/
CPA caused by non-fumigatus species is increasingly recognised, including a case of Aspergillus candidus infection in a lung cancer patient with heavy smoking history
👉 https://pubmed.ncbi.nlm.nih.gov/41495698/
Invasive Pulmonary Aspergillosis (IPA): Expanding Risk Groups
A comprehensive review describes how IPA is increasingly seen in non-neutropenic patients, challenging traditional risk models
👉 https://pubmed.ncbi.nlm.nih.gov/41515529/
ICU-acquired IPA is examined in a large multicentre cohort stratified by SARS-CoV-2 infection, demonstrating different clinical patterns in COVID-positive and COVID-negative patients
👉 https://pubmed.ncbi.nlm.nih.gov/41526761/
Longitudinal mycological data from ICU patients with influenza- and COVID-associated pulmonary aspergillosis (IAPA and CAPA) show how bronchoalveolar lavage galactomannan kinetics relate to outcome
👉 https://pubmed.ncbi.nlm.nih.gov/41508132/
IPA is not confined to traditional risk groups. A case report documents invasive aspergillosis in a child without identifiable risk factors, reinforcing the need for diagnostic vigilance
👉 https://pubmed.ncbi.nlm.nih.gov/41520716/
Rare manifestations continue to appear, including isolated renal aspergillosis after paediatric stem-cell transplantation
👉 https://pubmed.ncbi.nlm.nih.gov/41508322/
Diagnostics, Resistance, and Drug Safety
Concerns about antifungal resistance are reinforced by a clinical screening study demonstrating azole resistance in Aspergillus isolates
👉 https://pubmed.ncbi.nlm.nih.gov/41528781/
A molecular overview of clinical Aspergillus diversity beyond A. fumigatus highlights implications for pathogenicity and antifungal susceptibility
👉 https://pubmed.ncbi.nlm.nih.gov/41528247/
Drug–drug interactions are explored in depth in a study examining concurrent triazole use with chemotherapy and immunosuppressants in invasive aspergillosis, underlining the importance of monitoring
👉 https://pubmed.ncbi.nlm.nih.gov/41528615/
Unusual but serious adverse effects are illustrated by a case of voriconazole-induced hypoglycaemia in a non-diabetic patient
👉 https://pubmed.ncbi.nlm.nih.gov/41506798/
Cutaneous manifestations as early diagnostic clues are reviewed in a paper on skin signs of invasive fungal disease, including secondary cutaneous aspergillosis
👉 https://pubmed.ncbi.nlm.nih.gov/41505800/
New Therapies and Prevention Strategies
A nationwide French observational study reports real-world outcomes from compassionate use of olorofim in invasive mould infections, including refractory aspergillosis
👉 https://pubmed.ncbi.nlm.nih.gov/41531505/
Economic evidence is emerging alongside clinical data. A cost-utility analysis of preventing invasive aspergillosis in lung transplant recipients supports targeted prophylactic strategies
👉 https://pubmed.ncbi.nlm.nih.gov/41490563/
Fundamental and Translational Aspergillus Research
Several papers advance understanding of Aspergillus fumigatus virulence and regulation, including network-based gene regulation models
👉 https://pubmed.ncbi.nlm.nih.gov/41505094/
A detailed mechanistic study shows how CsdA–LaeB regulatory interactions influence virulence and secondary metabolite production
👉 https://pubmed.ncbi.nlm.nih.gov/41498629/
Vaccine-relevant work explores how cell-wall remodelling alters innate immune recognition in an experimental A. fumigatus strain
👉 https://pubmed.ncbi.nlm.nih.gov/41509434/
Population genomics continues to redefine Aspergillus diversity, including the Aspergillus flavus–oryzae complex, with relevance to both clinical and environmental exposure
👉 https://pubmed.ncbi.nlm.nih.gov/41522572/
One Health and Veterinary Aspergillosis
Aspergillosis remains a One-Health issue, with reports including fungal pericarditis due to Aspergillus fumigatus in dogs
👉 https://pubmed.ncbi.nlm.nih.gov/41521069/
and Aspergillus infections in endangered bird species, highlighting environmental and conservation links
👉 https://pubmed.ncbi.nlm.nih.gov/41497419/
Sinusitis in Patients with ABPA
When to suspect it, when to investigate, and when to refer
Why this matters
Patients with allergic bronchopulmonary aspergillosis (ABPA) are usually managed as having a lung disease. Diagnosis, monitoring, and treatment focus appropriately on the chest, immunology, and asthma control.
However, ABPA occurs within a single continuous airway, extending from the nose and sinuses to the lungs. Disease in the upper airway can coexist with, exacerbate, or complicate lower airway inflammation — yet sinus disease is not routinely assessed in ABPA care pathways.
This article outlines:
-
What is known about sinus disease in this context
-
Which symptoms should raise suspicion
-
When investigation or ENT referral should be considered
-
What GPs and non-specialists can reasonably do
The united airway: a brief reminder
The upper and lower airways share:
-
Type 2 (eosinophilic) inflammation
-
Immunoglobulin E–mediated immune responses
-
Common triggers, including allergens and fungi
Chronic rhinosinusitis is common in asthma and severe asthma, and treatment of sinus disease can improve lower airway outcomes in some patients.
ABPA sits within this same inflammatory spectrum, even though its management is lung-centred.
Sinus disease in ABPA: what is (and isn’t) known
What we know
-
Chronic rhinosinusitis is common in patients with asthma and severe asthma
-
Sinus disease may be symptomatic or relatively silent
-
ABPA guidelines do not mandate routine ENT review or sinus imaging
-
ENT involvement, therefore, varies widely between centres
What we do not know
-
Whether routine ENT assessment improves ABPA outcomes
-
Which ABPA patients benefit most from sinus intervention
-
The optimal timing for ENT referral in ABPA
As a result, clinical judgement remains central.
Symptoms that should prompt consideration of sinus disease
Sinusitis in ABPA patients does not always present with classic “blocked nose and facial pain”.
Key symptoms include:
Common but often overlooked
-
Persistent post-nasal drip
-
Foul, bitter, metallic, or “infected” taste in the mouth
-
Throat clearing, chronic cough
-
Thick or sticky mucus sensation
-
Symptoms are worse on waking or lying flat
More typical sinonasal features
-
Nasal blockage or congestion
-
Facial pressure or fullness
-
Reduced or altered sense of smell
-
Nasal crusting or discharge
Contextual clues
-
Poor durability of response to steroids or antifungals
-
Recurrent “flares” without clear chest triggers
-
Coexisting severe asthma or nasal polyps
-
Symptoms are worse in damp or mould-affected housing
A persistent foul taste in the mouth is a recognised symptom of chronic sinus disease, usually due to post-nasal drainage of inflamed secretions.
Damp homes and sinus disease
Living in damp or mould-affected environments is associated with:
-
Higher rates of chronic rhinosinusitis
-
Upper airway irritation and inflammation
-
Allergic sensitisation to fungal spores
In most cases, this results in inflammatory or allergic sinusitis, not invasive fungal infection.
Fungal involvement may act as an immune trigger, even when not labelled as “fungal sinusitis”.
Fungal sinusitis: rare vs under-recognised
It is important to distinguish between entities:
| Type | Frequency | Key point |
|---|---|---|
| Invasive fungal sinusitis | Rare | Usually immunocompromised; dramatic presentation |
| Fungal ball (mycetoma) | Uncommon | Usually obvious on CT |
| Allergic fungal rhinosinusitis | Likely under-recognised | Requires active suspicion |
Allergic fungal rhinosinusitis overlaps biologically with ABPA:
-
IgE-mediated
-
Eosinophilic inflammation
-
Thick allergic mucin
It is not routinely sought, so it may be under-diagnosed in at-risk groups.
What GPs and non-specialists can reasonably do
1. Take upper airway symptoms seriously
Especially in ABPA or severe asthma patients with:
-
Persistent post-nasal symptoms
-
Foul taste
-
Recurrent unexplained deterioration
2. Examine the nose and throat
-
Look for polyps, discharge, and crusting
-
Note mouth breathing or altered voice quality
-
Check dentition (to exclude dental causes)
3. Consider imaging when symptoms persist
-
CT sinuses (not plain X-ray) is the imaging of choice
-
Particularly appropriate if symptoms last >8–12 weeks or recur
4. Refer to ENT when:
-
Symptoms are persistent or progressive
-
CT shows significant sinus disease
-
There is a poor response to standard medical therapy
-
There is diagnostic uncertainty
Referral does not imply surgery — ENT input may be diagnostic or medical.
What this article is not saying
-
It does not suggest that all ABPA patients need an ENT referral
-
It does not claim that sinus treatment improves ABPA outcomes
-
It does not override existing guidelines
It does suggest that earlier consideration of the upper airway is reasonable in selected patients.
Key take-home points for clinicians
-
The airway functions as a single inflammatory system
-
Sinus disease may be subtle, under-reported, or atypical
-
A foul taste in the mouth is a meaningful symptom
-
Damp or mould exposure increases sinus disease risk
-
ENT referral is appropriate when symptoms persist or recur
-
Evidence gaps remain — but clinical vigilance is justified
In summary
ABPA is managed as a lung disease, but patients live with a whole airway.
Recognising when sinus disease may be contributing can help explain persistent symptoms and guide appropriate referral — without over-investigation or over-treatment.
ABPA and Work: What a Patient Poll Tells Us About Employment, Health, and Real-World Impact
An article for patients, GPs, and non-specialist healthcare professionals
Allergic bronchopulmonary aspergillosis (ABPA) is often discussed in terms of lung function, immunology, and imaging. Far less often do we talk about its impact on everyday life, particularly on a person’s ability to work.
A poll run within the National Aspergillosis Centre patient community asked a simple but powerful question:
Who is still able to work while living with ABPA – and who has had to stop or retire?
The responses provide an important insight into the functional and socioeconomic burden of ABPA.
Key findings from the poll (patient-reported)
-
Working full time: 17%
-
Working part time (days or hours): 18% combined
-
Not working: 30%
-
Retirement age: 21%
-
Retired early for health reasons: 12%
-
Currently on sick leave / full-time carer / pre-diagnosis: small but notable groups
Even allowing for the informal nature of a social media poll, the overall pattern is clear.
What this tells us
1. Sustained full-time work is uncommon in ABPA
Fewer than one in five respondents were able to work full time. Even among those still working, many described reduced hours, flexible arrangements, or fragile employment dependent on day-to-day health.
ABPA is often incompatible with predictable, high-demand working patterns.
2. ABPA frequently leads to work loss or early retirement
A substantial proportion of respondents were either:
-
No longer working at all, or
-
Retired earlier than planned specifically because of health
This is particularly striking given that ABPA often affects people during their working years and may coexist with asthma, bronchiectasis, or long-term steroid use.
3. “Retirement age” can hide health-forced exit
Some respondents selected “retirement age,” but accompanying comments revealed that many:
-
Left work earlier than expected
-
Changed careers or reduced responsibilities years before retirement
-
Worked through ill health until they no longer could
This matters when interpreting employment statistics: health-driven work loss may be underestimated.
4. Unpaid work and instability are often overlooked
The poll also highlighted:
-
People currently on prolonged sick leave
-
Full-time unpaid carers
-
Individuals still awaiting diagnosis but already struggling to work
These groups are frequently invisible in employment data, yet represent significant personal and societal impact.
Why ABPA affects the ability to work
For patients and non-specialists, it is important to understand that work difficulties in ABPA are not simply due to “asthma symptoms.”
Common contributors include:
-
Chronic breathlessness and cough
-
Severe fatigue and post-exertional exhaustion
-
Recurrent chest infections
-
Steroid side-effects (muscle weakness, bone disease, mood changes, diabetes risk)
-
Unpredictable flare-ups requiring rest, antibiotics, or hospital care
-
Cognitive and emotional burden of long-term illness
Together, these make consistent attendance, physical work, and high cognitive load difficult to sustain.
Implications for patients
-
Difficulty working is not a personal failure
-
Many others with ABPA face similar challenges
-
Adjustments, reduced hours, or stopping work altogether may be medically appropriate
-
Asking for support is reasonable and justified
Implications for GPs and non-specialist clinicians
-
Employment status should be considered a key outcome of disease control
-
Fit notes, occupational health input, and benefits documentation are part of holistic care
-
ABPA is a fluctuating condition – patients may cope for periods and then deteriorate
-
Statements such as “lung function is stable” do not always reflect real-world functioning
Understanding the work impact helps clinicians better support patients in consultations, reports, and advocacy.
Implications for systems and policy
This poll reinforces that ABPA carries a significant socioeconomic burden, including:
-
Reduced workforce participation
-
Early retirement
-
Increased reliance on health and social support systems
Any assessment of disability, employment capability, or long-term planning must take into account:
-
Variability over time
-
Treatment burden
-
Side-effects of necessary medications
In summary
This patient poll sends a consistent message:
ABPA commonly limits the ability to work, often leading to reduced hours, unstable employment, or early exit from the workforce.
For patients, this experience is shared and valid.
For clinicians, it is a reminder that ABPA is not just a radiological or immunological diagnosis, but a life-limiting condition with real-world consequences.
📢 Patient Speaker Opportunity – Breathe Clean Air Patient Conference 2026
The European Lung Foundation’s Breathe Clean Air Patient Conference is taking place online on 19 February 2026. This free event brings together patients, advocates, and experts to explore the impact of indoor air quality — including mould, moisture, and everyday environments — on lung health.
The organisers are looking for one patient speaker to share their lived experience in a 10-minute talk, either live or as a pre-recorded video. The theme is personal experience of mould, moisture or indoor air triggers affecting respiratory health — what it’s like day-to-day, how it’s impacted your life, and what advice you’d give others.
✨ This is a great chance to:
-
bring the patient voice to an international audience,
-
help raise awareness of how indoor air quality affects people with lung conditions,
-
and connect with others affected by similar issues.
The ELF team will provide support and a detailed brief ahead of the talk or video recording, so you won’t be doing it alone.
If you or someone you know might be interested, please let me know in the comments — happy to put you in touch with the organisers 🤍
Does when I eat cause fat gain if I have adrenal insufficiency?
Many people with adrenal insufficiency worry that eating at the “wrong time” — especially later in the day — will automatically cause weight gain or “steroid belly”.
This is understandable, but it’s important to separate myths from what actually happens in the body.
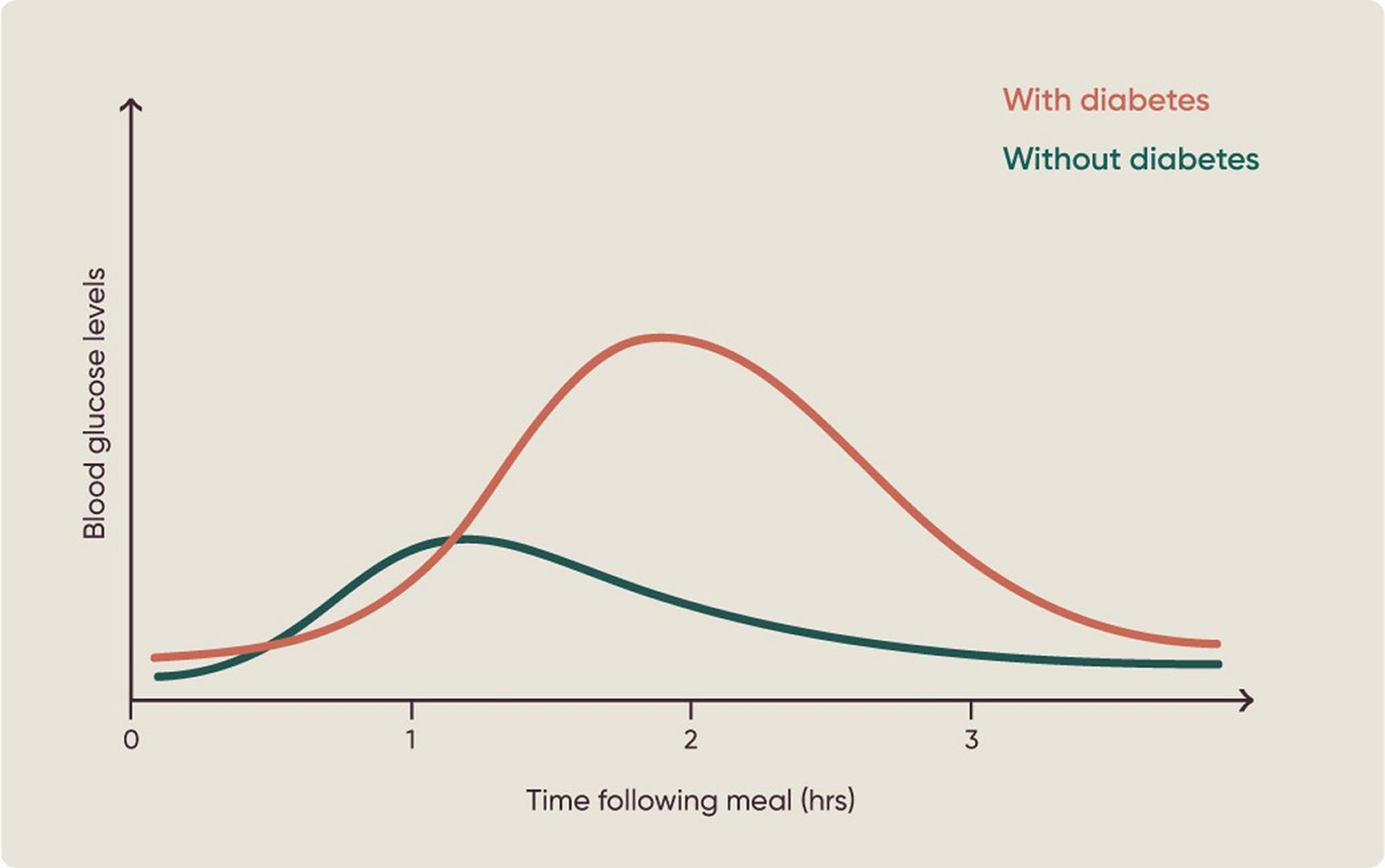

What doctors mean by “glucose response”
When clinicians or researchers talk about glucose response, they mean:
How your blood sugar rises and falls after eating
It does not mean that sugar is instantly being turned into fat.
A rise in blood glucose after eating is normal and happens in everyone.
Does eating later in the day automatically turn food into fat?
No.
Fat gain does not happen because of a single meal or snack — or because you ate at a particular time.
In most people:
-
Carbohydrates are first used for energy
-
Extra glucose is stored as glycogen in muscles and liver
-
Only repeated excess intake over time contributes to fat gain
Eating in the evening does not automatically cause fat storage.
Where insulin fits in (without the fear)
Eating raises blood glucose, which triggers insulin.
Insulin:
-
Helps move glucose into cells
-
Replenishes energy stores
-
Temporarily pauses fat burning
This pause is normal and reversible.
Insulin does not automatically create body fat.
Fat gain happens when:
-
Total calorie intake is consistently higher than needs
-
Steroid replacement is higher than required
-
This pattern continues over weeks or months
Why people with adrenal insufficiency feel confused about this
With adrenal insufficiency:
-
Cortisol replacement is taken in doses, not continuously
-
Symptoms, stress, poor sleep, or illness can affect appetite and energy
-
Some people are prone to low blood sugar, especially later in the day
Because of this:
-
Rigid food timing rules can make symptoms worse
-
Skipping meals or avoiding evening snacks can increase fatigue, dizziness, or night-time symptoms
A safer way to think about meal timing
Instead of strict rules, think in patterns:
-
Some people feel best with:
-
Larger meals earlier in the day
-
Lighter evenings
-
-
Others need:
-
A small evening snack
-
Protein or fat to keep blood sugar stable overnight
-
Both can be correct.
What matters most is:
-
How you feel
-
Whether your energy is stable
-
Whether sleep and symptoms improve
What usually matters more than timing
For people with adrenal insufficiency, weight changes are most often related to:
-
Total daily steroid dose
-
Repeated or prolonged stress dosing
-
Reduced activity due to illness or fatigue
-
Menopause, ageing, or other medical conditions
Food timing plays a much smaller role.
Key reassurance
If a food timing rule makes you feel worse, it is not the right rule for you.
-
A single glucose rise does not cause fat gain
-
Eating later does not automatically lead to weight gain
-
Safety, symptom control, and adequate steroid replacement come first
Please remember
Never change steroid dose or meal patterns intended to prevent hypoglycaemia without medical advice.
Underdosing steroids is far more dangerous than eating at the “wrong” time.
Take-home message
Focus on stability, nourishment, and feeling well — not fear of timing.
Hydrocortisone dosing in adrenal insufficiency
Why adrenal insufficiency can happen in people with aspergillosis
Many people with aspergillosis, particularly those with asthma-related conditions such as allergic bronchopulmonary aspergillosis (ABPA) or more severe chronic lung disease, need treatment with steroid medicines at some point. These treatments — often essential to control inflammation, protect the lungs, and improve breathing — may include repeated or long-term courses of steroids such as prednisolone.
When steroid treatment is used over time, it can reduce the body’s own production of cortisol by the adrenal glands. In some people, the adrenal glands do not fully recover, leading to adrenal insufficiency. Cortisol is a vital hormone that helps the body manage energy, illness, infection, and physical stress. When it cannot be made reliably, hydrocortisone replacement is needed to keep the body safe and functioning.
In this situation, hydrocortisone is prescribed to replace the cortisol your body can no longer make, usually after prednisolone has been reduced or stopped, or when prednisolone is no longer needed to control lung inflammation but adrenal support is still required.
Adrenal insufficiency in people with aspergillosis is not a failure and not something you have caused. It is a recognised consequence of necessary treatment for a serious, long-term condition. With the right information, a personalised dosing plan, and medical support, adrenal insufficiency can be managed safely alongside aspergillosis.
A patient guide to everyday (basal) dosing, higher-dose needs, and short-term stress dosing
If you take hydrocortisone because you have adrenal insufficiency, understanding how your dose works — both day to day and during illness or stress — is essential for your safety and wellbeing.
This guide explains:
-
What your basal (everyday) dose is for
-
Why some people need higher basal doses
-
When and how stress dosing is used — and why it is short term
-
Why some doctors may hesitate — and how to work safely with them
-
Where to find trusted patient and clinician resources
Very important first point ❗
Any changes to your hydrocortisone dose must be agreed in advance with a doctor or specialist nurse who knows your adrenal insufficiency.
This includes:
-
Your usual daily dose
-
Your stress-dosing (“sick day”) plan
-
Emergency injection instructions
This guide does not replace medical advice.
It is designed to help you understand your treatment and communicate clearly with healthcare professionals.
1) Your basal (everyday) hydrocortisone dose
What the basal dose is for
Your basal dose is the hydrocortisone you take on an ordinary day, when you are not ill or under unusual stress. Its purpose is to:
-
Replace the cortisol your body cannot make reliably
-
Support normal daily function (energy, blood pressure, mood)
-
Help your body feel stable and safe
-
Reduce the risk of chronic under-replacement
It is replacement, not treatment for inflammation.
A key point many patients are not told
Being consistently under-replaced does not help adrenal recovery.
Ongoing symptoms such as:
-
Constant exhaustion
-
Dizziness or nausea on standing
-
Brain fog or low mood
-
Poor tolerance of everyday stress
-
Frequent “crashes” or infections
can delay recovery, not speed it. Stability supports healing.
What doctors usually mean by a “physiological” dose
Most adults naturally produce the equivalent of about 15–25 mg of hydrocortisone per day.
Doctors aim for a dose in this range and adjust for:
-
Body size
-
Activity level
-
Other medical conditions
-
Individual response
This is replacement, not “high-dose steroids”.
How basal hydrocortisone is usually taken
To mimic the body’s natural rhythm, doses are often split:
-
A larger dose in the morning
-
Smaller doses later in the day
-
Avoiding late evening doses where possible
This supports:
-
Energy and blood pressure
-
Sleep
-
Mood and concentration
Signs your basal dose may be too low
Tell your doctor if you have persistent:
-
Severe fatigue despite rest
-
“Wired but empty” feeling
-
Dizziness, nausea, or salt craving
-
Poor concentration or memory
-
Low mood or anxiety
-
Frequent need for rescue or stress doses
These symptoms matter even if blood tests look reassuring.
Blood tests are only part of the picture
Cortisol and ACTH tests:
-
Help with diagnosis
-
Are less helpful for adjusting daily dose
-
Do not always reflect how well you function
Doctors experienced with adrenal insufficiency rely heavily on how you feel and cope day to day.
The right balance
Rather than “as low as possible,” a safer aim is:
Low enough to avoid overtreatment, but high enough to live a stable, functional life.
Living in constant deficit is not success.
2) When a higher basal dose may be appropriate
Some people with adrenal insufficiency — particularly those with chronic illness — may genuinely need a higher basal hydrocortisone dose (for example 25–30 mg/day).
This does not automatically mean overtreatment.
Well-recognised examples include:
Chronic inflammatory lung disease (including ABPA)
-
Ongoing airway inflammation and immune activation
-
Recurrent infective or inflammatory flares
-
The body may never be in a true “resting” state
-
Standard doses may leave patients under-replaced
-
A stable higher dose can reduce repeated stress dosing and improve daily function
Frequent infections or slow recovery
-
Repeated illness or prolonged recovery
-
Frequent “temporary” stress dosing just to cope with everyday life
Long-standing steroid-induced adrenal insufficiency
-
Years of prednisolone or similar treatment
-
Deep suppression of the adrenal system
Larger body size or higher metabolic demand
-
Cortisol needs vary with body size and activity
Autonomic symptoms or low blood pressure
-
Postural dizziness or faintness
-
Often benefit from a higher morning dose
Clinical clue:
If someone repeatedly needs stress dosing just to manage ordinary days, their basal dose may be too low for their current physiology.
Important reassurance
-
Higher basal doses can be appropriate, temporary, or longer-term
-
They do not automatically prevent recovery
-
Ongoing inflammation and repeated physiological stress suppress recovery more than adequate replacement
-
Doses should always be prescribed, documented, and reviewed
3) Stress dosing — when your body temporarily needs more
What stress dosing means
A healthy body automatically makes more cortisol during:
-
Illness or infection
-
Fever
-
Vomiting or diarrhoea
-
Injury or trauma
-
Severe pain
-
Surgery or medical procedures
-
Major physical stress
If you have adrenal insufficiency:
➡️ your body cannot do this, so doctors prescribe stress dosing in advance as part of your safety plan.
Stress dosing is essential — but it is short term
Stress dosing is meant to last only as long as the stress lasts.
It covers a temporary increase in need, not your everyday requirements.
What “short term” usually means
Stress dosing may last:
-
24–48 hours for minor illness or fever
-
Several days for infections or recovery from injury
-
During and immediately after surgery or procedures
Your doctor should advise:
-
When to increase
-
How much to increase
-
When and how to return to your usual dose
Why stress dosing should not continue indefinitely
If higher doses are needed for longer, something usually needs review:
-
Infection or inflammation has not settled
-
The basal dose may be too low
-
Another medical problem is present
If stress dosing is still needed after the original stress has passed, it’s time to talk to your doctor.
Stepping back down safely
-
Doctors usually advise returning to baseline
-
Sometimes a 1–2 day step-down is used
-
You should not remain on stress doses “just in case”
Stress dosing does NOT:
-
Stop adrenal recovery
-
Mean you are “failing”
-
Cause long-term harm when used correctly
Not stress dosing can:
-
Make you seriously unwell
-
Delay recovery
-
Lead to adrenal crisis


4) Why some doctors seem hesitant
Doctors outside endocrinology (GPs, A&E, ward teams):
-
Are trained to minimise steroid use
-
Often think of steroids only as anti-inflammatory drugs
-
May rarely manage adrenal insufficiency
What they may not realise immediately:
Your hydrocortisone is replacing a missing hormone — it is essential, not extra.
5) How to advocate safely (with medical backing)
It is appropriate to say:
“I have adrenal insufficiency. My doctor has advised stress dosing during illness to prevent adrenal crisis.”
If you have them, show:
-
Your Steroid Emergency Card
-
A written stress-dosing plan
-
A clinic letter or summary
6) Trusted resources & further support (with links)
The following organisations provide reliable, clinician-endorsed information on adrenal insufficiency, hydrocortisone replacement, stress dosing, and emergency care.
They are widely recognised by NHS endocrinology teams and safe to share with patients, families, and healthcare professionals.
UK patient and professional resources
Addison’s Disease Self-Help Group (ADSHG)
Website: https://www.addisonsdisease.org.uk
What it offers:
-
Clear explanations of basal vs stress dosing
-
Patient-friendly sick-day rules
-
Emergency hydrocortisone injection guidance
-
Downloadable patient leaflets used in NHS clinics
-
Webinars, helpline, and peer support
Why it’s useful:
ADSHG explicitly supports individualised dosing and crisis prevention.
Society for Endocrinology
Steroid Emergency Card & adrenal crisis guidance:
https://www.endocrinology.org/clinical-practice/steroid-emergency-card/
Why it’s useful:
-
Highly trusted by doctors, A&E, and ward teams
-
Clear professional wording that reassures non-specialists
-
Supports rapid decision-making in emergencies
NHS (England)
Steroid Emergency Card information:
https://www.nhs.uk/conditions/steroid-emergency-card/
Why it’s useful:
-
Official NHS backing
-
Useful for legitimacy in emergency or inpatient settings
International patient resources (useful supplements)
Endocrine Society
Patient information on adrenal insufficiency:
https://www.endocrine.org/patient-engagement/endocrine-library/adrenal-insufficiency
Why it’s useful:
-
Clear explanations of cortisol physiology
-
Conservative, authoritative tone
-
Helpful for patients seeking international consensus
National Adrenal Diseases Foundation (NADF)
Website: https://www.nadf.us
What it offers:
-
Practical sick-day rules
-
Emergency preparedness guidance
-
Injection training resources
Particularly helpful for patients with long-standing adrenal insufficiency or frequent illness.
Resources especially relevant for ABPA & chronic lung disease
National Aspergillosis Centre
Website: https://mft.nhs.uk/wythenshawe/services/infectious-diseases/national-aspergillosis-centre/
Why it’s relevant:
-
Specialist centre where ABPA and adrenal insufficiency often overlap
-
Supports personalised care plans in complex disease
Aspergillosis Trust
Website: https://www.aspergillosistrust.org
Why it’s useful:
-
Patient-focused education and advocacy
-
Helps explain the chronic physiological stress of ABPA
-
Supports conversations about higher basal hydrocortisone needs
Quick-access patient checklist (phone / wallet)
Patients are encouraged to keep:
-
Steroid Emergency Card
-
Sick-day rules (ADSHG)
-
Personal stress-dosing plan (agreed with doctor)
-
Clinic letter or summary
Many patients keep photos of these documents on their phone for emergencies.
Final reassurance
These resources support — not replace — medical advice.
They exist to help patients stay safe, informed, and confident when managing hydrocortisone and communicating with healthcare professionals.
Season’s Greeting
As the year draws to a close, we would like to send warm wishes to everyone in the aspergillosis community — patients, families, carers, clinicians, nurses, scientists, and all professionals working to improve care and understanding.
Living with aspergillosis, or supporting those who do, often requires resilience, patience, and compassion. Throughout this year, we have seen remarkable strength from patients, dedication from healthcare teams, and generosity of spirit across our wider community.
At this time of reflection and renewal — whether you mark Christmas, another festival, or simply the turning of the year — we hope you find moments of rest, comfort, and connection. May the days ahead bring steadier health where possible, renewed energy, and continued progress in care, research, and support.
Thank you for being part of this community.
With warmest wishes for peace, kindness, and hope — now and into the New Year.
New UK Best Practice Guidelines for Diagnosing Fungal Diseases – What Patients Need to Know
A major new set of UK guidelines has just been published on how doctors and laboratories should diagnose serious fungal diseases. These come from the British Society for Medical Mycology (BSMM) and aim to make diagnosis faster, more accurate, and more consistent across the country.
For patients and families, this is very good news.
Why does this matter?
Fungal diseases can be difficult to diagnose. Many symptoms overlap with other conditions, and traditional tests sometimes miss infections. This can lead to delays, uncertainty, or unnecessary treatments.
The new BSMM recommendations help make sure that:
-
The right tests are used at the right time
-
Results come back more quickly
-
Hospitals know which modern tests give the best answers
-
Doctors can decide sooner whether antifungal treatment is needed
-
Unnecessary or ineffective treatments can be avoided
Overall, this means quicker diagnoses, fewer missed cases, and better care.
What is new in these guidelines?
The guidance highlights several improvements that directly benefit patients:
1. More accurate tests
Doctors are encouraged to use modern tests such as PCR, antigen tests, and antibody tests—these can detect fungal infections earlier and more reliably than traditional culture alone.
2. Faster turnaround times
Hospitals are encouraged to report important results within hours, not days. Faster answers mean faster treatment.
3. Better testing for people with chronic lung conditions
People with asthma, bronchiectasis, COPD, cystic fibrosis, or other long-term lung problems are now recognised as groups who may need access to fungal testing sooner.
4. Clearer pathways for difficult-to-diagnose conditions
Conditions such as chronic pulmonary aspergillosis (CPA), allergic bronchopulmonary aspergillosis (ABPA), fungal bronchitis and invasive fungal infections now have clearer testing strategies.
5. Stronger links to antifungal stewardship
This means using antifungal medicines only when they are truly needed, helping prevent side-effects and resistance.
6. Guidance for hospitals with fewer resources
The document includes a step-by-step approach to help smaller or overseas hospitals improve gradually.
What does this mean for patients?
If you are living with, or being assessed for, a fungal condition, you should expect:
-
More consistent tests wherever you are treated in the UK
-
Better access to specialist testing when needed
-
Earlier and more confident diagnoses
-
More appropriate treatments, with less trial-and-error
-
Closer monitoring during treatment
These improvements could reduce anxiety, cut down on repeated appointments, and help your clinical team make clearer decisions.
Who should be following these guidelines?
-
Hospital laboratories
-
Doctors and nurses in respiratory, ICU, oncology, infectious diseases, and transplant services
-
GP practices referring patients for investigation
-
Healthcare commissioners
-
Hospitals outside the UK wanting to improve fungal care
Patients and carers can also play a part by asking:
“Does my hospital follow the latest BSMM recommendations for fungal testing?”
The bottom line
These new BSMM guidelines are a major step forward for anyone affected by fungal disease. They promote earlier diagnosis, better access to testing, safer treatment, and improved outcomes.
Putting these recommendations into everyday practice—across the UK and worldwide—has the potential to transform care and save lives.










