Real Questions. Clear Answers. Practical Support for People Living with Aspergillosis.
If you live with aspergillosis, or support someone who does, you will know how many questions can come up between appointments. Some are about symptoms. Some are about treatment. Some are about day-to-day life, side effects, monitoring, damp, mould, travel, fatigue, anxiety, or simply trying to understand what is happening.
That is why we have built and continue to expand our Patient Questions & Latest Articles section.
This part of our website brings together detailed answers to the kinds of questions patients and carers actually ask. Many of these articles began as real questions raised in our support meetings and online communities, then developed into fuller explanations so that others could benefit too.
Why this section is useful
Living with aspergillosis often means having to manage a lot of uncertainty. Information can be difficult to find, hard to interpret, or scattered across different places. We wanted to create a space that is easier to use: a place where people can browse practical topics, explore recent articles, and find trustworthy explanations written with patients and carers in mind.
Some people arrive with one urgent question. Others simply want to browse and see whether someone else has already asked the same thing. This section is designed to help with both.
What you will find there
- Answers to common patient and carer questions
- Detailed articles on symptoms, treatments, tests, monitoring, and side effects
- Practical explainers on issues such as damp, mould, air quality, medicines, and coping day to day
- Recent updates and newly published articles in one place
- Links into wider resources across our website
You do not need to read everything from start to finish. You can dip in when a particular question comes up, browse recent topics, or use the search function to look for something specific.
Built around real concerns
One of the strengths of this section is that it is grounded in real experience. The topics are not chosen at random. They reflect the concerns patients and carers bring to us repeatedly: the things people worry about, struggle to understand, or need explained more clearly.
That means the content is often practical, specific, and directly relevant to everyday life with aspergillosis.
Part of a wider knowledge base
If you want to browse recent questions and articles, this section is a very good place to start. If you are looking for more structured background information on aspergillosis, symptoms, tests, treatments, and monitoring, you can also move on from there to our wider Knowledge Hub.
Together, these resources are intended to make it easier for patients, families, and carers to find the information they need, when they need it.
Please use health information safely
As always, our website is here to support understanding, not replace medical advice from your own clinical team. We hope these articles help people feel better informed, better prepared, and better able to ask the right questions, but personal medical decisions should still be made with the professionals looking after your care.
Take a look
Explore the full section here:
Patient Questions & Latest Articles
If you think others may find it helpful, please feel free to share it.
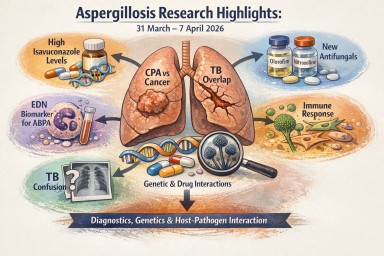
Weekly Aspergillosis Research Update: 31 March – 7 April 2026
This week’s research reinforces several consistent themes in aspergillosis: ongoing diagnostic confusion (particularly with tuberculosis and cancer), increasing movement toward precision medicine, and continued development of both antifungal therapies and biomarkers. There is also a growing emphasis on host-pathogen interactions rather than fungal burden alone.
Key Highlights
- Isavuconazole levels can become unexpectedly high due to genetics and drug interactions.
- Chronic pulmonary aspergillosis (CPA) can mimic lung cancer, risking delayed diagnosis.
- New biomarker (EDN) for ABPA shows promise for diagnosis and monitoring.
- Azole resistance research highlights differences between Aspergillus species.
- New antifungal approaches emerging (olorofim, nitroxoline).
- Host response is central – fibroblasts and immune pathways actively influence disease.
- TB vs aspergillosis confusion persists in real-world settings.
Contents
- Clinical & Diagnostic Studies
- Treatment & Pharmacology
- Biology, Immunology & Resistance
- Biomarkers & Diagnostics
- Wider Context
Clinical & Diagnostic Studies
CPA mistaken for lung cancer
Paper: PubMed
A case report describes chronic pulmonary aspergillosis presenting as suspected lung malignancy in a patient with asthma and ABPA overlap.
Why this matters: CPA continues to be misdiagnosed due to tumour-like imaging appearances. This reinforces the need to consider fungal disease in patients with underlying lung conditions.
Invasive sinus aspergillosis causing bone destruction
Paper: Free full text
Granulomatous invasive aspergillosis led to facial bone destruction and neurological symptoms.
Why this matters: Delayed diagnosis of invasive disease can lead to severe structural damage. Early imaging and specialist input are critical.
Aspergillus infection in suspected TB patients
Paper: PubMed
Study shows overlap between tuberculosis and aspergillosis in symptomatic patients.
Why this matters: Persistent global issue—shared symptoms delay correct diagnosis and treatment, particularly relevant for CPA pathways.
Invasive aspergillosis in critical illness
Paper: PubMed
Case of invasive pulmonary aspergillosis in a patient with severe viral illness.
Why this matters: Reinforces that aspergillosis is not limited to traditional risk groups and can complicate severe systemic illness.
Treatment & Pharmacology
Isavuconazole toxicity linked to genetics
Paper: Free full text
Case report of supratherapeutic isavuconazole levels linked to CYP3A5 genotype and interacting medications.
Why this matters: Even “predictable” antifungals show variability. Supports therapeutic drug monitoring and future personalised dosing approaches.
Olorofim pharmacokinetics
Paper: PubMed
Study demonstrates tissue distribution of olorofim in preclinical models.
Why this matters: Supports ongoing development of a key next-generation antifungal, particularly for resistant disease.
Nitroxoline shows antifungal activity
Paper: PubMed
Repurposed drug demonstrates activity against Aspergillus via copper disruption and oxidative stress.
Why this matters: Highlights potential for non-azole antifungal strategies in future treatment.
Biology, Immunology & Resistance
Azole resistance and Aspergillus genomics
Paper: PubMed
Genomic study of Aspergillus section Fumigati explores resistance mechanisms and pathogenicity.
Why this matters: Different species may respond differently to antifungals—accurate identification is increasingly important.
Fibroblasts actively support lung defence
Paper: PubMed
Study shows fibroblasts contribute to immune defence and tissue repair during infection.
Why this matters: Disease outcomes depend on host response, not just fungal burden—important for future therapies.
Immune pathway targeting in fungal keratitis
Paper: PubMed
PIM1 inhibition reduces inflammation via STING pathway signalling.
Why this matters: Supports growing interest in targeting immune pathways alongside antifungal therapy.
Biomarkers & Diagnostics
Eosinophil-derived neurotoxin (EDN) in ABPA
Paper: PubMed
EDN proposed as a biomarker for allergic bronchopulmonary aspergillosis.
Why this matters: Could improve diagnosis and monitoring, helping distinguish ABPA from asthma or sensitisation alone.
Commentary on ISHAM ABPA guidelines
Paper: PubMed
Discussion of updated international guidance on ABPA diagnosis and management.
Why this matters: Highlights ongoing refinement of diagnostic criteria and classification systems.
Wider Context
Fungal extracellular vesicles
Paper: PubMed
Review of fungal vesicles in pathogenesis and host interaction.
Why this matters: Emerging area that may influence future diagnostics and therapies.
Aspergillosis in broader disease settings
Why this matters: Aspergillosis continues to appear across a wide range of conditions, particularly in critically ill or immunocompromised patients.
Overall Interpretation
This week’s literature reinforces several strategic priorities:
- Earlier and more accurate diagnosis remains essential, particularly in distinguishing CPA from TB and cancer.
- Precision medicine is advancing, with growing roles for pharmacogenomics, drug monitoring, and species-level identification.
- New antifungal options are progressing, but remain largely in development.
- Host response is increasingly recognised as central to disease progression and outcomes.
Overall, the field continues to move toward more personalised, biology-driven approaches to diagnosis and management.
Mould, “Toxins” and Aspergillus: Understanding What’s True and What Isn’t
Last reviewed: April 2026
Key points
- Exposure to mould is not the same as having aspergillosis.
- Aspergillus is a common environmental fungus that most people inhale daily.
- “Mycotoxin” and “detox” explanations are common online but are not part of standard diagnosis.
- Symptoms usually arise from a combination of lung disease, infection, inflammation, and environment.
- Diagnosis relies on clinical tests (scans, blood tests, microbiology), not environmental testing alone.
- Specialist home testing is sometimes used—but only in specific clinical situations.
A guide for patients and carers
Many people with lung conditions come across information linking mould exposure, toxins, and Aspergillus infections. Some of this information can be helpful—but much of it can also be confusing.
This article explains the most common misunderstandings and what current medical practice actually relies on.
Important: Some tests measure exposure—but medical diagnosis requires evidence of disease.
Contents
- Why this topic is confusing
- Mould exposure vs Aspergillus disease
- Mycotoxins and “detox” claims
- Why “mould toxin tests” are not used diagnostically
- Why symptoms don’t have one single cause
- How Aspergillus conditions are actually diagnosed
- When home testing may be used
- What matters most for patients
Why this topic is confusing
- Mould is visible and easy to focus on
- Symptoms (fatigue, cough, breathlessness) are non-specific
- Online information often mixes different conditions together
This can lead to the idea that one single cause—such as “mould toxicity”—explains everything. In practice, respiratory conditions are usually more complex.
Mould exposure vs Aspergillus disease
Mould exposure
- Common in homes and outdoor air
- Can irritate airways
- May worsen symptoms
Aspergillus disease
- Occurs in specific medical contexts
- Related to lung structure and immune response
- Requires clinical diagnosis
Important context: Aspergillus spores are present in the air we breathe every day. Most people inhale them without developing disease.
Mycotoxins and “detox” claims
You may see references to:
- “Mycotoxin illness”
- “Die-off reactions”
- “Binders” to remove toxins
These ideas are widely discussed online, but they are not part of standard medical diagnosis or treatment for Aspergillus lung conditions.
This does not mean symptoms are not real. It means they are usually explained through:
- Inflammation
- Infection
- Underlying lung disease
Why “mould toxin tests” are not used diagnostically
Exposure is common
Many people have detectable environmental exposure, so results are not specific.
Results do not match symptoms reliably
Levels do not consistently correlate with disease or severity.
Lack of standardisation
Different labs use different methods, making interpretation difficult.
Not part of clinical guidelines
Major respiratory guidelines do not include these tests in diagnosing Aspergillus conditions.
Risk of confusion
- May cause unnecessary concern
- May lead to unproven treatments
Why symptoms don’t have one single cause
Symptoms often arise from a combination of:
- Underlying lung disease
- Inflammation
- Infection (fungal or bacterial)
- Environmental triggers (pollution, damp)
This is why symptoms can fluctuate and may not respond to a single explanation or treatment.
How Aspergillus conditions are actually diagnosed
Diagnosis is based on a combination of:
- CT imaging
- Blood tests (e.g. IgE, IgG)
- Microbiology (sputum or bronchoscopy)
Environmental testing alone is not sufficient to diagnose disease.
When home testing may be used
In some cases, specialist teams (for example via the Mycology Reference Centre Manchester) may arrange targeted environmental sampling.
This is different from commercial testing and focuses on:
- Specific clinically relevant fungi (e.g. Aspergillus species)
- Known allergens or pathogens
Why it is done
- Established Aspergillus-related disease
- Unexplained or persistent symptoms
- Concern about ongoing exposure
This testing is used to answer specific clinical questions and is interpreted alongside medical findings.
What matters most for patients
- Work with your clinical team for diagnosis and treatment
- Address damp and mould in your home
- Focus on practical steps that improve symptoms
- Avoid relying on a single explanation for complex symptoms
Final takeaway
Mould, environment, and Aspergillus are connected—but not in a simple cause-and-effect way.
Understanding this helps you focus on what is most likely to improve your health: appropriate treatment, good living conditions, and ongoing monitoring.
Author: aspergillosis.org
Note: Educational content only – not medical advice.
Indoor Air Quality, Damp, Mould and Aspergillus
Last reviewed: April 2026
Key points
- Indoor air problems usually relate to pollution, damp, or mould—these are different but can overlap.
- Aspergillus is a normal part of the environment and not automatically a sign of a problem indoors.
- Damp and mould can worsen breathing symptoms and should be taken seriously.
- Fixing the underlying moisture problem is more important than cleaning visible mould.
- Knowing who to contact (GP, landlord, council) is key to resolving problems.
Indoor Air Quality, Damp, Mould and Aspergillus
A practical guide for patients
Many people with aspergillosis or other lung conditions are concerned about the air inside their home. This article explains how indoor air quality works, how to recognise problems, and what steps you can take to improve your environment.
Contents
- What affects indoor air?
- Three common problems
- Aspergillus in the home
- Damp and mould
- Symptoms and health effects
- Identifying problems
- Getting the right help
- Practical steps
- Air purifiers
What affects indoor air?
Indoor air quality is influenced by both indoor and outdoor factors. People spend most of their time indoors, so even small issues can have a noticeable effect on health. :contentReference[oaicite:0]{index=0}
Indoor air can be affected by:
- Outdoor pollution entering the building
- Damp and moisture
- Mould growth
- Cooking, heating, and cleaning products
- Dust and particles
Three common indoor air problems
1. Indoor air pollution
- Particles from cooking, heating, and candles
- Outdoor pollution enters indoors
2. Damp (moisture)
- Condensation, leaks, poor ventilation
- Creates conditions for mould growth
3. Mould
- Visible fungal growth on surfaces
- Releases spores into the air
Aspergillus in the home
Aspergillus is a common environmental fungus found in air, dust, and soil.
This means:
- Everyone breathes in Aspergillus spores regularly
- Indoor exposure is not unusual
Important: Exposure does not equal disease. Aspergillosis depends on lung health and immune response—not just environment.
Damp and mould
Damp and mould are important because they can affect respiratory health. Damp homes are linked with increased respiratory symptoms and illness. :contentReference[oaicite:1]{index=1}
UK guidance highlights that damp and mould can produce irritants and spores that affect the lungs and should be addressed promptly. :contentReference[oaicite:2]{index=2}
Symptoms and health effects
Indoor air problems may contribute to:
- Cough
- Breathlessness
- Wheeze
- Chest tightness
- Fatigue
These symptoms often overlap with infection or underlying disease, making them difficult to interpret.
Identifying problems
Signs of damp
- Condensation on windows
- Cold or damp walls
- Peeling paint or wallpaper
- Musty smell
Signs of mould
- Black or green patches
- Mould returning after cleaning
- Growth behind furniture or in corners
Hidden issues
- Persistent damp smell
- Symptoms are worse in certain rooms
Getting the right help
1. Your healthcare team
- If symptoms worsen
- If you suspect your environment is affecting your health
2. Landlord or housing provider
- Report problems early
- Keep records (photos, dates)
UK guidance makes clear that damp and mould should be addressed promptly and not ignored.
3. Local council
- If landlord does not act
- If health is affected
Further reading
See more detailed guidance and support resources here:
Housing, damp and mould articles (aspergillosis.org)
Practical steps
- Improve ventilation (especially kitchens and bathrooms)
- Reduce moisture
- Address leaks quickly
- Clean small mould areas safely
Air purifiers
Air purifiers can reduce airborne particles, but:
- They do not fix damp
- They do not remove mould from surfaces
- They do not treat aspergillosis
Final takeaway
Indoor air problems are common and often manageable. The key is to:
- Recognise the signs early
- Address moisture and mould properly
- Seek help when needed
Author: aspergillosis.org
Note: This article is for general education and does not replace medical advice.
Outdoor Air Pollution and Aspergillosis
Last reviewed: April 2026
Key points
- Harmful air pollution can be visible or invisible.
- The Air Quality Index (AQI) is more reliable than how the air looks.
- Air pollution comes from natural, industrial, and everyday urban sources.
- Staying indoors can reduce exposure, but it does not remove it completely.
- People with aspergillosis, asthma, or bronchiectasis may be more sensitive to polluted air.
- Simple steps such as checking AQI, closing windows, and using filtration can help reduce exposure.
A practical guide for patients with lung conditions
Air pollution can make breathing symptoms worse for many people, especially those with existing lung disease. If you live with aspergillosis, asthma, bronchiectasis, chronic obstructive pulmonary disease (COPD), or other long-term lung problems, outdoor air quality can make a noticeable difference to how you feel day to day.
This guide explains what outdoor air pollution is, where it comes from, how to judge risk, what practical steps may help reduce exposure, and how to think sensibly about buying an air purifier.
Contents
- What is outdoor air pollution?
- Where does it come from?
- Visible or invisible pollution
- Understanding AQI (Air Quality Index)
- Where to check AQI
- Why air pollution matters in aspergillosis
- Dust storms, haze and extreme events
- How much protection does staying indoors give?
- Practical ways to reduce exposure
- Masks and air filtration
- Buying an air purifier: what matters and what does not
- Travel and changing environments
- Common misconceptions
- Managing your condition during high air pollution
- When to seek medical help
- References
What is outdoor air pollution?
Outdoor air pollution is a mixture of particles and gases in the air that can irritate the lungs and worsen breathing symptoms.
The main pollutants discussed in health guidance include:
- PM2.5 – very fine particles small enough to travel deep into the lungs
- PM10 – slightly larger particles that can still irritate the airways
- Nitrogen dioxide (NO2) – often linked to traffic and combustion
- Ozone (O3) – a gas that can irritate the lungs, especially in hot weather
- Smoke, dust and soot – depending on local conditions
Some of these pollutants are easy to see, but some are not. This is one reason why it is important not to rely only on appearance when judging air quality.
Where does it come from?
Outdoor air pollution usually comes from a combination of sources, not just one.
Natural sources
- Desert dust or sandstorms
- Wildfire smoke
- Wind-blown soil and dust
- Pollen (not usually included in AQI in the same way, but still relevant for symptoms)
Commercial and industrial sources
- Factories
- Power stations
- Shipping emissions
- Aviation emissions
- Construction and demolition dust
Domestic and urban sources
- Road traffic
- Heating systems
- Wood burners and solid fuel burning
- Cooking emissions in densely populated areas
In some situations, one source clearly dominates. For example, a severe dust event in North Africa or the Middle East may be mainly caused by natural desert dust. In a busy city, day-to-day pollution may be more strongly linked to traffic, heating, and industry. In many real-life situations, however, several sources are contributing at the same time.
Visible or invisible pollution
One of the most important things for patients to know is that harmful air pollution can be visible or invisible.
Visible pollution
- Dust or sand in the air
- Smoke
- Grey or brown smog
- Heavy haze
Invisible pollution
- Fine particles such as PM2.5
- Gases such as ozone or nitrogen dioxide
Clear sky does not always mean clean air. Equally, hazy air is not always the only dangerous situation. Some invisible pollutants can be especially harmful because they are easy to miss and can still reach deep into the lungs.
Key message: If you can see it, it may be harmful. If you cannot see it, it still may be harmful.
Understanding AQI (Air Quality Index)
The Air Quality Index (AQI) is often the most useful practical tool for patients. It gives a simple way of describing how healthy or unhealthy the air is likely to be.
| AQI | Meaning |
|---|---|
| 0–50 | Good |
| 51–100 | Moderate |
| 101–150 | Unhealthy for sensitive groups |
| 151–200 | Unhealthy |
| 201–300 | Very unhealthy |
| 300+ | Hazardous |
Very high readings can occur in severe dust events, smoke events, or heavily polluted urban conditions. For patients with lung disease, even levels well below the highest categories may still trigger symptoms.
Why AQI matters more than appearance
AQI can help you judge risk more reliably than what you can see or smell. Dust may have no obvious smell. Fine particles may be invisible. Air that looks like fog may actually be carrying a heavy particle load. Because of this, it is usually safer to trust the AQI reading rather than appearance alone.
Where to check AQI
Reliable sources for checking air quality include:
- QAir AirVisual
– widely used internationally, with real-time air quality data, forecasts, and health guidance - UK Department for Environment, Food & Rural Affairs (DEFRA) – UK-AIR
– official UK air quality data and health advice - Plume Labs
– useful for air-quality maps and forecasts - Windy
– helpful for visualising dust, smoke and weather patterns
For most people, it is best to use one app consistently, and occasionally cross-check with a second source if readings seem unusual.
Why air pollution matters in aspergillosis
People living with aspergillosis may already have lungs that are inflamed, structurally damaged, or more easily irritated. This may include people with:
- Allergic bronchopulmonary aspergillosis (ABPA)
- Chronic pulmonary aspergillosis (CPA)
- Bronchiectasis
- Asthma
- Chronic obstructive pulmonary disease (COPD)
Air pollution can potentially worsen:
- Cough
- Breathlessness
- Chest tightness
- Wheeze
- Mucus production
- General irritation of the airways
It can also make it harder to work out what is causing symptoms. A flare in symptoms is not always due to infection. Sometimes symptoms may be made worse by air pollution, airway irritation, or inflammation, even when there is no new infection.
Dust storms, haze and extreme events
In some parts of the world, especially in North Africa and the Middle East, very high AQI readings are often caused by dust and sand in the air. This can sometimes be mistaken for fog.
Clues that this may be dust rather than fog include:
- Very high AQI readings
- Dry-looking haze rather than damp mist
- Dust collecting on surfaces
- Weather reports mentioning dust, sand, or reduced visibility
Dust does not always have a noticeable smell, so the absence of smell does not mean the air is safe.
How much protection does staying indoors give?
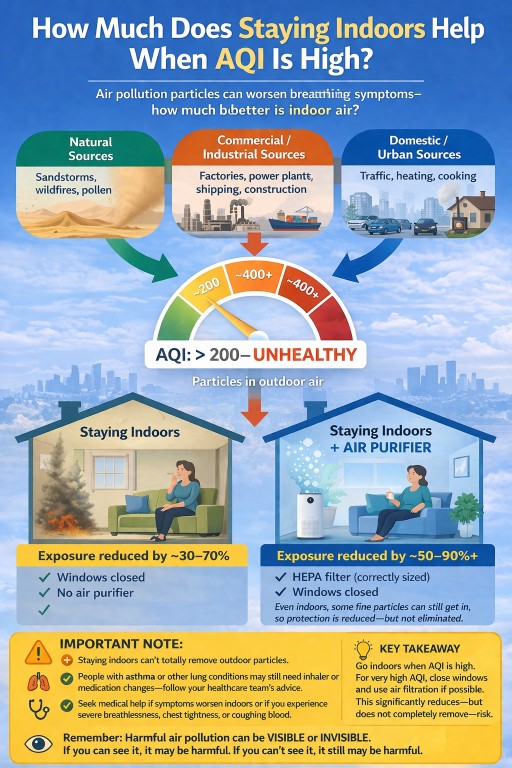
Staying indoors usually does help, but the amount of protection varies.
Typical home with windows closed
In general terms, staying indoors with windows closed may reduce exposure by around 30% to 70%, depending on:
- How well sealed the building is
- Whether windows or doors are being opened
- The age and condition of the property
- How long the pollution event lasts
Indoors with HEPA filtration
Using a correctly sized High Efficiency Particulate Air (HEPA) purifier can improve indoor air further. In some situations, this may reduce indoor particle exposure by around 50% to 90% or more.
Important limits
- Fine particles can still get indoors
- Older or drafty buildings may offer less protection
- Opening windows can quickly increase indoor particle levels again
The realistic goal is usually to reduce exposure as much as possible, not to expect complete protection.
Practical ways to reduce exposure
When outdoor AQI is high, the following steps may help:
- Stay indoors as much as possible
- Keep windows and doors closed
- Avoid strenuous outdoor activity
- Use air filtration if you have it
- Delay non-essential trips outdoors if practical
- Check AQI more than once per day during unstable conditions
For some people, symptoms may still occur even indoors. If so, it may help to reduce physical activity, keep rescue medication available if prescribed, and monitor symptoms closely.
Masks and air filtration
Masks
Well-fitted particle-filtering masks such as FFP2 or FFP3 may reduce exposure to fine particles when you need to go outdoors. Their usefulness depends heavily on fit, comfort, and wearing them correctly.
Not everyone can tolerate masks easily, especially if they are already short of breath, so they are not always a complete solution.
Air purifiers
A HEPA purifier may be especially useful in the room where you spend the most time, such as a bedroom or living room. Performance depends on:
- Correct room size
- Good maintenance of filters
- Keeping windows closed during high pollution periods
Buying an air purifier: what matters and what does not
Air purifiers can help reduce indoor particle levels, but it is important to be realistic about what they can and cannot do.
Important: An air purifier does not treat aspergillosis, does not cure asthma or bronchiectasis, and does not remove all particles. It is best thought of as one way to reduce exposure.
What matters most
- True HEPA filtration (or a clearly specified high-efficiency particle filter)
- Room-size coverage that matches the room where it will be used
- Clean Air Delivery Rate (CADR), where provided
- Noise level, especially if used in a bedroom
- Replacement filter cost and availability
What these terms mean
True HEPA: This usually means the purifier is designed to remove very small airborne particles efficiently. For patients concerned about dust and polluted air, this is generally more important than extra “smart” features.
Room size: A purifier that is too small for the room may not make much difference. It is usually better to buy the correct size for the room where you spend the most time than to buy a very cheap device that cannot handle the space.
CADR: This is a measure of how quickly a purifier can clean the air. Higher CADR usually means faster particle removal, though room size and real-life use still matter.
What to be cautious about
- Very cheap devices with vague claims but no clear filter specification
- Small USB-powered “desk purifiers” claiming to clean large rooms
- Ioniser-only devices with no proper particulate filter
- Products that make sweeping medical claims
Practical buying advice
- Start with the room you use most, often the bedroom or living room
- Choose a purifier that is rated for that room size
- Check how often filters need replacing and how much replacements cost
- Read the real specifications, not just the marketing headline
- If you are noise-sensitive, look at the sleep-mode or low-speed sound level
Reasonable expectations
Used properly, a good air purifier may make a meaningful difference during high pollution periods, especially when combined with keeping windows closed. However, it is only one part of managing exposure.
Travel and changing environments
Air quality can change dramatically between regions and countries. Some places have persistent urban pollution, while others may experience sudden dust events, wildfire smoke, or seasonal changes.
If you are travelling, it may help to:
- Check local AQI before travel and during your stay
- Be aware of seasonal dust or wildfire risks
- Know where you can spend time indoors if air quality worsens
- Keep regular medications with you
This can be particularly important if you already know that poor air quality tends to worsen your symptoms.
Common misconceptions
Myth: “If I cannot see pollution, it is safe.”
Fact: Invisible pollution such as PM2.5 and gases can still be harmful.
Myth: “If the air looks foggy, it must just be water vapour.”
Fact: Hazy air can sometimes be caused by dust or pollution rather than fog.
Myth: “Staying indoors completely solves the problem.”
Fact: Staying indoors usually reduces exposure, but does not remove it completely.
Myth: “If my symptoms worsen, it must be an infection.”
Fact: Pollution and airway irritation can also worsen symptoms.
Myth: “Air pollution is always caused by traffic.”
Fact: Pollution may come from natural, industrial, and domestic sources, often in combination.
Managing your condition during high air pollution
For people with aspergillosis, asthma, bronchiectasis or other lung conditions, air pollution can trigger symptoms even when there is no infection. Having a simple plan can help you feel more in control.
Continue your regular medication
- Take your prescribed medications as usual, including inhalers or antifungal treatments
- Do not stop or reduce medication without medical advice
Have your reliever medication available
- Carry your reliever inhaler (if prescribed) when leaving the house
- Make sure it is within easy reach, not packed away
- Check that it is in date and not empty
Plan ahead before going outdoors
- Check the AQI before leaving home
- Avoid going out during peak pollution times if possible
- Consider shorter trips or lower-exertion activities
Adjust activity levels
- Reduce strenuous activity when AQI is high
- Walk more slowly and allow extra time
- Take breaks if you feel breathless
Know your early warning signs
Be aware of symptoms that may indicate your lungs are being affected:
- Increased breathlessness
- More coughing or wheezing
- Chest tightness
- Unusual fatigue
Have a simple action plan
- Know what you usually do if symptoms worsen
- Follow any personalised plan provided by your clinical team
- Seek advice if symptoms are different from your usual pattern
Practical tip: On high pollution days, think ahead before leaving home—take medication with you, plan shorter trips, and allow extra time so you do not have to rush.
When to seek medical help
Seek medical advice promptly if you notice:
- Worsening breathlessness
- New or worsening wheeze
- Chest tightness that is not settling
- A major increase in cough
- Coughing blood (haemoptysis)
- Symptoms that are more severe than your usual pattern
If you have a severe flare, marked breathing difficulty, or significant haemoptysis, seek urgent medical help.
Final takeaway
Outdoor air pollution is an important and often under-recognised trigger for people with lung disease. For patients with aspergillosis and related lung conditions, it can worsen symptoms even when there is no new infection.
The most helpful practical points are:
- Use AQI rather than appearance alone
- Remember that pollution may be visible or invisible
- Stay indoors when AQI is high
- Use air filtration where possible
- Keep an eye on changing symptoms and seek help if needed
References
- World Health Organization. WHO Global Air Quality Guidelines. 2021.
- UK Department for Environment, Food & Rural Affairs (DEFRA). UK-AIR: Air Information Resource.
- IQAir AirVisual air quality app.
- European Respiratory Society statements and guidance on air pollution and respiratory health.
- British Thoracic Society resources on respiratory health and environmental exposure.
Author: Prepared for aspergillosis.org
Review note: This article is intended for general education and should not replace advice from your own clinical team.
Voriconazole interactions: what patients need to know
Key points
- Voriconazole interacts with many medicines.
- It affects several liver enzyme pathways, including CYP3A4, CYP2C19, and CYP2C9.
- Its behaviour can vary more from one person to another than some other azoles.
- Some medicines can make voriconazole stronger, while others can make it less effective.
- Visual side effects and sensitivity to sunlight are well recognised with voriconazole.
What is voriconazole?
Voriconazole is an azole antifungal often used in serious fungal infections, including aspergillosis. It can be very effective, but it also has a relatively complex interaction profile.
Why voriconazole interacts with so many medicines
Voriconazole affects several liver enzyme systems, including CYP3A4, CYP2C19, and CYP2C9. It can increase the levels of some medicines, while some other medicines can lower voriconazole levels and reduce its effectiveness.
Because voriconazole metabolism varies between patients, the same combination can affect people differently.
The interaction groups most likely to matter
Steroids
Voriconazole can increase exposure to some steroids, including inhaled steroids, which may increase the risk of steroid side effects.
Medicines that reduce voriconazole levels
Some medicines, including certain anti-seizure medicines and rifampicin-type antibiotics, can reduce voriconazole levels so much that the antifungal may not work properly.
Blood thinners
Some blood thinners may become stronger when taken with voriconazole, increasing bleeding risk.
Heart rhythm medicines
Voriconazole can contribute to QT prolongation, so combinations with other medicines that affect heart rhythm may be particularly important.
Statins
Some statins can become stronger when taken with voriconazole, increasing the risk of muscle side effects.
Immunosuppressants
Medicines such as tacrolimus and ciclosporin can rise significantly with voriconazole and often require specialist monitoring and dose adjustment.
Sedatives and some mental health medicines
Some sedatives and psychiatric medicines can become stronger when combined with voriconazole, increasing the risk of drowsiness, confusion, or other side effects.
Voriconazole-specific issues patients should know
Visual changes
Temporary visual disturbances are well recognised with voriconazole. Patients may notice blurred vision, brighter vision, or changes in colour perception.
Photosensitivity
Voriconazole can increase sensitivity to sunlight. Patients should use sensible sun protection and report new skin changes, especially during long-term treatment.
Variable drug levels
Voriconazole levels can vary between patients, which is one reason some teams use therapeutic drug monitoring in selected situations.
What patients should do in practice
- Tell your clinical team and pharmacist that you are taking voriconazole.
- Check before starting new medicines, including over-the-counter or herbal products.
- Report visual changes, significant sensitivity to sunlight, or a change in symptoms after a medicine change.
- Do not change treatment without advice.
When to seek medical advice
Seek medical advice urgently for severe bleeding, fainting, severe palpitations, marked confusion, or rapid worsening after a medicine change.
Important
This page is educational and does not list every interaction. For a full check, use the BNF interaction checker or speak to a pharmacist or clinician.
References
Itraconazole interactions: what patients need to know
Key points
- Itraconazole can interact with many other medicines.
- It usually does this by affecting liver enzymes, especially CYP3A4.
- Some combinations need careful monitoring, while others are best avoided.
- Capsules and liquid formulations are not handled by the body in exactly the same way.
- This page highlights the interactions most likely to matter to people with aspergillosis.
What is itraconazole?
Itraconazole is an azole antifungal used in aspergillosis and other fungal infections. It has a relatively high interaction burden, so medicine checks are especially important before starting it and whenever another medicine is added, stopped, or changed.
Why itraconazole interacts with so many medicines
Itraconazole can increase the levels of some other medicines by affecting liver enzymes, especially CYP3A4. This means that some medicines may become stronger than intended, which can increase the risk of side effects or toxicity.
The interaction groups most likely to matter
Steroids and inhalers
This is one of the most important groups for many aspergillosis patients, especially those with asthma or allergic bronchopulmonary aspergillosis. Itraconazole can increase exposure to some steroids, including inhaled steroids, which may increase the risk of steroid side effects.
Possible symptoms to report: unusual weight gain, easy bruising, increased facial rounding, mood changes, worsening blood sugar control, weakness, or marked fatigue.
Statins
Some statins can become much stronger when taken with itraconazole. This can increase the risk of muscle toxicity.
Possible symptoms to report: new muscle pain, muscle weakness, or dark urine.
Blood thinners
Itraconazole can increase the effect of some blood thinners, which may raise bleeding risk.
Possible symptoms to report: unusual bruising, bleeding that is hard to stop, black stools, vomiting blood, or coughing up much more blood than usual.
Heart and rhythm medicines
Some combinations can increase the risk of serious heart rhythm problems. Itraconazole is also used cautiously in people with a history of ventricular dysfunction or heart failure unless the infection is serious.
Possible symptoms to report: fainting, palpitations, marked dizziness, increasing ankle swelling, or worsening breathlessness.
Sleeping tablets, sedatives, and some mental health medicines
Some sedatives and psychotropic medicines can become stronger when combined with itraconazole, increasing the risk of drowsiness, confusion, falls, or breathing problems.
Immunosuppressants
Medicines such as tacrolimus and ciclosporin can rise significantly when taken with itraconazole and usually need specialist monitoring.
Medicines that can make itraconazole less effective
Some medicines lower itraconazole levels, which means the antifungal may not work as well. This can happen with some anti-seizure medicines, rifampicin-type antibiotics, and some antiviral medicines.
Acid-reducing medicines and itraconazole capsules
Reduced stomach acid can lower the absorption of itraconazole capsules. This means reflux medicines, antacids, and some acid-suppressing treatments can affect how well the capsules work. The liquid formulation behaves differently and should not be treated as interchangeable with capsules.
What patients should do in practice
- Keep a current medicines list and bring it to appointments.
- Tell clinicians and pharmacists if you are taking itraconazole.
- Ask specifically about inhalers, steroid tablets, statins, blood thinners, and reflux treatment.
- Do not change doses or stop medicines without advice.
- If your itraconazole formulation changes, ask whether it should be taken with food or on an empty stomach.
When to seek medical advice
Seek urgent medical advice for severe bleeding, black stools, vomiting blood, severe muscle pain, fainting, marked palpitations, or rapidly worsening breathlessness.
Important
This page is not a full interaction database. For a complete medicine-by-medicine check, use the BNF interaction checker or speak to a pharmacist or clinician.
References
Amphotericin B interactions: what patients need to know
Key points
- Amphotericin B comes in different formulations, and they are not interchangeable.
- Its main interaction risks are different from the azoles.
- The most important problems are usually kidney stress, low potassium, low magnesium, and additive toxicity with other medicines.
- These risks matter most with intravenous treatment.
- If you hear “amphotericin B”, it is important to know which formulation is being used.
What is amphotericin B?
Amphotericin B is an antifungal used mainly for serious fungal infections. In modern UK practice this often means liposomal amphotericin B, but conventional amphotericin B deoxycholate is also a recognised formulation.
Why amphotericin B interactions are different from azoles
Unlike azole antifungals, amphotericin B does not mainly cause medicine interactions through liver enzymes. Its most important interaction risks usually relate to kidney injury, low potassium, low magnesium, and infusion-related effects.
The interaction groups most likely to matter
Other medicines that can damage the kidneys
This is one of the most important groups. Combining amphotericin B with other nephrotoxic medicines can increase the risk of kidney injury.
Diuretics, steroids, and other medicines that lower potassium
Amphotericin B can lower potassium, and some other medicines can make this worse. This may increase the risk of weakness, cramps, or heart rhythm problems.
Digoxin and heart-rhythm-sensitive situations
Low potassium caused by amphotericin B can make digoxin-related toxicity more likely and may increase the importance of electrolyte monitoring.
Flucytosine
When combined with flucytosine, specialist monitoring may be needed because toxicity can increase.
Some cancer medicines and intensive hospital treatments
In hospital, additive toxicity with other intensive treatments may be particularly important, especially where kidneys and electrolytes are already under strain.
White blood cell transfusions
Acute lung reactions are a recognised specialist concern if amphotericin B is given during or soon after leukocyte transfusions.
Why the formulations matter
Amphotericin B formulations are not interchangeable. Conventional amphotericin B deoxycholate and liposomal amphotericin B have different dosing, different handling by the body, and different safety profiles. Using the wrong formulation in the wrong dose has caused serious and even fatal errors.
In general, liposomal amphotericin B is less nephrotoxic than conventional amphotericin B deoxycholate, but it still requires careful monitoring.
What patients should do in practice
- Ask which amphotericin B formulation is being used.
- Tell the clinical team about all medicines, especially kidney-risk medicines, diuretics, steroids, and digoxin.
- Expect blood tests to monitor kidney function, potassium, and magnesium during intravenous treatment.
- Report weakness, reduced urine output, worsening swelling, palpitations, or marked dizziness.
When to seek medical advice
Seek urgent medical help for severe breathlessness, fainting, major palpitations, or a rapid deterioration during treatment.
Important
This page is educational and does not list every possible interaction. Amphotericin B treatment is usually managed by specialist teams, especially when given intravenously.
References
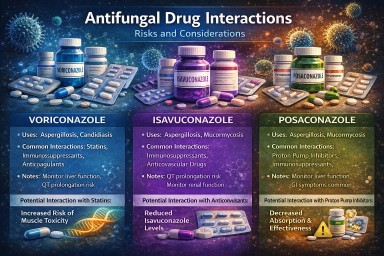
Isavuconazole interactions: what patients need to know
Key points
- Isavuconazole can interact with other medicines, but its interaction profile is often less complex than older azoles.
- It mainly interacts through CYP3A4.
- Some medicines can increase isavuconazole levels, while others can reduce its effectiveness.
- It has an important difference from some other azoles: it can shorten the QT interval.
- It is still essential to check new medicines carefully.
What is isavuconazole?
Isavuconazole is a newer azole antifungal used in invasive aspergillosis and some other serious fungal infections. It is often seen as having a more predictable interaction profile than some older azoles, but it still has important interactions.
Why isavuconazole interacts with other medicines
Isavuconazole is mainly linked to CYP3A4. This means some medicines can become stronger when combined with it, while other medicines can lower isavuconazole levels and reduce its effectiveness.
The interaction groups most likely to matter
Medicines that reduce isavuconazole effectiveness
Some medicines, including rifampicin-type antibiotics and certain anti-seizure drugs, can lower isavuconazole levels and may make treatment ineffective.
Steroids
Some steroid levels may rise with isavuconazole, although the interaction pattern is often less intense than with some older azoles.
Immunosuppressants
Medicines such as tacrolimus and ciclosporin may increase and usually need specialist review and monitoring.
Blood thinners
Some blood thinners may become stronger, increasing bleeding risk.
Statins
Some statin levels may increase, which can raise the risk of muscle side effects.
Important isavuconazole-specific points
QT shortening
Unlike several other azole antifungals, isavuconazole can shorten the QT interval. This is an important difference and should be considered when other heart medicines are being reviewed.
Generally simpler interaction profile
Compared with itraconazole and voriconazole, isavuconazole is often considered a little easier to manage in patients taking several medicines, although checks are still essential.
What patients should do in practice
- Tell your pharmacist or clinician if you are taking isavuconazole.
- Check before starting prescription, over-the-counter, or herbal medicines.
- Ask specifically about anti-seizure drugs, antibiotics, blood thinners, and heart medicines.
- Do not stop or change medicines without advice.
When to seek medical advice
Seek medical advice urgently for severe bleeding, fainting, severe palpitations, severe muscle pain, or rapid worsening after a medicine change.
Important
This page is educational and not a complete interaction database. For a full check, use the BNF interaction checker or speak to a pharmacist or clinician.
References
- BNF: isavuconazole monograph and interactions
- UK isavuconazole SmPCs - not available
Posaconazole interactions: what patients need to know
Key points
- Posaconazole can interact with other medicines, although its interaction pattern is often a little simpler than itraconazole or voriconazole.
- It mainly interacts through CYP3A4 inhibition.
- Important interaction groups include immunosuppressants, steroids, blood thinners, and some heart medicines.
- Some medicines can reduce posaconazole levels and make treatment less effective.
- Tablets and oral suspension are not interchangeable in the same way.
What is posaconazole?
Posaconazole is an azole antifungal used in aspergillosis and in some high-risk patients for prevention of fungal infection. It is often seen as somewhat easier to manage than some older azoles, but important interactions still exist.
Why posaconazole interacts with other medicines
Posaconazole mainly affects CYP3A4, a key liver enzyme involved in handling many medicines. This means some drugs can become stronger, while some combinations can lower posaconazole levels and make it less effective.
The interaction groups most likely to matter
Steroids
Posaconazole can increase exposure to some steroids, including inhaled or oral steroids, which may increase the risk of steroid side effects.
Immunosuppressants
Medicines such as tacrolimus and ciclosporin can rise significantly with posaconazole and usually need close specialist monitoring.
Blood thinners
Some blood thinners may become stronger, increasing bleeding risk.
Statins
Some statins can rise in level, increasing the risk of muscle problems.
Heart rhythm medicines
Some combinations can increase the risk of heart rhythm problems and need careful review.
Medicines that reduce posaconazole effectiveness
Some medicines, including rifampicin-type antibiotics and certain anti-seizure drugs, can lower posaconazole levels and may make treatment less effective.
Posaconazole formulations and absorption
Posaconazole comes in different forms, including tablets, oral suspension, and infusion. The oral suspension and tablets are not handled identically by the body and should not be assumed to be interchangeable dose-for-dose without clinical advice.
In practice, the tablets tend to be more predictable than the suspension.
What patients should do in practice
- Tell your pharmacist or clinician if you are taking posaconazole.
- Ask about new medicines, especially blood thinners, steroids, statins, and heart medicines.
- If your formulation changes, ask whether there are any special instructions.
- Do not stop or swap medicines without advice.
When to seek medical advice
Seek medical advice urgently for severe bleeding, fainting, major palpitations, severe muscle pain, or rapid worsening after a medicine change.
Important
This page does not list every interaction. For a full check, use the BNF interaction checker or speak to a pharmacist or clinician.