Trials, systematic reviews, and state-of-the-science reviews from ~2016–2026 on damp housing, mould, and health
Executive summary (what 10 years of evidence consistently shows)
1) Damp and mouldy housing is a causal driver of respiratory disease
-
Strong, repeated associations with asthma incidence, asthma exacerbations, wheeze, chronic cough, and poorer lung function, especially in children.
-
Effects persist across countries, climates, and housing systems.
-
Evidence is strongest for asthma and allergic respiratory disease, but extends to bronchitis, infections, and symptom burden in people with existing lung disease.
2) Health effects are dose-related, not binary
-
Risk increases with extent, persistence, and visibility of dampness/mould (patch size, odour, condensation, repeated water damage).
-
No safe threshold has been identified → “any dampness matters.”
3) Mental health impacts are now well-established
-
Damp and mould exposure is associated with depression, anxiety, stress, sleep disturbance, and reduced wellbeing.
-
Pathways are both biological (inflammation, immune activation) and psychosocial (lack of control, stigma, housing insecurity).
4) Children are disproportionately affected
-
Strong paediatric evidence links damp homes to asthma development, poorer asthma control, and higher healthcare use.
-
Early-life exposure appears particularly important.
5) Damp housing is a marker of structural inequality
-
Concentrated in low-income, overcrowded, poorly maintained, or privately rented housing.
-
Acts as a health inequality amplifier, not just an environmental exposure.
6) Remediation works—but prevention works better
-
Interventions that fix the building (leaks, insulation, ventilation) improve symptoms.
-
Education alone is insufficient if the housing defect remains.
Thematic synthesis of the literature
1. Respiratory health (strongest evidence base)
Consistent findings across reviews (2016–2025):
-
Dampness and mould exposure increases:
-
Asthma onset in children
-
Asthma severity and exacerbations
-
Wheeze, cough, breathlessness
-
-
Associations hold even after adjusting for smoking, socioeconomic status, and outdoor pollution.
Key insight
Damp housing is not merely an “asthma trigger” — it is a risk factor for developing disease, especially in childhood.
2. Childhood lung health (very strong, clinically relevant)
-
Paediatric reviews emphasise that clinicians routinely see children whose symptoms are driven or sustained by housing conditions.
-
Poor housing undermines:
-
Controller medication effectiveness
-
Self-management plans
-
Long-term lung development
-
Clinical implication
Asking about housing conditions should be as routine as asking about pets or smoking in paediatric respiratory clinics.
3. Mental health and wellbeing (rapidly strengthening evidence)
Recent state-of-the-science reviews conclude:
-
Damp and mould exposure is associated with:
-
Depression
-
Anxiety
-
Psychological distress
-
-
Effects persist even when respiratory disease is accounted for.
Mechanisms proposed
-
Chronic inflammation and immune signalling
-
Sleep disruption
-
Loss of control and “housing stress”
-
Fear for children’s health
Important shift
Damp housing is no longer viewed as purely a respiratory issue—it is a whole-person health exposure.
4. Measurement and exposure assessment (important but imperfect)
What works reasonably well
-
Visual inspection and standard dampness indices
-
Structured questionnaires (especially for asthma cohorts)
-
ERMI (Environmental Relative Moldiness Index) as a research tool
What does NOT yet exist
-
A clinically agreed safe exposure threshold
-
A single test that rules exposure in or out
Consensus
Absence of a perfect test does not mean absence of harm.
5. Built environment, ventilation, and remediation
Clinical trials and housing intervention studies show:
-
Improved ventilation and moisture control:
-
Reduces indoor humidity
-
Improves reported physical and mental health
-
-
Poorly executed energy efficiency measures can worsen damp if ventilation is not addressed.
Critical point
“Warmth without ventilation” is a known failure mode.
6. Housing as a social determinant of health
Major public health frameworks now explicitly define healthy housing as:
-
Warm
-
Dry
-
Well-ventilated
-
Free from mould and toxins
-
Secure and accessible
Shift in framing
Damp housing is not an individual lifestyle issue—it is a system-level health determinant.
What the evidence does not support (important for countering misinformation)
-
No convincing evidence that:
-
“Detox” supplements treat mould exposure
-
Binding agents reverse health effects
-
Genetic susceptibility alone explains illness without exposure
-
-
Evidence strongly favours environmental remediation, not biomedical “workarounds”.
Implications for practice, policy, and patient support
For clinicians
-
Ask about damp and mould explicitly.
-
Document housing conditions when symptoms are unexplained or refractory.
-
Support patients with letters or reports—this is evidence-based advocacy, not speculation.
For public health & housing services
-
Damp housing remediation is preventive medicine.
-
Children’s respiratory health and mental health outcomes justify investment.
For patients
-
Symptoms are not imagined.
-
The problem is the building, not personal failure.
-
Improvement often requires structural change, not just treatment escalation.
Bottom line (10-year consensus)
Damp and mouldy housing causes avoidable disease, worsens inequality, and undermines medical care.
Fixing homes is one of the most effective—and underused—public health interventions available.
References
-
Bentley R, Mason K, Jacobs D, Blakely T, Howden-Chapman P, Li A, Adamkiewicz G, Reeves A.
Housing as a social determinant of health: a contemporary framework. Lancet Public Health. 2025;10(10):e855–e864. doi:10.1016/S2468-2667(25)00142-2. PMID: 40953578. -
Moorcroft C, Whitehouse A, Grigg J.
Damp and mouldy home: impact on lung health in childhood. Archives of Disease in Childhood. 2025;110(6):419–421. doi:10.1136/archdischild-2023-326035. PMID: 39814530. -
Gatto MR, Mansour A, Li A, Bentley R.
A state-of-the-science review of the effect of damp- and mold-affected housing on mental health. Environmental Health Perspectives. 2024;132(8):086001. doi:10.1289/EHP14341. PMID: 39162373. -
Patti MA, Henderson NB, Phipatanakul W, Jackson-Browne M.
Recommendations for clinicians to combat environmental disparities in pediatric asthma. Chest. 2024;166(6):1309–1318. doi:10.1016/j.chest.2024.07.143. PMID: 39059578. -
Punyadasa D, Adderley NJ, Rudge G, Nagakumar P, Haroon S.
Self-reported questionnaires to assess indoor home environmental exposures in asthma patients: a scoping review. BMC Public Health. 2024;24:2915. doi:10.1186/s12889-024-20418-8. PMID: 39434085. -
Kozajda A, Miśkiewicz E.
Exposure to bioaerosol in the residential environment. Medycyna Pracy. 2024;75(6):545–560. doi:10.13075/mp.5893.01508. PMID: 39688367. -
Vesper SJ.
The development and application of the Environmental Relative Moldiness Index (ERMI). Critical Reviews in Microbiology. 2025;51(2):285–295. doi:10.1080/1040841X.2024.2344112. PMID: 38651788. -
Nabaweesi R, Hanna M, Muthuka JK, Samuels AD, Brown V, Schwartz D, Ekadi G.
The built environment as a social determinant of health. Primary Care. 2023;50(4):591–599. doi:10.1016/j.pop.2023.04.012. PMID: 37866833. -
Grant TL, Wood RA.
The influence of urban exposures and residence on childhood asthma. Pediatric Allergy and Immunology. 2022;33(5):e13784. doi:10.1111/pai.13784. PMID: 35616896. -
Coulburn L, Miller W.
Prevalence, risk factors and impacts related to mould-affected housing: an Australian integrative review. International Journal of Environmental Research and Public Health. 2022;19(3):1854. doi:10.3390/ijerph19031854. PMID: 35162876. -
Wimalasena NN, Chang-Richards A, Wang KI, Dirks KN.
Housing risk factors associated with respiratory disease: a systematic review. International Journal of Environmental Research and Public Health. 2021;18(6):2815. doi:10.3390/ijerph18062815. PMID: 33802036. -
Ali SH, Foster T, Hall NL.
The relationship between infectious diseases and housing maintenance in Indigenous Australian households. International Journal of Environmental Research and Public Health. 2018;15(12):2827. doi:10.3390/ijerph15122827. PMID: 30545014. -
Wolkoff P.
Indoor air humidity, air quality, and health – an overview. International Journal of Hygiene and Environmental Health. 2018;221(3):376–390. doi:10.1016/j.ijheh.2018.01.015. PMID: 29398406. -
Mendell MJ, Kumagai K.
Observation-based metrics for residential dampness and mold with dose–response relationships to health: a review. Indoor Air. 2017;27(3):506–517. doi:10.1111/ina.12342. PMID: 27663473. -
Francisco PW, Jacobs DE, Targos L, Dixon SL, Breysse J, Rose W, Cali S.
Ventilation, indoor air quality, and health in homes undergoing weatherization: a randomized trial. Indoor Air. 2017;27(2):463–477. doi:10.1111/ina.12325. PMID: 27490066. -
Barnes CS, Horner WE, Kennedy K, Grimes C, Miller JD.
Home assessment and remediation. Journal of Allergy and Clinical Immunology: In Practice. 2016;4(3):423–431.e15. doi:10.1016/j.jaip.2016.01.006. PMID: 27157934. -
Chew GL, Horner WE, Kennedy K, Grimes C, Barnes CS, Phipatanakul W, Larenas-Linnemann D, Miller JD.
Procedures to assist health care providers to determine when home assessments for potential mold exposure are warranted. Journal of Allergy and Clinical Immunology: In Practice. 2016;4(3):417–422.e2. doi:10.1016/j.jaip.2016.01.013. PMID: 27021632. -
Vesper S, Wymer L.
The relationship between Environmental Relative Moldiness Index values and asthma. International Journal of Hygiene and Environmental Health. 2016;219(3):233–238. doi:10.1016/j.ijheh.2016.01.006. PMID: 26861576.
Invitation: Patient & Carer Discussion on Living with ABPA. New type of treatment.
🕙 10:00am, Thursday 12th
Get details on how to join us by clicking on the link below and choosing Thursday 12th Patients Support Meeting - you will be sent a link to the meeting via email.
https://outlook.office.com/book/[email protected]/
We are inviting people living with Allergic Bronchopulmonary Aspergillosis (ABPA), and those who care for them, to take part in an open, informal online discussion with argenx, a research-focused biotechnology company.
argenx would like to listen directly to patients and carers to better understand what day-to-day life with ABPA is really like. There is no need to prepare anything in advance — you are welcome simply to listen, or to share as much or as little as you feel comfortable.
They are particularly interested in hearing about:
-
Patients’ and carers’ journeys living with ABPA
-
Which symptoms are most burdensome in everyday life (for example breathlessness, cough, fatigue, thick mucus or mucus plugs)
-
Where current treatments fall short from a patient or carer perspective
-
What would make patients or carers feel motivated or reassured about taking part in a future clinical trial of a new ABPA therapy
The purpose of this conversation is to help researchers design future studies that reflect what matters most to patients, including which outcomes are meaningful and how trials can be made more patient-friendly.
📅 Date: Thursday 12th
🕙 Time: 10:00am
💬 Format: Open, informal discussion
📝 Preparation: None required
If you are living with ABPA, or care for someone who is, and would be interested in attending, please let us know.
A short explainer: what is ARGX-118?
argenx is developing an investigational (research-stage) treatment called ARGX-118. It is not yet a licensed medicine and is not currently available outside of research studies.
In ABPA, many people experience very thick, sticky mucus and mucus plugs that block airways and contribute to breathlessness, cough, and flare-ups. Research has shown that this mucus can sometimes contain microscopic crystals formed from proteins released by certain white blood cells involved in allergic inflammation. These crystals can make mucus denser and harder to clear.
ARGX-118 is designed to target and break down these crystals, with the aim of making mucus less thick and easier to clear from the lungs. This is a different approach from current treatments, which mainly focus on suppressing inflammation (such as steroids or biologics) or reducing fungal burden (antifungal medicines).
Because ARGX-118 is still in early development, we do not yet know how effective it will be, who might benefit most, or how it would fit alongside existing treatments. That is exactly why argenx wants to hear from patients and carers now — to understand real-world symptoms, treatment gaps, and what would genuinely matter if a future clinical trial were developed.
👉 Attending this meeting does not commit you to any trial and will not affect your care. It is simply an opportunity to share experiences and help shape future research, if you wish.
Systemic fungal infections: why speed, diagnosis and stewardship matter
Systemic fungal infections — including aspergillosis, candidiasis, cryptococcosis, mucormycosis and pneumocystis pneumonia — are medical emergencies. When diagnosis or treatment is delayed, mortality rises sharply. This comprehensive review brings together current understanding of how these infections arise, why they are so difficult to diagnose, and what is needed to improve outcomes.
Why fungal infections are often missed
Unlike many bacterial infections, systemic fungal infections can be hard to confirm quickly. Fungal organisms are often present in low numbers, may be released intermittently into the bloodstream, and can be difficult to grow in standard cultures. As a result, no single test is usually sufficient, and clinicians often need a combination of imaging, cultures, antigen tests, molecular tests (PCR), and histopathology.
Because delay can be fatal, antifungal treatment is frequently started on clinical suspicion alone — especially in critically ill or immunocompromised patients. The paper emphasises that this approach is often necessary, but it must be paired with a clear diagnostic strategy.
Antifungal stewardship: knowing when to stop
A central message of the paper is that diagnostic tests are just as important for stopping treatment as for starting it. Antifungal drugs can be toxic, interact with many other medicines, and drive antifungal resistance if used unnecessarily.
The authors stress that:
-
Diagnostic results should be actively reviewed
-
Antifungal therapy should be stopped or stepped down if infection is not supported by evidence
-
This approach protects patients and preserves antifungal effectiveness
Antifungal resistance is a growing threat
Antifungal resistance is no longer rare. The review highlights:
-
Azole resistance in Aspergillus, including cryptic species
-
Rising resistance in several Candida species
-
The global spread of multidrug-resistant Candida auris
Because of this, the authors recommend that all clinically relevant fungal isolates are identified to species level and tested for antifungal susceptibility wherever possible. Making assumptions about drug sensitivity is increasingly unsafe.
Aspergillosis: a broad spectrum of disease
The paper clearly outlines the many forms of aspergillosis, ranging from:
-
Allergic disease (such as allergic bronchopulmonary aspergillosis)
-
Chronic pulmonary aspergillosis, often in people with underlying lung damage
-
Subacute and acute invasive disease, particularly in immunocompromised or critically ill patients
Importantly, the review notes that aspergillosis is not limited to severely immunocompromised people. Chronic and subacute forms often occur in individuals with structural lung disease who are otherwise immunocompetent.
Climate change and emerging fungal risks
One of the most forward-looking sections of the paper addresses how climate change and natural disasters are altering fungal disease patterns. Rising environmental temperatures, flooding, storms and environmental disruption are:
-
Increasing exposure to environmental fungi
-
Enabling fungi to adapt to higher temperatures
-
Contributing to outbreaks after natural disasters and trauma
-
Expanding fungal diseases into new geographic regions
The authors argue that fungal infections must be considered part of future public health and healthcare resilience planning.
Key take-home messages
-
Systemic fungal infections are time-critical medical emergencies
-
Diagnosis usually requires multiple tests, not a single result
-
Early antifungal treatment is often necessary — but must be reviewed
-
Diagnostics are essential for safe antifungal stewardship
-
Antifungal resistance is a real and growing problem
-
Climate change is reshaping fungal epidemiology and risk
Free access to the full article
Elsevier has provided free access to the full paper for a limited time (no registration required):
👉 https://authors.elsevier.com/a/1mZqR4qdNoJLH2
🗓️ Available until 28 March 2026
This article is recommended reading for patients wanting a deeper understanding of fungal disease, as well as clinicians, microbiology teams, and healthcare planners.
Weekly Aspergillosis Update (2–9 February 2026)(Week 5).
This week’s papers cluster around: (1) ICU/viral-pneumonia–associated invasive pulmonary aspergillosis (IPA),
(2) tuberculosis (TB)–chronic pulmonary aspergillosis (CPA) overlap,
(3) diagnostic criteria and emerging detection approaches, and
(4) antifungal drug interaction risk.
Top highlights (quick take)
- CAPA criteria matter: case rates vary substantially depending on which definition is used (AspICU vs ISHAM vs EORTC).
- Viral illness + immune dysfunction = early IPA risk: data add to the “risk stacking” story (including SFTS and broader viral pneumonia).
- TB–CPA remains a major clinical challenge: CPA can be misread as TB relapse; delayed recognition worsens outcomes.
- Safety: rifapentine can markedly reduce voriconazole exposure (important in TB–aspergillosis co-infection).
1) ICU, Viral Pneumonia & CAPA / IPA
Decoding CAPA: A Comparative Study of Aspicu, Isham, and Eortc Criteria in Critical COVID-19 Patients Requiring Mechanical Ventilation (Preprint)
Taleb C, Lelubre C, Biston P, Piagnerelli M. Preprints.org. 04 Feb 2026. PPR: PPR1150994
- What they did: compared CAPA classification using AspICU, ISHAM and EORTC-style criteria in ventilated COVID-19 patients.
- Key point: CAPA “incidence” changes materially depending on the criteria applied; distributions differed across COVID-19 waves.
- Why it matters: reinforces that audits, research comparisons and ICU protocols must state which definition is used (and why).
Characteristics of T-lymphocyte subsets in patients with severe fever with thrombocytopenia syndrome complicated with invasive pulmonary aspergillosis: a retrospective study
Xu Y, Liu Y, Qian Y, et al. Front Immunol. 09 Feb 2026. PMCID: PMC12876148
- What they found: SFTS patients complicated by IPA showed marked T-cell subset abnormalities and high reported secondary IPA rates.
- Clinical takeaway: another example of viral immune dysregulation predisposing to IPA—analogous to influenza-associated IPA and CAPA.
- Practice relevance: supports heightened fungal vigilance in severe viral syndromes with immune suppression features.
Immunocompromise and early-onset invasive pulmonary aspergillosis in viral pneumonia: a retrospective cohort study
Sun B, Shen J, Dong M, et al. Front Public Health. 02 Feb 2026. PMCID: PMC12852324
- Theme: early IPA can emerge in viral pneumonia in the setting of immunocompromise (not only classic neutropenia).
- Why it matters: backs the “risk stacking” concept—viral lung injury + immune dysfunction (often steroids) can accelerate IPA risk.
- Use: helpful citation for ICU pathways and education materials.
The COVID-19 pandemic: an underlying factor for increased Stenotrophomonas maltophilia infections—A literature review and case study analysis (Review)
Pompilio A, Di Bonaventura G. Front Microbiol. 06 Feb 2026. PMCID: PMC12867275
- What’s relevant to aspergillosis: notes co-detection of Stenotrophomonas maltophilia in COVID-19 patients with invasive aspergillosis.
- Why it matters: underlines polymicrobial complexity in ICU; prompts questions about dysbiosis and pathogen interactions in severe disease.
Pulmonary Cavitation as a Late and Self-Limited Complication of COVID-19 Pneumonia: A Case Report
Osório M, Silveira M. Cureus. 02 Feb 2026. PMCID: PMC12852039
- Clinical reminder: post-COVID cavitation has a broad differential including CAPA and mucormycosis; requires careful exclusion of fungal disease.
- Why it matters: useful for follow-up imaging discussions and MDT differential diagnosis teaching.
2) TB–CPA overlap & antifungal pharmacology
Clinical features, diagnostic test performance, treatment and outcome of pulmonary tuberculosis patients with chronic pulmonary aspergillosis in China: a retrospective, observational study
Li J, Wu N, Mei C, et al. Front Cell Infect Microbiol. 06 Feb 2026. PMCID: PMC12864492
- Main message: CPA in TB patients is common and can be mistaken for TB relapse; diagnostic delay is consequential.
- Why it matters: strong global relevance—TB remains one of the biggest drivers of CPA burden.
- Use: good reference for post-TB lung disease pathways and CPA awareness materials.
A clinically significant interaction between voriconazole and rifapentine: a case report and review of evidence
Chen T, Chen X, Zhang Q. Front Med (Lausanne). 09 Feb 2026. PMCID: PMC12875967
- What happened: TB–aspergillosis co-infection complicated by rifapentine–voriconazole interaction.
- Key point: rifapentine (a potent enzyme inducer) can substantially reduce voriconazole exposure → risk of treatment failure.
- Why it matters: high-impact safety message; supports use of therapeutic drug monitoring and/or alternative strategies in TB co-treatment.
3) Diagnostics & detection methods
Combined Biospectroscopy with Multivariate Analysis for the Differential Diagnosis of Leptospirosis Disease: A Pilot Study
Zambrano A, Trilleras J, Arana Rengifo V, et al. ACS Omega. 09 Feb 2026. PMCID: PMC12878783
- Why it’s here: includes a small aspergillosis group among comparator infections.
- What it suggests: biospectroscopy + multivariate modelling may separate infections via biochemical “fingerprints” (early-stage concept).
- Bottom line: promising research direction, but not near-term clinical practice.
Research progress on the current status of respiratory pathogen infections and their detection methods (Review)
Zhu F, Peng M, Chen A, Zhu Q. Front Microbiol. 09 Feb 2026. PMCID: PMC12876234
- Scope: broad overview of respiratory pathogen detection, including invasive and allergic aspergillosis concepts.
- Useful for: background reading for non-specialists and training materials (diagnostic modalities and limitations).
4) Aspergillus biology, pathology & wider fungal immunology
Characterization of a bZIP Transcription Factor ZipD in Aspergillus flavus
Jeong D, Cho H, Park H. Mycobiology. 06 Feb 2026. PMCID: PMC12865826
- What it is: basic science on gene regulation (ZipD) in Aspergillus flavus.
- Why it matters: contributes to long-term understanding of fungal stress responses and potential future targets.
Mechanistic Insights into Calcium Oxalate Crystals in Aspergillosis of the Maxillary Sinus
Trimukhe A, Bhatt K, Mridha AR, et al. Head Neck Pathol. 02 Feb 2026. PMID: 41627592
- Key message: calcium oxalate crystal deposition is a mechanistic contributor to local inflammation/tissue injury in sinus aspergillosis.
- Clinical relevance: useful for ENT/pathology audiences; supports recognition of crystals as an important clue.
Adjunctive GM-CSF therapy enhances host defense against systemic Candida auris infection in immunosuppressed mice
Mattos E, Das Gupta K, Quintanilla D, et al. Front Immunol. 06 Feb 2026. PMCID: PMC12862068
- Why included: host-directed immunotherapy concepts often discussed alongside invasive aspergillosis.
- Takeaway: GM-CSF improved antifungal host defense in a preclinical model—supporting interest in adjunctive approaches (not clinical guidance).
The therapeutic potential of high-dose inhaled nitric oxide for antimicrobial effects: a narrative review and future directions (Review)
Berra L, Kamenshchikov N, Tal A, et al. Intensive Care Med Exp. 05 Feb 2026. PMCID: PMC12872992
- Scope: experimental antimicrobial strategy, mainly ICU-focused.
- Relevance: future-facing adjunct discussion rather than current aspergillosis practice.
5) Case reports & broader context (selected)
Case Report: Triple autoimmune overlap: rheumatoid arthritis, systemic lupus erythematosus, and hypereosinophilic asthma with systemic manifestations
Front Immunol. 02 Feb 2026. PMCID: PMC12852425
- Aspergillosis relevance: ABPA considered in complex eosinophilic/asthma phenotypes; reminder that ABPA can present atypically (e.g., without classic bronchiectasis early on).
- Use: supports education on diagnostic nuance in asthma/eosinophilic lung disease.
HIV-associated neurological infections in a Brazilian tertiary care center: clinical-epidemiological features and predictors of in-hospital mortality
Ramos L, Ninomiya D, Sequeira M, et al. Rev Inst Med Trop Sao Paulo. 02 Feb 2026. PMCID: PMC12858172
- Context: opportunistic infection landscape in advanced HIV; useful epidemiological background (limited direct aspergillosis focus).
Note: This page summarises research and does not replace clinical guidance. If you are a patient and have concerns about symptoms or treatment, contact your clinical team.
Genes and aspergillosis: why the same fungus causes different problems in different people
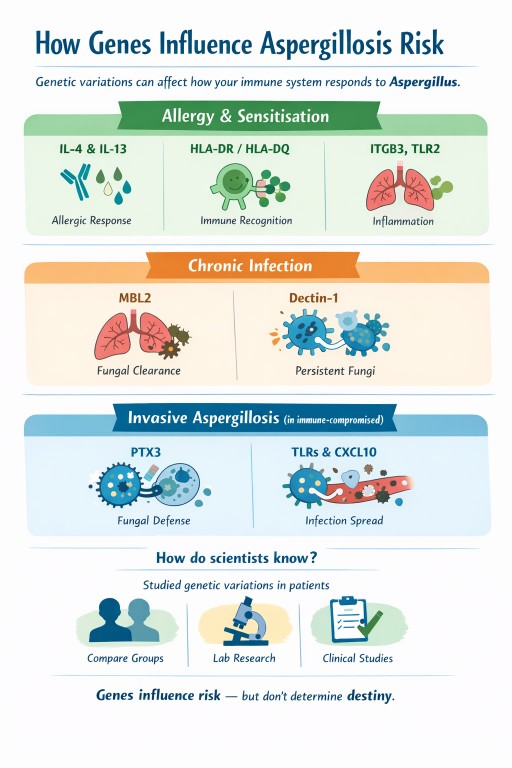
Why look at genes when talking about aspergillosis?
The theme of World Aspergillus Day 2026 was “How can the genomics revolution help patients with chronic aspergillosis?”
To answer that, we need to look briefly at genes and what they tell us about how the body resists infection.
Genes are the body’s instruction manual. They help control how our immune system works, how inflammation is managed, and how well we clear infections. Humans have around 25,000 genes, with two copies of each in almost every cell — and billions of cells using these instructions every day.
Small, natural differences in genes help explain why people respond differently to Aspergillus: some develop allergy, others chronic infection, and many clear it without any illness at all. Genes don’t determine outcomes, but they help us understand why the immune response differs between people.
Many people ask an understandable question:
“If we all breathe in Aspergillus spores, why do only some people get aspergillosis – and why does it look so different from person to person?”
Part of the answer lies in genes.
Genes do not cause aspergillosis on their own, but they can influence how the immune system responds once the fungus is encountered.
A simple way to think about genes
Genes act like settings, not switches.
They can influence:
-
how strongly your immune system reacts
-
whether that reaction is allergic, chronic, or weak
-
how well fungi are cleared from the lungs
Genes do not override:
-
lung damage (asthma, bronchiectasis, old infections)
-
steroid or immunosuppressive treatment
-
mould exposure levels
They help explain patterns of illness, not certainty.
Risk stacking: why combinations matter more than any single factor
Aspergillosis rarely develops because of one single cause. Instead, it usually arises through risk stacking, where several small risk factors overlap at the same time.
Each factor may add only a little vulnerability on its own, but together they can tip the balance from resistance to disease.
This helps explain why aspergillosis often appears after years of stability, or during periods of change such as illness, medication adjustment, or increased environmental exposure.
What does risk stacking look like in practice?
A person might have:
-
mild genetic tendencies toward allergic inflammation or reduced fungal clearance
-
asthma, bronchiectasis, or old lung damage
-
long-term inhaled or oral corticosteroid treatment
-
periods of higher mould exposure (for example, damp housing or renovation work)
None of these alone guarantees illness.
But stacked together, they increase the chance that Aspergillus:
-
is recognised as an allergen rather than ignored
-
is not cleared efficiently from the lungs
-
triggers ongoing inflammation or chronic infection
Where genes fit into risk stacking
Genes usually act as background modifiers, not primary causes.
In people with healthy lungs and normal immunity, genetic differences rarely matter.
In people who already have lung disease, immune suppression, or repeated exposure, those same genetic differences can add to the overall risk stack.
This also explains why there is no single genetic test that can predict aspergillosis — risk depends on combinations, not on one gene.
Just as risks can add up, risk reduction also adds up. Improvements in airway clearance, asthma control, steroid management, and home environment can all meaningfully reduce overall risk.
Why this matters in aspergillosis
Aspergillosis is not one condition. It includes:
-
fungal sensitisation and allergy
-
chronic pulmonary infection
-
invasive disease in people with weakened immunity
Different genes influence different stages of the immune response, which helps explain why people experience very different forms of disease.
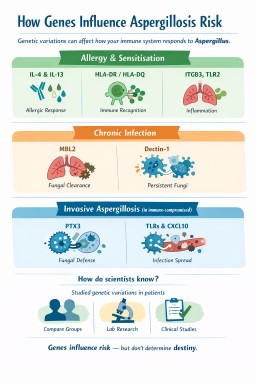
1. Genes linked to fungal allergy and sensitisation
These genes affect whether the immune system treats Aspergillus as a strong allergen.
IL-4, IL-13 and the IL-4 receptor
What they do
Control allergic inflammation, including:
-
immunoglobulin E (IgE)
-
eosinophils
-
mucus production
-
airway inflammation
What this means
Certain natural gene variants increase the likelihood of:
-
fungal sensitisation
-
asthma with fungal sensitisation
-
allergic bronchopulmonary aspergillosis (ABPA)
This fits closely with what patients experience clinically: high IgE, eosinophilia, steroid responsiveness, and response to biologic treatments.
HLA-DR and HLA-DQ
What they do
Help the immune system decide which proteins deserve attention.
What this means
Some HLA types present Aspergillus proteins in a way that:
-
encourages persistent allergic inflammation
-
increases the chance of ABPA
This helps explain why only a minority of people with asthma develop ABPA.
ITGB3 (integrin beta-3)
What it does
Helps airway and immune cells:
-
attach to surrounding tissue
-
communicate danger signals
-
interact with fungal-recognition pathways
What this means
Certain versions are linked to:
-
mould sensitisation
-
stronger immune signalling when fungal particles are present
This does not mean ITGB3 causes aspergillosis.
It helps explain why some people become sensitised more easily.
TLR2
What it does
Recognises fungal cell-wall components and triggers early immune responses.
What this means
Different versions can amplify or dampen inflammation, influencing sensitivity to fungi.
2. Genes linked to chronic pulmonary aspergillosis (CPA)
These genes influence how well fungi are cleared, especially in damaged lungs.
MBL2 (mannose-binding lectin)
What it does
Marks fungi so the immune system can remove them.
What this means
Low MBL activity may allow Aspergillus to persist once lung cavities or scarring exist.
Dectin-1 (CLEC7A)
What it does
Detects fungal cell-wall sugars and triggers antifungal responses.
What this means
Reduced detection can allow slow, long-term infection rather than allergy.
TLR4
What it does
Regulates inflammation in response to microbes.
What this means
Certain variants may influence how chronic inflammation and tissue damage evolve.
3. Genes linked to invasive aspergillosis
These matter most in people with weakened immune systems (for example, during chemotherapy or after transplant).
PTX3 (pentraxin-3)
What it does
Acts as an early fungal sensor and helps immune cells kill Aspergillus.
What this means
Reduced PTX3 activity is one of the strongest known genetic risk factors for invasive aspergillosis in high-risk medical settings.
TLR3 and interferon pathways (including CXCL10)
What they do
Coordinate immune communication and antifungal killing.
What this means
Impairment can delay fungal control and increase the risk of spread.
How do scientists know these genes are involved?
Researchers study natural genetic variations that:
-
are common in healthy people
-
are present from birth
-
usually cause small functional differences, not disease by themselves
They:
-
compare people with aspergillosis to similar people without it
-
identify gene variants linked to specific disease patterns
-
test how those genes affect fungal recognition, inflammation, or killing
-
confirm findings in laboratory and clinical studies
These are risk modifiers, not disease-causing genes.
Does this mean my family is at risk?
This is a very common concern. The reassuring answer for most people is:
No – aspergillosis does not usually run in families.
Why this is reassuring
-
These gene variants are common in the general population
-
Most people who carry them never develop aspergillosis
-
Aspergillosis requires other factors, such as lung disease, immune suppression, or heavy exposure
-
There is no consistent pattern of aspergillosis being passed from parent to child
Even strong genetic signals (such as PTX3) only increase risk in specific high-risk medical situations, not in healthy relatives.
Putting it all together
| Pattern of disease | Genes most often involved |
|---|---|
| Fungal sensitisation | IL-4, IL-13, IL-4 receptor, ITGB3, TLR2 |
| ABPA | IL-4/IL-13 pathway, HLA-DR/DQ, TLR3 |
| Chronic pulmonary aspergillosis | MBL2, Dectin-1, TLR4 |
| Invasive aspergillosis | PTX3, interferon pathways |
What this means for patients and families
-
Genetic testing is not routinely needed
-
These genes do not predict individual outcomes
-
Family members are not usually at increased risk
The most important factors remain:
-
good lung care
-
appropriate treatment
-
sensible mould exposure reduction
Genes influence risk — they do not determine destiny.
Can blood tests help predict if chronic pulmonary aspergillosis will come back?
This study from the National Aspergillosis Centre (NAC) looked at people with chronic pulmonary aspergillosis (CPA) who had completed antifungal treatment and asked a simple question:
Can blood tests tell us who is more likely to relapse after treatment stops?
What the researchers did
Doctors reviewed patients with CPA who had:
-
Taken antifungal treatment for at least 6 months
-
Stopped treatment because they were clinically stable
They then followed these patients to see who stayed well and who relapsed, and compared this with their blood test results at the time treatment stopped.
What they found
-
About 1 in 4 patients had a relapse after stopping treatment
-
People whose Aspergillus IgG blood test was still high at the end of treatment were much more likely to relapse
-
Patients whose IgG level had fallen to a lower level did not relapse in this study
-
Signs of Aspergillus allergy or sensitisation also increased relapse risk
-
CT scan appearances and treatment length alone were not reliable predictors
Why this matters for patients
This means that:
-
Blood tests may help doctors decide when it is safe to stop treatment
-
Some people may need closer follow-up or longer treatment
-
Follow-up can be more personalised, rather than “one size fits all”
Importantly, a relapse does not mean treatment failed — it reflects how persistent this infection can be in damaged lungs.
Key takeaway
A simple blood test at the end of treatment may help predict who needs closer monitoring for CPA relapse.
This research supports a more individualised approach to long-term CPA care.
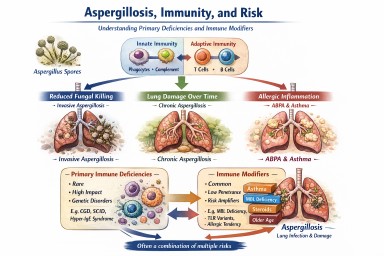
Aspergillosis, immunity, and risk
Primary immune deficiencies and immune modifiers explained
A single, comprehensive explainer for expert patients, carers, and non-specialists
Why this article exists
Aspergillus is a mould that everyone breathes in every day. Most people clear it without difficulty.
A small number of people develop aspergillosis because the balance between the fungus, the lungs, and the immune system is disturbed.
This article explains:
-
Rare primary (inherited) immune deficiencies that are clearly linked to aspergillosis
-
Common immune “modifier” factors that can increase risk or severity but do not cause disease on their own
-
How these factors stack together in real life
Key reassurance up front
There are 500+ recognised primary immune deficiencies
Only ~20–30 are clearly linked to aspergillosis
Most people with aspergillosis do not have any inherited immune disorder
The unifying concept: three immune pathways to aspergillosis
Almost all immune–aspergillus relationships fall into three mechanisms. Understanding these matters more than memorising names.
1. Reduced ability to kill the fungus
Some immune cells fail to destroy Aspergillus spores effectively.
→ Risk of invasive aspergillosis, sometimes severe or life-threatening.
2. Lung damage over time
Repeated infections or inflammation damage airways or leave cavities.
→ Risk of chronic pulmonary aspergillosis (CPA) or aspergillomas.
3. Excessive allergic inflammation
The immune system over-reacts to Aspergillus rather than failing to fight it.
→ Allergic bronchopulmonary aspergillosis (ABPA) and severe fungal-sensitised asthma.
Many conditions overlap more than one pathway.
Section 1: Primary (inherited) immune deficiencies clearly linked to aspergillosis
Rare, high-impact, and sometimes life-changing when present
These are the conditions clinicians usually mean when they talk about “immune causes of aspergillus disease”.
A. Phagocyte defects
Strongest association with invasive aspergillosis
-
Chronic granulomatous disease (CGD)
-
Autosomal recessive forms of CGD
-
Severe congenital neutropenia
-
Cyclic neutropenia
-
Leukocyte adhesion deficiency type I
Typical pattern
-
Aspergillosis at a young age
-
Invasive lung disease ± spread beyond lungs
-
Often no other obvious risk factors
B. Hyper-IgE and severe allergy syndromes
Allergic, chronic, and cavity-associated disease
-
STAT3 hyper-IgE syndrome
-
DOCK8 deficiency
-
PGM3 deficiency
-
ZNF341 deficiency
-
IL6ST deficiency
Typical pattern
-
Severe asthma and allergy
-
Thick mucus, recurrent infections
-
ABPA, later CPA or aspergillomas
C. Combined immunodeficiencies
Immune coordination problems
-
Severe combined immunodeficiency (milder or surviving forms)
-
Omenn syndrome
-
ZAP-70 deficiency
-
Major histocompatibility complex class II deficiency
-
CD40 ligand deficiency (hyper-IgM syndrome)
Typical pattern
-
Broad infection susceptibility
-
Aspergillosis can behave aggressively
D. Defects of fungal recognition and innate signalling
Often dramatic or unexpected presentations
-
CARD9 deficiency
-
Dectin-1 (CLEC7A) complete deficiency
-
MALT1 deficiency
Typical pattern
-
Severe or unusual aspergillosis
-
Lung, brain, or deep tissue involvement
-
Sometimes first presents in adulthood
E. Immune dysregulation syndromes
Mixed infection, inflammation, and autoimmunity
-
CTLA-4 haploinsufficiency
-
LRBA deficiency
-
STAT1 gain-of-function mutations
-
IPEX syndrome (FOXP3 deficiency)
Typical pattern
-
Inflammatory lung disease
-
Chronic or invasive aspergillosis emerging over time
F. Antibody deficiencies (indirect risk via lung damage)
-
Common variable immunodeficiency
-
X-linked agammaglobulinaemia
-
Activated PI3K-delta syndrome
Important nuance
Antibodies do not normally kill Aspergillus.
Risk arises after years of lung damage, not early in life.
Section 2: Immune modifier-types that can amplify risk
Common, low-penetrance, and often invisible on routine testing
These are not immune deficiencies, but they can influence who struggles, how severely, and why disease persists.
Mannose-binding lectin (MBL) deficiency
-
Common (≈5–10% of population)
-
Affects fungal recognition and complement activation
-
Usually mild on its own
-
Becomes relevant with lung disease, steroids, or other immune issues
Partial fungal-recognition receptor variants
-
Heterozygous dectin-1 variants
-
Toll-like receptor polymorphisms (for example TLR2, TLR4)
Effect
-
Slower fungal recognition
-
Increased colonisation or allergic response
-
Act as risk amplifiers, not causes
Cytokine balance variants
Small genetic differences affecting immune “signal strength”, including:
-
Interleukin-6
-
Interleukin-10
-
Tumour necrosis factor-alpha
These modify:
-
Inflammation intensity
-
Tissue damage vs clearance balance
Allergy-biased (Th2-skewed) immunity
Not a disease, but a recognised immune tendency.
Features:
-
Asthma
-
Eczema
-
Nasal polyps
-
High immunoglobulin E levels
-
Eosinophilia
Strongly associated with:
-
Fungal sensitisation
-
ABPA
-
Difficult-to-control asthma
Impaired mucociliary clearance
A functional immune–mechanical issue.
Seen in:
-
Severe asthma
-
Bronchiectasis
-
Chronic sinus disease
Effect:
-
Aspergillus spores are not physically cleared
-
Prolonged immune exposure
-
Increased colonisation and allergy
Age-related immune change (immunosenescence)
-
Normal reduction in immune speed and coordination with age
-
Particularly relevant to chronic pulmonary aspergillosis
Not a disease, but an important modifier of outcome.
Airway epithelial vulnerability
Subtle weaknesses in:
-
Airway lining integrity
-
Antimicrobial peptide production
-
Local immune signalling
Can increase:
-
Fungal adherence
-
Chronic colonisation
-
Allergic sensitisation
Section 3: Risk stacking – how this works in real life
Aspergillosis rarely results from one single factor.
Instead, several modest risks align:
-
Mild MBL deficiency
-
Severe asthma
-
Corticosteroid exposure
-
Bronchiectasis
-
Age-related immune change
→ Together, they create real disease risk, even though none alone would.
This explains why:
-
Two people with similar scans can behave very differently
-
One patient relapses while another stabilises
-
“Why me?” often has no single answer
Section 4: When clinicians investigate immune causes
Testing is targeted, not routine. It is usually considered when there is:
-
Aspergillosis at a young age
-
Invasive or unusually severe disease
-
Disease without classic risk factors
-
Recurrent infections plus severe asthma or allergy
-
A family history of unusual infections
Section 5: Why identifying (or excluding) immune factors helps
Understanding immune contribution can:
-
Explain disease pattern and behaviour
-
Guide antifungal choice and duration
-
Inform long-term prevention strategies
-
Reduce future lung damage
-
Reassure patients when no immune defect is found
Key take-home messages
-
Aspergillus exposure is universal; immune causes are rare
-
Only ~20–30 inherited immune deficiencies are clearly linked to aspergillosis
-
Modifier-type immune factors are common and usually harmless alone
-
Aspergillosis often reflects risk stacking, not a single diagnosis
-
Understanding patterns matters more than labels
-
Specialist care improves precision and outcomes
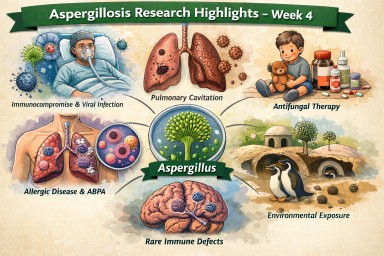
Latest Aspergillosis & Related Research Updates (Week 4).
Executive overview (what stands out this fortnight)
Key signals
-
Immune dysregulation—not just classic immunosuppression—continues to emerge as a central driver of invasive aspergillosis.
-
Allergic bronchopulmonary aspergillosis (Allergic Bronchopulmonary Aspergillosis) is appearing in atypical and early phenotypes, including absence of bronchiectasis.
-
Antifungal toxicity and pharmacokinetic variability remain clinically important.
-
Paediatric invasive aspergillosis evidence is improving.
-
Environmental and One Health studies continue to inform exposure risk.
-
Overlap with non-tuberculous mycobacteria and microbiome disruption is increasingly evident.
1. Immunocompromise, viral infection, and invasive aspergillosis
Immunocompromise and early-onset invasive pulmonary aspergillosis in viral pneumonia
Sun B et al., Frontiers in Public Health, 2026
Relevance
- Directly informs understanding of early invasive pulmonary aspergillosis in severe viral pneumonia.
- Extends COVID-associated pulmonary aspergillosis concepts to non-COVID viral infections.
Key points
- Viral pneumonia causes early immune dysregulation, including lymphopenia.
- Invasive aspergillosis may develop before classic intensive care risk factors.
- Supports earlier fungal surveillance rather than late rescue testing.
Pulmonary cavitation as a late and self-limited complication of COVID-19 pneumonia
Osório M, Silveira M, Cureus, 2026
Relevance
- Highlights post-viral structural lung damage as a substrate for aspergillosis.
Key points
- Cavitation discussed alongside COVID-associated pulmonary aspergillosis and mucormycosis.
- Fungal risk may persist after apparent clinical recovery.
2. Allergic disease and ABPA – expanding phenotypes
Triple autoimmune overlap: rheumatoid arthritis, systemic lupus erythematosus, and hypereosinophilic asthma with ABPA features
Frontiers in Immunology, 2026 (Case Report)
Relevance
- Challenges rigid diagnostic frameworks for Allergic Bronchopulmonary Aspergillosis.
- Supports emerging views that ABPA can occur before bronchiectasis develops.
Key points
- ABPA considered despite normal chest imaging.
- Diagnosis driven by immunological and eosinophilic markers.
Diagnosis of bronchopulmonary candidiasis—refractory airway hyperresponsiveness and severe pneumonia
Zhang D et al., Frontiers in Medicine, 2026
Relevance
- Important differential diagnosis for suspected ABPA.
Key points
- Bronchopulmonary candidiasis can closely mimic ABPA.
- Normal Aspergillus serology does not exclude other fungal airway disease.
3. Rare immune defects and aspergillosis
Complete and partial forms of X-linked MCTS1 deficiency in patients with mycobacterial disease
Zhou Q et al., Journal of Human Immunity, 2026
Relevance
- Expands the list of primary immunodeficiencies associated with Aspergillus infection.
Key points
- Central nervous system aspergillosis identified as a rare but severe phenotype.
- Suggests impaired cellular immunity as the underlying mechanism.
4. Antifungal therapy – toxicity, variability, and paediatrics
Voriconazole-associated peripheral polyneuropathy: A case report
González BJ et al., Archives of Argentine Pediatrics, 2026
(No PMC full text currently available)
Relevance
- Highlights clinically important non-hepatic toxicity of azole therapy.
Key points
- Peripheral neuropathy developed during voriconazole treatment.
- Symptoms may be insidious and progressive.
RE: Factors affecting voriconazole pharmacokinetic variability in critically ill patients
Langbeen J et al., Critical Care, 2026
Relevance
- Explains why fixed dosing of voriconazole is often unsafe.
Key points
- Critical illness alters drug metabolism and clearance.
- Drug–drug interactions are common.
- Supports therapeutic drug monitoring and specialist pharmacy input.
Phase 2 clinical trial of posaconazole in paediatric invasive aspergillosis
Kang HJ et al., Antimicrobial Agents and Chemotherapy, 2026
(No PMC full text currently available)
Relevance
- Rare prospective antifungal data in children.
Key points
- Posaconazole showed acceptable safety.
- Clinical responses were encouraging in a high-risk population.
5. Diagnostics, microbiology, and co-infection
Clinical characteristics, molecular diagnosis, and drug resistance profiles of nontuberculous mycobacteria infections
Wang K et al., Clinical and Translational Science, 2026
Relevance
- Highly relevant to bronchiectasis patients where NTM and aspergillosis frequently coexist.
Key points
- Molecular diagnostics improve species identification.
- Resistance patterns complicate treatment strategies.
Impaired systemic antibody response against gut microbiota pathobionts in critical illness
Cho NA et al., Intensive Care Medicine Experimental, 2026
Relevance
- Links immune–microbiome disruption to susceptibility to Aspergillus fumigatus.
Key points
- Critical illness impairs antibody responses.
- Loss of immune balance increases infection risk.
6. Pathogenesis and basic science
Arp2/3 complex contributes to actin-dependent uptake of Aspergillus terreus conidia
Mach N et al., PLOS One, 2026
Relevance
- Improves understanding of early host–fungus interactions.
Key points
- Epithelial cells actively internalise Aspergillus conidia.
- Species differences may influence pathogenicity.
7. Environmental and One Health perspectives
Seasonal variation in Aspergillus abundance in captive penguin burrow sands
Takanobu S et al., Frontiers in Veterinary Science, 2026
Relevance
- Demonstrates dynamic environmental exposure risk.
Key points
- Clear seasonal peaks in Aspergillus burden.
- Correlates with increased disease risk.
Mycotoxins – biomonitoring method including gliotoxin
Berger M et al., MAK Collection for Occupational Health and Safety, 2026
Relevance
- Gliotoxin explored as a potential biomarker for invasive aspergillosis.
Key points
- LC-MS/MS methods validated.
- Currently research-grade rather than clinical.
Wearable devices and aspergillosis
Are they useful yet – and which ones are the most accurate?
The short answer
Wearable devices do not diagnose aspergillosis and cannot tell what is causing symptoms.
However, some wearables are now good enough to provide useful background information about how your body is coping over time.
Their value lies in:
-
spotting gradual deterioration
-
recognising patterns over weeks or months
-
supporting conversations with your clinical team
They are not a replacement for scans, blood tests, sputum cultures, lung function tests, or specialist review.
What wearables can realistically help with
For people with:
-
Chronic Pulmonary Aspergillosis (CPA)
-
Allergic Bronchopulmonary Aspergillosis (ABPA)
-
Aspergillus bronchitis
-
Aspergillosis with bronchiectasis or asthma
wearables can sometimes help answer:
-
“Am I slowly getting worse, or is this just a bad patch?”
-
“Has my recovery from exertion changed?”
-
“Are my nights becoming more disrupted?”
They are most useful for long-term trends, not day-to-day decisions.
The signals that matter most
From both patient experience and respiratory clinical practice, these signals tend to be most meaningful:
1. Activity tolerance
-
Falling step count over weeks
-
Needing longer to recover after usual activity
-
Avoiding activity you previously managed
➡ Often one of the earliest signs of deterioration.
2. Resting heart rate
-
A persistent rise from your own baseline
-
Especially if not explained by infection, fever, medication or stress
➡ Often reflects physiological strain before symptoms become obvious.
3. Sleep quality
-
Frequent night waking
-
Shortened or fragmented sleep
-
Feeling unrefreshed despite enough hours in bed
➡ Poor sleep often accompanies worsening respiratory symptoms or medication effects.
4. Oxygen saturation (SpO₂) trends
-
Repeated low readings
-
Drops overnight or with exertion
-
Patterns that persist over days or weeks
➡ Trends matter far more than single readings.
➡ Dedicated oxygen monitors are usually more reliable than watches.
What about ECG and breathing rate?
These features are often misunderstood. They are not useless, but they are supportive rather than central in aspergillosis care.
ECG (heart rhythm)
Some wearables can record a single-lead ECG, which may detect:
-
atrial fibrillation
-
sustained rhythm abnormalities
This can be helpful if someone develops:
-
new palpitations
-
breathlessness out of proportion to lung symptoms
-
dizziness or faintness
➡ ECG does not provide information about Aspergillus activity or lung disease progression.
Breathing (respiratory) rate
Most wearables estimate breathing rate indirectly, usually during sleep.
Breathing-rate trends may:
-
support a sense that breathing effort has increased
-
highlight disrupted sleep linked to respiratory load
➡ It cannot distinguish fungal disease from asthma, infection, anxiety or medication effects.
Medication and age matter — a lot
When clinicians interpret wearable data, they always consider:
-
antifungal medicines (e.g. azoles)
-
steroids (current or past)
-
asthma and allergy treatments
-
other long-term conditions
-
age-related physiological change
Common medication effects seen on wearables
-
higher resting heart rate
-
poorer sleep
-
fatigue
-
reduced activity tolerance
These are common and expected and do not automatically mean disease progression.
As we age:
-
recovery slows
-
sleep becomes lighter
-
heart-rate variability reduces
-
oxygen dips more easily overnight
➡ Always compare data to your own baseline, not to “normal” values.
Environment and everyday factors strongly affect readings
Often more than lung disease itself
This makes them highly sensitive to environment and daily circumstances.
Many “abnormal” readings reflect conditions around you, not worsening aspergillosis.
Temperature (especially cold)
Cold causes blood vessels in the skin to narrow, which can lead to:
-
falsely low oxygen readings
-
erratic heart-rate data
-
missing or failed measurements
Common situations:
-
cold bedrooms
-
winter walks
-
sleeping with arms outside the duvet
➡ A low oxygen reading in the cold is often technical, not medical.
Altitude and air pressure
At higher altitude (even modest):
-
oxygen saturation normally falls
-
breathing rate may rise
-
sleep may worsen
Examples:
-
flying
-
holidays in hilly or mountainous areas
-
high-rise accommodation
➡ This is normal physiology, not disease progression.
Air quality, humidity and heat
Poor air quality or high humidity can cause:
-
faster breathing
-
increased heart rate
-
worse sleep
-
reduced activity tolerance
➡ Wearables detect body stress, not its cause.
Sleep environment
Sleep data is very sensitive to:
-
noise
-
light
-
room temperature
-
uncomfortable bedding
A poor sleep score often reflects environmental disruption, not lung decline.
Movement, posture and coughing
Night-time data can be affected by:
-
coughing
-
restless sleep
-
sleeping on the arm wearing the device
➡ Night data is often noisy and imperfect.
Hydration, alcohol and meals
-
dehydration → higher heart rate
-
alcohol → worse sleep and altered breathing rate
-
heavy evening meals → raised heart rate
These effects are temporary and not signs of deterioration.
Stress and anxiety
Stress can:
-
raise heart rate
-
increase breathing rate
-
worsen sleep
Wearables cannot distinguish stress from illness, and worrying about readings can make readings worse — a common feedback loop.
What wearables cannot do (important)
-
They cannot diagnose aspergillosis
-
They cannot identify fungal flares
-
They cannot separate cause from effect
-
They cannot replace specialist investigations
They provide context, not answers.
The most accurate consumer devices (2025–26)
Best overall smartwatches
-
Apple Watch (Series 9 / Ultra 2) – excellent heart-rate accuracy, ECG, good sleep trends
-
Withings ScanWatch 2 – health-focused, ECG and oxygen, long battery life
-
Garmin Venu 3 / Epix Pro – excellent activity and recovery tracking
Best non-watch wearables
-
Oura Ring (Gen 3) – strong overnight physiology and sleep trends
-
Wellue O2Ring / similar continuous oximeters – more reliable oxygen trends than watches
These are listed because of better accuracy and consistency, not because they are diagnostic devices.
Can wearable data cause over-worry?
Yes — and this is common, especially in people with long-term lung disease.
Wearables can sometimes:
-
increase anxiety
-
encourage constant checking
-
turn normal variation into worry
-
make people feel unwell even when stable
This is not a personal weakness.
When wearables help
-
checked occasionally
-
viewed over weeks or months
-
used to support (not replace) symptoms
When wearables stop helping
-
if they increase anxiety
-
if they disrupt sleep
-
if numbers override how you feel
➡ It is entirely reasonable to reduce use or stop.
How specialists actually prioritise information
In real aspergillosis care, clinicians still focus on:
-
How you feel
-
What you can do
-
Symptoms and sputum
-
Imaging and tests
-
Medication history
Wearable data sits well below these.
The bottom line
-
✔ Wearables are becoming useful for monitoring trends
-
✔ ECG and breathing rate add context and safety, not answers
-
✔ Medication, age and environment strongly affect readings
-
❌ Wearables do not diagnose aspergillosis
-
✔ If a device increases anxiety, stepping back is sensible
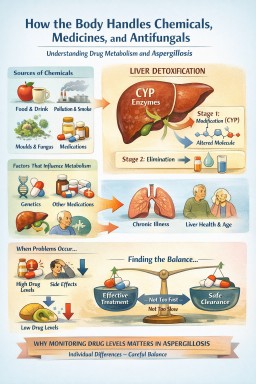
How the Body Handles Chemicals, Medicines, and Antifungals
Why metabolism differs between people — and why this matters in aspergillosis
The big idea (in one sentence)
Your body uses an ancient liver detox system to handle chemicals from food, air, and medicines — and differences in that system explain why people with aspergillosis respond so differently to antifungal drugs.
What is metabolism?
Every day, your body is exposed to chemicals from many sources:
-
Food and drink
-
Air pollution and moulds
-
Natural plant chemicals
-
Hormones your body makes itself
-
Medicines, including antifungals and steroids
Many of these chemicals cannot be safely removed in their original form.
They first need to be chemically modified so they can be excreted in urine or bile.
This process is called metabolism, and it happens mainly in the liver.
The liver’s chemical processing system
The liver contains a large family of enzymes called cytochrome P450, often shortened to CYP.
Important clarification
CPA = Chronic Pulmonary Aspergillosis (a lung disease)
CYP = Cytochrome P450 (liver enzymes)
They sound similar but are completely different things.
What CYP enzymes really do
CYP enzymes did not evolve to deal with medicines.
They evolved to protect us from chemicals in the environment.
They help process:
-
Plant toxins and food chemicals
-
Smoke and air pollution
-
Mould and fungal by-products
-
Alcohol and caffeine
-
Hormones such as cortisol and sex hormones
-
Medicines (which are treated as “foreign chemicals”)
Medicines simply use a system that already existed.
How CYP enzymes “recognise” chemicals
CYP enzymes do not recognise chemicals like the immune system recognises germs.
Instead, they recognise chemical patterns, such as:
-
Fat-solubility (hard to excrete)
-
Size and shape
-
Reactive chemical groups
If a molecule:
-
Fits into the enzyme’s binding pocket, and
-
Can be chemically modified,
then CYP will act on it.
This makes CYP enzymes:
-
Broad (they work on many substances)
-
Flexible
-
Imperfect by design
The two main stages of metabolism (simplified)
Stage 1 – Modification
-
Mainly done by CYP enzymes
-
The chemical is altered (often oxidised)
-
This may:
-
Reduce activity
-
Prepare it for removal
-
Occasionally create a more toxic intermediate
-
Stage 2 – Packaging for removal
-
The altered chemical is “tagged”
-
It becomes water-soluble
-
It can now leave the body safely
Why metabolism differs between people
This is especially important for aspergillosis patients.
1. Genetics (the biggest factor)
People inherit different versions of CYP enzymes.
Some people:
-
Break drugs down slowly → higher levels → side effects
-
Break drugs down quickly → low levels → reduced effectiveness
Two people on the same antifungal dose can have very different blood levels.
2. Other medicines
Some medicines:
-
Block CYP enzymes (slowing breakdown)
-
Speed up CYP enzymes (lowering drug levels)
Antifungals, steroids, antibiotics, antidepressants, and heart drugs often interact.
3. Inflammation and chronic illness
During infection or chronic inflammation:
-
CYP activity is often reduced
-
Drug levels may rise unexpectedly
This matters in:
-
Chronic Pulmonary Aspergillosis (CPA)
-
Allergic Bronchopulmonary Aspergillosis (ABPA)
-
Bronchiectasis
-
Severe asthma
Drug handling can change during disease flares.
4. Liver health and age
-
Liver disease can slow metabolism
-
Older adults often process drugs differently
Why something can become a “poison”
A substance can cause harm if it:
-
Escapes CYP processing
-
Is metabolised too slowly
-
Overwhelms the system at high dose
-
Blocks CYP so other substances build up
-
Is converted into a toxic by-product
This explains:
-
Why some foods are toxic to dogs but safe for humans
-
Why “natural” substances are not automatically safe
-
Why dose really matters
A key question:
Why not design medicines that CYP can’t break down?
This is a real goal in drug development, and your instinct is correct.
If a drug:
-
Is broken down very slowly, or
-
Avoids CYP metabolism altogether,
then:
-
It stays in the body longer
-
Blood levels are steadier
-
Fewer doses are needed
This is why some medicines are once-daily, once-weekly, or long-acting injections.
But there is a trade-off
CYP metabolism is not just an inconvenience — it is also a safety system.
If a drug:
-
Cannot be metabolised, and
-
Cannot be excreted easily,
then:
-
It may accumulate
-
Side effects last much longer
-
Toxicity is harder to reverse
-
Stopping the drug does not stop the problem quickly
So completely avoiding CYP can increase long-term risk, especially when medicines are taken for months or years.
How drug designers manage this balance
Most modern drugs aim for a middle ground:
-
Broken down slowly, not zero
-
More predictable metabolism
-
Fewer interactions with major CYP enzymes
-
Alternative clearance routes where possible
-
Long-acting formulations (slow release, depots) rather than permanent persistence
In other words:
Long enough to work — but short enough to stay safe
Why this is especially relevant in aspergillosis
Antifungal drugs are particularly challenging because:
-
Fungi are biologically similar to humans
-
Drugs often interact with human CYP enzymes
-
Treatment is long-term
-
Patients often take multiple other medicines
Because of this:
-
Blood level monitoring is common
-
Dose adjustments are expected
-
Side effects do not mean failure
-
Low levels do not mean non-compliance
This variability reflects normal biology, not poor care.
A simple way to think about it
-
Your liver is a chemical processing plant
-
CYP enzymes are general-purpose machines
-
Everyone’s machines run at slightly different speeds
-
Illness and other drugs change how they behave
-
Antifungals depend on these machines being “just right”
Key take-home messages for patients
-
CYP enzymes are part of your body’s everyday detox system
-
They evolved to handle food chemicals, pollution, moulds, and hormones
-
Medicines use the same system
-
People differ because of genetics, illness, and other drugs
-
In aspergillosis, variable drug levels are expected
-
Monitoring and dose adjustment are signs of good specialist care
-
Drugs are not designed to avoid metabolism completely — safety matters as much as convenience