When ‘chest infections’ don’t respond: when to suspect ABPA or CPA (Patient guide)
Patients presenting with recurrent “chest infections” that do not respond to antibiotics are common in primary and secondary care. In a subset of these cases, the underlying cause may not be bacterial, but related to fungal disease or immune-mediated responses to Aspergillus.
This article summarises when to suspect allergic bronchopulmonary aspergillosis (ABPA) or chronic pulmonary aspergillosis (CPA), and how to move from repeated empirical treatment to a more structured diagnostic approach.
Key clinical message
Repeated antibiotic-treated exacerbations with limited response, particularly when symptoms improve with steroids and then relapse, should prompt reconsideration of the diagnosis.
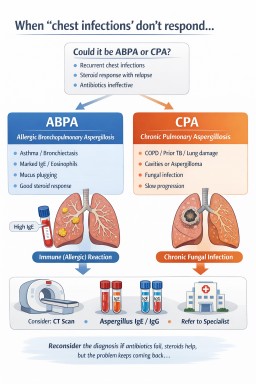
When to suspect ABPA or CPA
Consider aspergillosis-related disease in patients with:
- Recurrent “chest infections” with poor or inconsistent antibiotic response
- Steroid-responsive symptoms with relapse on reduction or cessation
- Persistent or unexplained radiological abnormalities
- Underlying lung disease:
- Asthma
- Bronchiectasis
- Chronic obstructive pulmonary disease (COPD)
- Previous tuberculosis or lung damage
- Raised or previously documented abnormalities in:
- Total IgE
- Eosinophils
- Aspergillus-specific markers (if previously tested)
These features are not diagnostic in isolation but should raise suspicion when seen together.
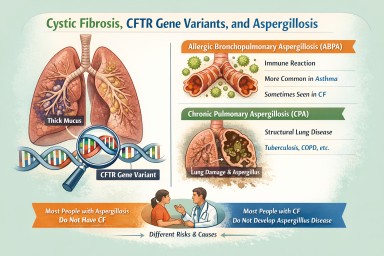
ABPA vs CPA: clinical distinction
| Feature | ABPA | CPA |
|---|---|---|
| Primary mechanism | Immune-mediated (allergic) | Chronic fungal infection |
| Typical background | Asthma, bronchiectasis | Structural lung disease, prior TB, COPD |
| Steroid response | Often marked | Variable (may improve symptoms but not disease) |
| Antibiotic response | Limited | Limited |
| Radiology | Mucus plugging, bronchiectasis | Cavities, fungal balls, fibrosis |
Common pitfalls in practice
- Repeated empirical antibiotics despite poor response
- Short courses of steroids without a long-term management plan
- Reliance on chest X-ray alone in persistent or atypical cases
- Failure to recognise patterns across multiple consultations or admissions
These patterns can lead to prolonged diagnostic delay, which is well described in CPA and ABPA.
Suggested diagnostic approach
1. Reassess the working diagnosis
When standard treatment fails, explicitly reconsider whether the presentation remains consistent with bacterial infection.
2. Imaging
- Escalate from chest X-ray to CT thorax where appropriate
- Look for:
- Cavitation
- Fungal ball (aspergilloma)
- Mucus plugging
- Bronchiectasis
3. Blood tests
- Total IgE
- Eosinophil count
- Aspergillus-specific IgE and IgG (where available)
4. Microbiology / further testing
Depending on context, consider sputum culture, fungal markers, or specialist input.
The steroid–relapse pattern
A common clinical scenario:
Exacerbation → steroids → improvement → relapse
This should raise suspicion of an underlying inflammatory or fungal-driven process rather than recurrent bacterial infection alone.
When to consider referral
Referral to a specialist centre (e.g. National Aspergillosis Centre, Manchester) may be appropriate where:
- Diagnosis remains uncertain
- Symptoms are persistent or progressive despite treatment
- Antifungal therapy is being considered or not tolerated
- Radiology suggests CPA or complex disease
Referral decisions should be made in the context of overall patient condition, comorbidities, and goals of care.
Why diagnosis is often delayed
- Overlap with common respiratory conditions
- Partial response to standard therapies
- Fragmentation across care settings
- Limited exposure to aspergillosis in routine practice
Recognising the pattern is often the key step in reducing delay.
Practical takeaways
- If antibiotics are not working, reconsider the diagnosis
- If steroids repeatedly improve symptoms, ask why
- Use CT imaging to clarify persistent abnormalities
- Aim for a clear, shared management plan
Guidelines and further reading
Which guideline should I use?
In UK practice, the British Thoracic Society (BTS) statement provides the most directly applicable clinical guidance. The ISHAM guidelines offer detailed international consensus, particularly for ABPA. Both are complementary and widely used.
- British Thoracic Society (BTS)
Clinical Statement on Aspergillus-related chronic pulmonary disease
UK guidance covering diagnosis and management of ABPA, CPA, and related conditions.
View BTS clinical statement
- International Society for Human and Animal Mycology (ISHAM)
Revised clinical practice guidelines for allergic bronchopulmonary aspergillosis/mycoses
International consensus guidance on diagnosis, classification, and management of ABPA.
View ISHAM guideline (European Respiratory Journal)
- Chronic Pulmonary Aspergillosis (CPA) guidelines
Denning DW et al. Chronic pulmonary aspergillosis: rationale and clinical guidelines for diagnosis and management
View PubMed record
Further professional resources
For clinician-focused guidance, referral pathways, and educational material:
https://aspergillosis.org/information-for-professionals/
This article is intended for educational purposes and should be interpreted in the context of individual clinical judgement.
Weekly Aspergillosis Research Update (7–11 April 2026)
Last reviewed: April 2026
Audience: Patients, carers, GPs, specialist nurses, and healthcare professionals
Key Points (Summary)
- Invasive pulmonary aspergillosis (IPA) can closely mimic other serious infections, including miliary tuberculosis, particularly in highly immunocompromised patients.
- Metagenomic next-generation sequencing (mNGS) is emerging as a valuable diagnostic tool in complex or unclear cases, though it is not yet widely available.
- Bruton tyrosine kinase (BTK) inhibitors are associated with a measurable risk of invasive fungal infections, with aspergillosis the most frequently reported.
- Host genetics (e.g. toll-like receptor variants) may influence susceptibility to invasive fungal disease, but this is not yet used clinically.
- Azole antifungal drugs remain high-risk for drug–drug interactions, particularly in patients receiving cancer therapies.
- Basic science research continues to identify new fungal targets and pathways, which may inform future treatments.
Contents
- Diagnosis and difficult cases
- Treatment-related risk and immunosuppression
- Genetics and susceptibility
- Asthma and Aspergillus sensitisation
- Drug interactions
- Emerging research and future treatments
- What this means for patients
- When to seek medical advice
Diagnosis and difficult cases
When aspergillosis looks like something else
A case report
(Ji H et al., 2026 – full text)
describes a patient with acute leukaemia who developed widespread “miliary” lung nodules—an imaging pattern classically associated with tuberculosis.
Despite this, the final diagnosis was invasive pulmonary aspergillosis (IPA).
Clinical interpretation
- Radiological appearances in immunocompromised patients can be non-specific.
- Aspergillosis may present without classic features such as halo signs or cavitation.
- Coinfection (e.g. TB + fungal disease) can further complicate interpretation.
This reinforces an important principle in clinical practice: lack of response to treatment should prompt reconsideration of the diagnosis.
The emerging role of mNGS
In this case, metagenomic next-generation sequencing (mNGS) helped establish the diagnosis by detecting fungal DNA directly from clinical samples.
Strengths of mNGS
- Broad pathogen detection (fungi, bacteria, viruses)
- Useful in culture-negative infections
- Can identify unexpected pathogens
Current limitations
- Limited availability outside specialist centres
- Cost and turnaround time
- Interpretation challenges (distinguishing infection from colonisation)
Bottom line: mNGS is promising, but currently complements rather than replaces standard diagnostics (culture, PCR, antigen testing).
Treatment-related risk and immunosuppression
BTK inhibitors and invasive fungal infection
A systematic review and meta-analysis
(PMID: 41954633)
including over 23,000 patients found that:
- Aspergillosis was the most commonly reported invasive fungal infection
- Central nervous system involvement was reported in a subset
Why BTK inhibitors increase risk
- They impair B-cell signalling
- They affect macrophage and neutrophil function
- This reduces the body’s ability to control fungal spores
Clinical implications
- Risk stratification is important
- Some patients may require antifungal prophylaxis
- Early recognition of symptoms is critical
This aligns with increasing recognition that modern targeted therapies can have unintended immunological effects.
Genetics and susceptibility
The role of innate immunity
A systematic review
(PMID: 41962654)
examined toll-like receptor (TLR) polymorphisms and fungal infection risk.
TLRs are part of the innate immune system and are responsible for recognising fungal components such as:
- β-glucans
- Cell wall proteins
Key insight
- Certain genetic variants were more frequently reported in patients with invasive aspergillosis
Important context:
- This does not yet translate into routine testing
- It may become relevant in the future for personalised risk prediction
Asthma and Aspergillus sensitisation
A 12-week prospective study
(PMID: 41949214)
found that:
- ~29% of asthma patients were sensitised to Aspergillus fumigatus
- No significant short-term differences in outcomes were seen compared to non-sensitised patients
Interpretation
- Sensitisation alone does not necessarily indicate active disease
- Clinical context remains critical
Important distinction:
- Sensitisation = immune response to Aspergillus
- ABPA (Allergic Bronchopulmonary Aspergillosis) = a specific inflammatory disease requiring treatment
Drug interactions
Itraconazole and erlotinib interaction
A case report
(PMID: 41953502)
demonstrated increased exposure to erlotinib when co-administered with itraconazole.
Mechanism
- Itraconazole inhibits CYP3A4
- This reduces drug metabolism
- Drug levels rise, increasing risk of toxicity
Clinical message
- Always review medications when starting antifungals
- Particular caution is needed in cancer, transplant, and multi-morbid patients
Useful tool:
Antifungal Interactions Checker
Emerging research and future treatments
Epigenetic regulation in Aspergillus fumigatus
A study
(PMID: 41928566)
identified the role of HosA in regulating fungal growth and virulence.
This type of work helps identify:
- Potential drug targets
- Mechanisms of antifungal resistance
New experimental antifungal compounds
A review
(Full text (PMC))
discusses sodium new houttuyfonate (SNH), which has shown activity in animal models of invasive aspergillosis.
Important caution:
- This is early-stage research
- It is not available as a treatment
What this means for patients
- Aspergillosis can sometimes be difficult to diagnose, especially if symptoms overlap with other conditions.
- If treatment is not working, your medical team may need to review or repeat tests.
- Some medications (especially for cancer or immune conditions) can increase risk of fungal infection.
- Antifungal medications are effective but require careful monitoring for interactions.
- New research is promising, but most advances take time to reach routine care.
When to seek medical advice
Seek medical attention urgently if you have:
- Worsening breathlessness
- Persistent fever
- Coughing up blood
- New chest pain
- Symptoms not improving with treatment
If you are immunocompromised, symptoms may progress quickly and should be assessed promptly.
References
- Ji H et al. Front Fungal Biol, 2026
- Srisurapanont K et al. Blood Adv, 2026
- Pereira Staiger MF et al. Clin Microbiol Infect, 2026
- Yokota H et al. Case Rep Oncol Med, 2026
- Chauhan NK et al. Monaldi Arch Chest Dis, 2026
- Zhou Z et al. Virulence, 2026
- Fang L et al. Front Pharmacol, 2026
Author & Review
This article is part of the Aspergillosis.org weekly research update series. It is intended for general educational purposes and reflects a structured summary of recent research.
Disclaimer: This content is not a substitute for medical advice. Always consult your healthcare team for individual care decisions.
Inflammation and Aspergillosis: Understanding “Stable”, “Flare”, and “Improving” Disease
Last reviewed: April 2026
Key points
- Inflammation is part of the body’s response to Aspergillus, but it does not always mean damage is actively worsening.
- “Stable” disease means no clear progression over time, not that the condition has disappeared.
- Symptoms in aspergillosis often vary because of other infections, especially in the lungs.
- Test results (such as IgE or CRP) can change without symptoms changing.
- Doctors make decisions based on the overall pattern over time, not a single test result.
Table of contents
- What is inflammation and why does it matter?
- Inflammation in different types of aspergillosis
- Clear definitions: active, flare, stable, improving, remission
- What does “stable disease” mean in practice?
- Why other infections cause flare-ups
- Understanding test results (CRP, IgE, scans)
- When test results worsen but symptoms do not
- How doctors decide what is happening
- Common patient questions
- When to seek medical advice
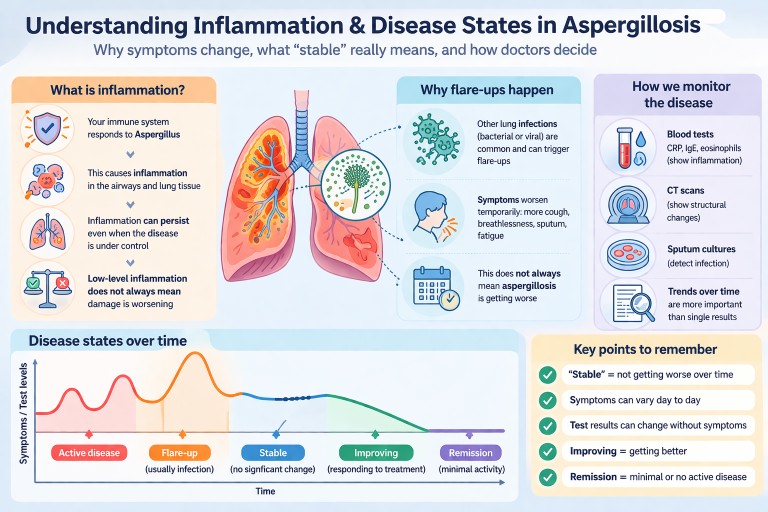
What is inflammation and why does it matter?
Inflammation is the body’s way of responding to something it sees as harmful. In aspergillosis, this is usually the fungus Aspergillus.
This response involves immune cells, chemicals, and changes in the lungs that aim to control the fungus. However, if inflammation continues over a long period (chronic inflammation), it can also contribute to:
- Ongoing symptoms (cough, breathlessness, fatigue)
- Mucus production
- Damage to lung tissue over time
Important: inflammation can be present at a low level without causing active damage. This is common in chronic conditions.
Inflammation in different types of aspergillosis
The type of inflammation depends on the form of aspergillosis:
- Allergic Bronchopulmonary Aspergillosis (ABPA): driven by an overactive allergic response. Blood markers such as IgE and eosinophils are often used to monitor this.
- Chronic Pulmonary Aspergillosis (CPA): caused by long-term infection in damaged lung tissue, leading to ongoing inflammation and structural changes.
- Aspergillus bronchitis: persistent infection with inflammation, often causing chronic cough and sputum.
In all cases, inflammation may improve with treatment but often does not disappear completely.
Clear definitions: disease states
Doctors use the following terms to describe how the disease is behaving:
- Active disease: symptoms, tests, or scans are getting worse over time
- Flare-up: a short-term worsening, often triggered by infection or another stress on the body
- Stable: no clear overall change over time
- Improving / responding to treatment: symptoms and/or tests are getting better
- Remission: minimal or no signs of active disease (used more often in ABPA)
Key point: these states are not fixed — patients may move between them.
What does “stable disease” mean in practice?
“Stable” means that, over a period of time (weeks to months), there is no clear evidence that the disease is progressing.
This usually includes:
- No worsening of key symptoms
- No new complications (e.g. haemoptysis, significant weight loss)
- Imaging (CT scans) showing no progression
- No need to increase treatment
What stable does NOT mean:
- It does not mean symptoms are absent
- It does not mean inflammation is zero
- It does not mean you will feel the same every day
Many patients with stable disease still experience day-to-day variation in symptoms.
Why other infections cause flare-ups
People with aspergillosis are more vulnerable to other lung infections (bacterial or viral).
This is because:
- Lung structure may already be damaged
- Mucus clearance is less effective
- The immune system is already active
When another infection occurs, it can trigger a flare-up, causing:
- Increased cough and breathlessness
- More or thicker sputum
- Fatigue and feeling unwell
- Raised inflammatory markers (e.g. CRP)
Crucial point: this does not necessarily mean the aspergillosis itself is worsening. It is often a temporary additional problem.
Understanding test results
Doctors use several types of tests to monitor inflammation and disease activity:
- CRP / ESR: general markers of inflammation
- IgE: particularly important in ABPA
- Eosinophils: linked to allergic inflammation
- CT scans: show structural changes in the lungs
- Sputum cultures: detect infection
Important limitations:
- No single test gives a complete picture
- Results can fluctuate for many reasons
- Changes must be interpreted over time
When test results worsen but symptoms do not
This situation is common, especially in ABPA.
For example, IgE levels may rise without any noticeable change in symptoms.
This may happen because of:
- Natural biological variation
- Exposure to allergens
- A mild or early flare that has not yet caused symptoms
Key point: a change in a single test result does not automatically mean the disease is worsening.
Doctors will usually:
- Repeat tests
- Look for consistent trends
- Assess symptoms and scans
If symptoms remain stable and no other changes are seen, the condition may still be considered stable — but monitored more closely.
How doctors decide what is happening
Clinicians do not rely on a single result. Instead, they assess the pattern over time:
- Are symptoms changing?
- Are test results consistently rising or falling?
- Are scans stable or changing?
- Is the patient responding to treatment?
This combined assessment is called the clinical picture.
Common questions
If I feel better, what is that called?
This is usually described as improving or responding to treatment. In some cases (especially ABPA), it may be called remission.
Does inflammation always mean damage?
No. Low-level inflammation can persist without causing further harm.
Why do my symptoms change from day to day?
This is common and often relates to infections, environment, or general health rather than disease progression.
Can aspergillosis affect the whole body?
It can have wider effects, but it mainly affects the lungs in most patients.
When to seek medical advice
Seek medical advice if you notice:
- Persistent worsening of symptoms
- New haemoptysis (coughing up blood)
- Significant weight loss
- Symptoms not improving after a suspected infection
- Concerns about test results
Author and review
Author: Aspergillosis Patient Education Team
Reviewed by: National Aspergillosis Centre (UK)
References
- Denning DW et al. Chronic pulmonary aspergillosis guidelines
- ISHAM ABPA guidelines
This article is for general information only and is not a substitute for medical advice.
Can overseas patients access specialist centres like NAC? A clear guide for patients and families
Last reviewed: April 2026
Key points
- Specialist centres such as the National Aspergillosis Centre (NAC) cannot provide individual medical advice directly to patients, especially from overseas.
- This is due to safety, legal, and clinical responsibility requirements.
- The correct route is through a local doctor, using a formal referral process.
- A referral does not guarantee that a patient will be seen.
- Even when doctors contact specialist centres, there are limits to what can be offered.
- NAC does not offer private care, although some other centres do.
Table of contents
- Why can’t specialist centres respond directly?
- The correct way to get help
- What does a referral mean?
- Will a referral always be accepted?
- Do personal requests help?
- Limits even when doctors are involved
- What about private care?
- Common myths
- When to seek urgent help
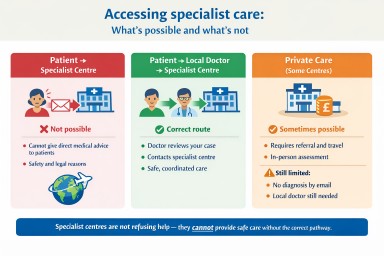
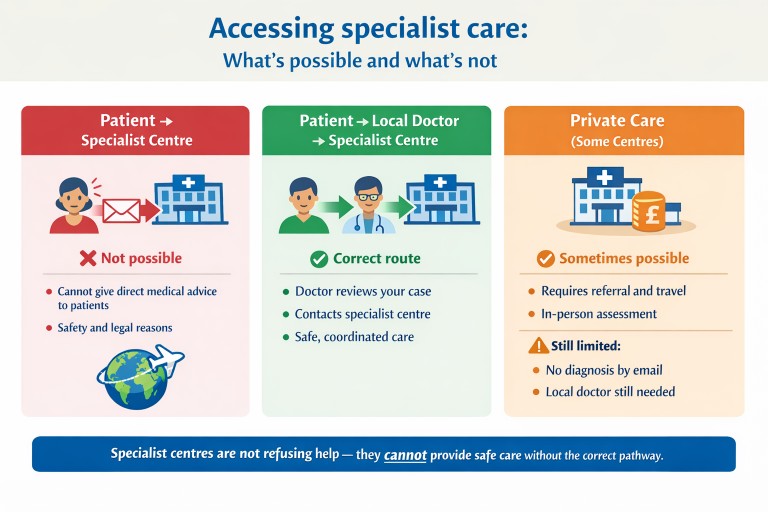
Why can’t specialist centres respond directly to patients?
It is natural to want to contact a specialist centre directly, especially when dealing with a complex condition such as aspergillosis. However, centres like NAC cannot provide individual medical advice directly to patients.
This is not a matter of choice—it is because they cannot safely or legally do so.
Patient safety
- Safe medical advice requires full access to medical records, test results, and examination findings
- Doctors must be able to monitor progress and adjust treatment
- This cannot be done through messages or emails alone
Legal and regulatory requirements
- Doctors must follow strict rules set by regulators such as the General Medical Council (GMC) in the UK
- They cannot take responsibility for a patient without a formal clinical relationship
Responsibility for care
- Any doctor giving advice must be able to take responsibility for outcomes
- This requires recognised clinical pathways and follow-up arrangements
In summary: Specialist centres are not refusing help—they cannot provide care outside safe and regulated systems.
The correct way to get help
The safest and most effective way to access specialist expertise is through your local doctor.
Step 1 – See a local specialist
- Respiratory doctor
- Infectious disease specialist
- Internal medicine specialist
Step 2 – Assessment and initial care
- Your doctor reviews your symptoms and history
- Tests are arranged where needed
- Treatment may be started
Step 3 – Referral or specialist advice
- Your doctor may refer you to a specialist centre
- Or they may contact a centre such as NAC for advice
This approach ensures your care is coordinated, safe, and based on full clinical information.
What does a referral mean?
A referral is when your doctor formally asks another specialist or centre to review your case or consider seeing you.
This is not just a message—it is a structured clinical process.
What does a referral usually include?
- Your medical history
- Details of your symptoms
- Results of tests (such as scans or laboratory results)
- Treatments you have already received
- A clear reason for referral
This allows the specialist centre to understand your situation safely and properly.
What happens next?
After reviewing the referral, the specialist centre may:
- ✔️ Accept the referral and arrange an appointment
- ✔️ Provide advice to your doctor without seeing you
- ✔️ Suggest a more appropriate service
- ❌ Decline the referral
A referral is like a formal handover between doctors—it does not guarantee an appointment.
Will a referral always be accepted?
Even when your doctor makes a referral, it is important to understand that the referral may not always be accepted.
Why might a referral not be accepted?
Eligibility criteria
- Specialist centres often have strict criteria for the patients they can see
- Some services are commissioned only for specific conditions
Geographic and funding rules
- Access may depend on healthcare system or funding arrangements
Clinical suitability
- The centre may decide your care can be managed locally
- They may offer advice instead of accepting the referral
Capacity
- Specialist centres often manage large numbers of complex patients
What happens if it is not accepted?
- Your doctor may still receive expert advice
- You may be directed to another service
- Your care continues locally
This does not mean you are being refused help.
Why specialist centres may not be able to confirm anything to you
Patients and families sometimes ask a specialist centre whether a referral has been received, reviewed, or accepted.
It is important to understand that the centre may not be able to confirm or discuss this with you directly.
Why is this?
Confidentiality and data protection
- Medical information is protected by strict confidentiality rules
- In the UK, this includes laws such as data protection legislation and professional duties of confidentiality
- Centres must be certain they are communicating with the correct person and through approved channels
Communication is usually between doctors
- Referrals are handled as clinician-to-clinician communication
- Responses are normally sent back to the referring doctor, not directly to the patient
No confirmation does not mean no action
- If you do not receive a reply from the centre, it does not necessarily mean your referral has been ignored
- Advice or decisions may already have been communicated to your doctor
What should you do?
- Contact your own doctor for updates
- Ask whether a referral has been sent and if a response has been received
- Discuss next steps with your clinical team
Summary
Specialist centres usually communicate with your doctor, not directly with patients. This is to protect your privacy and ensure safe, appropriate communication.
Do personal requests from patients or families help?
Personal requests are completely understandable, but specialist centres cannot provide individual medical advice directly to patients, even in urgent situations.
This is because they cannot safely or legally do so without a doctor involved.
These requests may still help with general information and guidance, but they do not usually lead to diagnosis or treatment advice.
Limits even when doctors are involved
When your doctor contacts a specialist centre, this is the correct route—but there are still limits.
What specialist centres can offer
- Expert opinion
- Suggestions for diagnosis
- Guidance on tests
- Interpretation of results
What they cannot usually provide
- Direct patient care
- Full responsibility for treatment
- Definitive diagnosis without full assessment
Your local doctor remains responsible for your care.
What about private care?
The National Aspergillosis Centre (NAC) does not offer private care.
- You cannot arrange to be seen there privately
- Access is through NHS referral pathways only
Some other specialist centres may offer private consultations. However:
- A referral and medical records are usually required
- An in-person assessment is typically needed
- Private care does not bypass safety or legal requirements
Common myths
- “I can contact a specialist centre directly for help”
→ Centres cannot provide individual advice directly to patients - “If it’s urgent, they will make an exception”
→ The same safety rules apply to all patients - “A referral guarantees I will be seen”
→ Referrals are reviewed and may not always be accepted - “Private care means I can be seen anywhere”
→ Not all centres offer private care (NAC does not) - “A detailed email is enough for diagnosis”
→ Diagnosis requires full clinical assessment
When to seek urgent medical help
If you or someone you care for has any of the following, seek urgent local medical care:
- Severe breathlessness
- Chest pain
- Coughing up blood
- Confusion or extreme drowsiness
- High fever that is not improving
Do not delay seeking help while trying to contact overseas specialists.
Summary
Specialist centres such as NAC play an important role in supporting complex conditions like aspergillosis. However, they must work within systems designed to keep patients safe.
The most effective way to access their expertise is through your own doctor, using formal referral pathways and specialist advice where needed.
Author & review information
Prepared for patient education purposes.
Aligned with UK specialist centre practice and patient safety guidance.
References & further reading
- General Medical Council (UK) – Good medical practice
- NHS England – Specialist services referral guidance
- European Lung Foundation – Aspergillosis information
- National Aspergillosis Centre – aspergillosis.org
AntifungalInteractions.org – A Specialist Resource for Safer Antifungal Treatment
Last reviewed: April 2026
Managing antifungal medications can be complex. Many antifungal drugs interact with other medicines, foods, and even supplements.
To support both patients and healthcare professionals, a dedicated resource is available:
AntifungalInteractions.org.
Key Points
- A specialist database focused specifically on antifungal drug interactions
- More detailed and targeted than general resources such as the British National Formulary (BNF)
- Includes guidance designed for both healthcare professionals and patients
- Regularly updated (typically several times per month)
- Maintained by an experienced clinical pharmacist and prescriber
- Owned and supported by the Fungal Infection Trust
What is AntifungalInteractions.org?
AntifungalInteractions.org is a dedicated online database designed to help users understand how antifungal medications interact with:
- Other prescribed drugs
- Over-the-counter medications
- Herbal supplements
- Certain foods and drinks
Unlike general drug reference tools, this resource focuses specifically on antifungal medicines, making it particularly useful for conditions such as aspergillosis, where treatment often involves long-term or complex therapy.
Why This Resource Matters
1. Antifungal drugs are complex
Common antifungal medications such as azoles (e.g. itraconazole, voriconazole, posaconazole) are known to interact with many other drugs.
These interactions can:
- Increase side effects
- Reduce treatment effectiveness
- Require dose adjustments or monitoring
2. General resources may not go far enough
Widely used tools like the British National Formulary (BNF) are essential, but they are designed for broad use across all medicines.
AntifungalInteractions.org provides:
- More detailed interaction explanations
- Practical interpretation of risk
- Condition-specific relevance
3. It supports informed discussions
The database is not a replacement for clinical advice, but it can help patients and clinicians:
- Prepare for consultations
- Understand potential risks
- Ask more informed questions
Who Maintains the Database?
AntifungalInteractions.org is maintained by:
Saarah Niazi-Ali
MPharm, PG Cert (General Pharmacy Practice), PG Dip (Advanced Clinical Pharmacy Practice),
Independent Pharmacist Prescriber, Non-Medical Prescribing (Level 7), Final Medical Signatory
The database is updated frequently—typically 3–4 times per month, often on a weekly basis—ensuring that information remains current and clinically relevant.
Governance and Ownership
The resource is owned and supported by the Fungal Infection Trust, a UK-based organisation dedicated to improving the understanding, diagnosis, and treatment of fungal diseases.
This ensures that the database:
- Remains focused on patient benefit
- Is aligned with specialist fungal disease care
- Supports both clinical practice and patient education
Who Is It For?
Patients and carers
- To better understand their medications
- To check for potential interactions
- To support conversations with their clinical team
Healthcare professionals
- Infectious disease specialists
- Respiratory clinicians
- Pharmacists
- GPs managing complex patients
It is particularly valuable for clinicians managing conditions such as:
- Chronic pulmonary aspergillosis (CPA)
- Allergic bronchopulmonary aspergillosis (ABPA)
- Other fungal infections requiring long-term antifungal therapy
How Does It Compare to Other Resources?
| Feature | AntifungalInteractions.org | General Drug References (e.g. BNF) |
|---|---|---|
| Focus | Antifungal-specific | All medicines |
| Level of detail | High (specialist) | Moderate (broad coverage) |
| Patient-friendly explanations | Yes | Limited |
| Update frequency | Frequent (monthly/weekly) | Regular but broader scope |
Important Notes for Patients
While this database is a valuable resource, it should be used appropriately:
- Do not stop or change medication based on what you read
- Always discuss concerns with your doctor, pharmacist, or specialist team
- Use the information to support—not replace—medical advice
When to Seek Medical Advice
Contact your healthcare provider if you:
- Start a new medication while on antifungal treatment
- Experience new or worsening side effects
- Are unsure whether a supplement or food is safe
- Have been advised of a potential interaction
Summary
AntifungalInteractions.org is a highly valuable, specialist resource that fills an important gap in antifungal care.
Its combination of:
- Expert clinical oversight
- Frequent updates
- Patient-accessible explanations
- Specialist focus
makes it an important tool for both patients and healthcare professionals managing fungal disease.
Further Reading
- British National Formulary (BNF)
- Aspergillosis treatment guidance (aspergillosis.org)
Author & Review
Prepared for Aspergillosis patient and healthcare education.
Content aligned with UK specialist practice and reviewed for clarity and safety.
Help shape the future of aspergillosis care across Uk & Europe
“Can patients do more than just cope with this condition?”
The answer is yes.
The European Lung Foundation (ELF)
and its
Aspergillosis Patient Advisory Group (PAG)
give patients and carers a chance to contribute to something bigger: better awareness, better information, better research, and better care.
What is ELF?
ELF is a Europe-wide organisation that brings patients, carers, healthcare professionals and researchers together to improve lung health information, treatment and care.
One of ELF’s strongest advantages is that it works across Europe, not just in one country. It also makes key information available in several languages, helping more people access reliable information about lung conditions, including aspergillosis.
You can read ELF’s patient information on aspergillosis here:
Aspergillosis – European Lung Foundation.
What is the Aspergillosis Patient Advisory Group?
The Aspergillosis PAG is part of ELF’s wider network of
Patient Advisory Groups.
These groups bring together people with experience of specific lung conditions, or experience as carers, so that patient views can help improve treatment and healthcare.
The Aspergillosis PAG works to raise awareness of aspergillosis and improve diagnosis, treatment and care. It also works alongside healthcare professionals and researchers involved in the Chronic Pulmonary Aspergillosis Network (CPAnet), helping identify research priorities and information gaps for both patients and professionals.
Why does this matter?
Aspergillosis is still not well understood in many places. Diagnosis can be delayed, information can be hard to find, and patients often feel that few people truly understand what living with the condition is like.
By involving patients and carers directly, ELF helps ensure that real-life experience is not left out of the conversation. This can influence education, awareness work, research priorities and wider discussions about care across Europe.
What is in it for the patient or carer?
This is an important question, because volunteering your time and energy is a big ask, especially when you are already managing illness, fatigue, appointments, uncertainty or caring responsibilities.
So it is only fair to be clear and honest about what people may gain from taking part.
1. A chance to make your experience count
Many people with aspergillosis have learned difficult lessons the hard way. Getting involved gives you a chance to turn that experience into something useful — helping improve information, shape priorities and make life a little easier for future patients.
2. Better understanding and confidence
Being involved can help you better understand how research, awareness work and patient representation operate. Some people find that this gives them more confidence when speaking about their condition and navigating their own care.
3. Connection beyond your local area
Because ELF is Europe-wide, patients are not limited to the perspective of one hospital, one region or one country. For people living with a relatively uncommon condition, that wider connection can feel valuable and reassuring.
4. The opportunity to be heard
Many patients are used to feeling overlooked. PAGs are designed so that patient and carer perspectives are actively included in projects and discussions, rather than being an afterthought.
5. A sense of purpose
Some people find that involvement helps them move from simply living with a difficult condition to doing something constructive with that experience. It will not suit everyone, but for some it can be meaningful.
6. Support and training
ELF says it provides support, guidance and training to help people share their perspective and get involved in projects. It also encourages interested patients and carers to use its free online European Patient Ambassador Programme (EPAP), which introduces the skills and knowledge needed to represent yourself and others effectively.
What it is not
It is also important to be realistic.
- It is not medical care.
- It does not replace your doctor, nurse or specialist team.
- It is not a route to faster treatment.
- It is not a paid role.
ELF states that PAG involvement is voluntary and that it is unable to pay for people’s time.
Who can join?
ELF says most PAGs are open to new members from European countries. In general, people are invited to get involved if they are over 18, have experience as a patient or carer, live in a European country, can communicate in English, are interested in improving healthcare and treatment across Europe, and are willing to share their perspective.
That said, this should not feel like an all-or-nothing commitment. Not everyone can give a lot of time, and health can change. Even modest involvement can still be worthwhile.
Why mention this to our groups?
Many people in aspergillosis support communities have exactly the kind of insight that is valuable here: the reality of diagnosis, treatment, daily management, side effects, uncertainty, isolation, and learning how to cope.
Those experiences matter. They can help improve what is researched, what is explained, and how future patients are supported.
Interested?
You can explore more here:
- ELF information on aspergillosis
- ELF Patient Advisory Groups
- ELF Aspergillosis Patient Advisory Group
You do not need to be an expert. You do not need to be highly confident. You do not need to commit to everything.
But if you have lived with aspergillosis, or cared for someone who has, your experience may be more valuable than you think.
In short: this is a voluntary opportunity to help improve understanding, research and care for aspergillosis across Europe, while connecting with a wider patient community and making sure lived experience is heard.
Why do doctors ask me to repeat my history — and sometimes not read my summary?
Many patients, especially those with long-term or complex health problems, say the same thing: “Why do I always have to repeat everything?” Some also say that even when they bring a short written summary, it is put to one side and does not seem to be read straight away.
This can feel frustrating, tiring, and dismissive. In most cases, though, it is not because the doctor does not care. It is usually because of how medical appointments are structured, how clinicians are trained, and the time pressures built into the healthcare system.
This article explains what may be happening, why doctors often work this way, and what can sometimes help patients get more out of appointments.
Key points
- Doctors are usually not ignoring you or your efforts.
- They are trained to hear the story directly from the patient for safety and accuracy.
- Medical notes can be long, incomplete, hard to access quickly, or spread across different systems.
- A written summary can still be helpful, but it may work best as a support tool rather than a replacement for discussion.
- The system is often particularly difficult for people with complex, long-term conditions.
Contents
- Why do doctors ask patients to repeat information?
- Why might a written summary be set aside?
- What are doctors trying to achieve?
- Why is this harder for people with long-term conditions?
- What can help in appointments?
- Common questions
- When to seek further support
Why do doctors ask patients to repeat information?
Many patients assume that if something is already in the notes, the doctor should simply read it and move on. In practice, clinicians are usually taught not to rely only on previous notes. They are expected to take a current history directly from the patient wherever possible.
1. They need a fresh history
Symptoms can change. A note written last week, last month, or even earlier the same day may no longer fully reflect what is happening now. A doctor needs to understand the present situation, not just the recorded one.
This matters because:
- new symptoms may have appeared
- older symptoms may have improved or worsened
- important details may have been left out of earlier notes
- previous notes can sometimes contain misunderstandings or errors
2. Hearing the story directly is part of diagnosis
Doctors do not just collect facts. They also listen to how symptoms are described, what concerns the patient most, what order things happened in, and whether there are any clues that do not fit the previous record.
For example, hearing a patient say “this is much worse than usual” or “the pain has changed completely” may matter just as much as what is already written down.
3. Each clinician is responsible for their own decisions
Every doctor is professionally responsible for the decisions they make in that consultation. Because of that, they usually need to confirm the key information for themselves rather than relying entirely on another person’s notes or summary.
4. Notes are not always as clear or complete as patients imagine
Patients often assume the notes tell the whole story. Sometimes they do not. They may be brief, scattered across different entries, missing key context, or written in a way that does not quickly explain the current problem.
Why might a written summary be set aside?
Patients who have made the effort to prepare a summary can feel especially upset when a doctor places it to one side. It may look like the document is being ignored. Often, however, the doctor is following a routine approach to consultation rather than rejecting the information.
1. Many consultations follow a familiar structure
Doctors are often trained to work in a rough sequence:
- listen to the patient’s account
- ask follow-up questions
- clarify the current concern
- review supporting information and records
- decide what to do next
So a written summary handed over at the start may be used later, not immediately.
2. They may not want the written summary to shape their thinking too early
Clinicians are often cautious about being overly influenced by somebody else’s wording before they have heard the patient directly. This is part of clinical reasoning and risk management. They may want to form their own understanding first, then compare it with the summary.
3. Reading while listening can be difficult
In a short appointment, a doctor may feel they cannot properly read a document, listen closely, ask questions, and maintain eye contact all at the same time. They may therefore choose to focus first on the conversation.
4. Some summaries are not easy to use quickly
Even a well-meant summary may be hard to absorb if it is too long, too detailed, or does not make the current issue obvious straight away. Doctors are often trying to answer one urgent question first:
What is the main problem today?
If that is not immediately clear from the page, they may return to direct questioning.
What are doctors trying to achieve?
From a patient’s point of view, repeating information can feel inefficient and unnecessary. From a clinician’s point of view, the consultation is often trying to achieve several things at once:
- Accuracy: understanding what is happening now, not just what happened before
- Safety: checking for changes, gaps, or warning signs
- Clarity: identifying the most important issue for that appointment
- Responsibility: making decisions based on information they have personally checked
That does not make the experience any less frustrating for patients, but it can help explain the behaviour.
Why is this harder for people with long-term conditions?
This problem is often worse for people who have:
- complex diagnoses
- multiple health conditions
- long medical histories
- many medications
- appointments with different teams or hospitals
If you live with a chronic condition, you may have repeated the same history many times. You may also already be tired, breathless, in pain, stressed, or worried. In that situation, being asked again to explain everything can feel overwhelming.
This is a real systems problem. It is not a sign that you are failing to explain yourself properly, and it is not unreasonable to find it difficult.
What can help in appointments?
There is no perfect solution, but some approaches can make appointments easier and increase the chance that useful information is taken in.
Start with the main issue today
A helpful opening sentence can be:
“The main issue today is…”
This quickly gives the doctor a focus and may reduce the need to retell everything in full.
Keep written summaries short
A one-page summary is usually easier to use than a longer document. It can include:
- main diagnoses
- current medications
- important recent events
- relevant allergies or major problems
- today’s main concern at the top
Offer the summary rather than relying on it
Instead of assuming it will be read immediately, it may help to say:
“I have brought a short summary in case it is useful.”
or:
“Would it help if I gave you a one-page summary of my background?”
This fits better with how many consultations flow.
Use the summary as a support tool
A written summary often works best as:
- a memory aid for the patient
- a quick reference for the doctor
- a back-up in case important facts are missed
It may be less effective if used as a substitute for the whole conversation.
Say when repetition is difficult
If repeating your history is hard because of pain, fatigue, breathlessness, stress, memory problems, or the complexity of your condition, it is reasonable to say so politely.
“I’m happy to summarise, but I do find repeating the full history difficult.”
That can help the doctor understand the burden on you.
Common questions
Are doctors not reading my notes at all?
Sometimes they have read some of them, but not everything. Sometimes they have skimmed the record for key issues. Sometimes the most relevant information is hard to find quickly. The fact that they ask questions does not always mean they have read nothing.
Why do they not just read my summary instead of asking me?
Because they are usually expected to hear the current story directly, check the important facts for themselves, and understand what matters most right now.
Is there any point bringing a written summary?
Yes. A good summary can still be very helpful. It may save time, improve accuracy, and make it easier to communicate complex information. It is just not always used in the way patients expect.
Does putting my summary to one side mean it has been ignored?
Not necessarily. It may be read later in the appointment, after the consultation, or used as a reference rather than read word for word in front of you.
When to seek further support
You may want more support if:
- you repeatedly feel that important information is not being understood
- your condition is complex and hard to explain in a short appointment
- you feel too unwell or distressed to repeat your history each time
- you think communication problems are affecting your care
Possible options include:
- asking whether a clinic letter or care plan summary can be used
- bringing a relative, friend, or advocate to appointments
- keeping a short up-to-date medical summary with you
- asking whether the main issue for that appointment can be agreed early on
Final thought
It can be upsetting to feel that your effort has been overlooked or that you are being asked to say the same things again and again. Usually, though, what you are seeing is not indifference but the way modern consultations are structured around safety, time pressure, and direct assessment.
Your summary is still worth bringing. The most useful expectation is often not that it will replace the conversation, but that it may support it.
Important note
This article is general educational information. It is not medical advice and cannot explain every individual consultation or healthcare setting.
Author and review information
Prepared as general patient information to help explain common experiences in healthcare appointments.
Aspergillosis and Diet: coping with weight loss, poor appetite, food avoidance and stomach symptoms
For: patients, carers, general practitioners, specialist nurses and other non-specialists
Last reviewed: 8 April 2026
Important: This page is general information. It does not replace advice from your own clinical team.
Key points
- Eating difficulties are common in aspergillosis, especially in chronic pulmonary aspergillosis (CPA) and in people who also have other lung disease.
- The problem is often not simply “poor appetite”. Breathlessness, cough, fatigue, reflux, nausea, altered taste and medicine side effects can all make eating difficult.
- Some people gradually cut out more and more foods because eating feels uncomfortable or because they have been told certain foods are “bad” for lung symptoms.
- For many patients, the main nutritional goal is not a “perfect” diet. It is getting enough energy, protein and fluids in ways that feel manageable.
- “Little and often”, food fortification and nourishing drinks are often more realistic than trying to eat three large meals a day.
- Ongoing weight loss, a very restricted diet, persistent nausea, reflux or difficulty eating most days should be discussed with a doctor, specialist team or dietitian.
Why diet can become a major problem in aspergillosis
Many people living with aspergillosis find that eating becomes much harder than it used to be. This is particularly important in chronic pulmonary aspergillosis (CPA), where weight loss, fatigue and general ill health are common features of the illness. In practical terms, the body may need more energy while the person is less able to eat comfortably.
Several problems can overlap:
- Breathing takes more effort, which can increase energy needs.
- Coughing or breathlessness can interrupt meals.
- Tiredness can make shopping, cooking and eating feel like hard work.
- Inflammation and chronic illness can reduce appetite and contribute to muscle loss.
- Antifungal treatment and other medicines can cause nausea, altered taste, indigestion or poor appetite.
- Reflux, bloating or early fullness may mean that even small meals feel uncomfortable.
For some patients this creates a vicious circle: eating becomes unpleasant, intake falls, weight drops, strength falls, and eating may then feel even more difficult.
Who is most affected?
Not every patient with aspergillosis has major nutritional problems, but some groups are more likely to struggle. This includes people with:
- Chronic pulmonary aspergillosis (CPA)
- pre-existing lung disease such as chronic obstructive pulmonary disease (COPD), bronchiectasis or previous tuberculosis
- long-term fatigue, breathlessness or coughing
- persistent nausea or reflux symptoms
- a history of recent unplanned weight loss
- side effects from antifungal or other medicines
- anxiety around eating because meals repeatedly trigger symptoms
Some people with allergic bronchopulmonary aspergillosis (ABPA) also report poor intake or nutritional difficulties, although the pattern may differ from CPA. In ABPA, steroid treatment, asthma burden, medicine effects and general symptom load may all influence diet.
How eating can become difficult
People often describe eating problems in ways that do not sound like a classic “nutrition” issue. They may say things like:
- “I get full after a few mouthfuls.”
- “I cannot face a proper meal.”
- “Eating makes me cough.”
- “I feel uncomfortable after food.”
- “Some foods seem to sit badly.”
- “I only eat a few safe foods now.”
These experiences are important. They suggest that the real problem may be a mixture of breathlessness, upper gastrointestinal symptoms, medicine effects and learned food avoidance, not simply a lack of willpower or poor food choices.
When eating shrinks into a “minimal diet”
Some patients end up eating very little, often because that feels safer or more manageable than trying to eat normally. A “minimal diet” may look like:
- very small amounts of food only once or twice a day
- mostly soft or liquid foods
- reliance on tea, toast, soup or yoghurt
- long gaps without eating
- skipping meals because eating feels exhausting
This is understandable, but it can become a serious problem. Small intake over time may lead to:
- weight loss
- loss of muscle mass
- greater weakness and fatigue
- slower recovery from illness
- reduced ability to cope with infections or treatment
If a patient is managing only tiny amounts of food, the first goal is often not to rebuild a “normal” diet immediately. It is to make intake easier, more comfortable and more nourishing.
Avoiding many food types
Another common pattern is gradual food restriction. Patients may stop eating several food groups because they believe these foods worsen mucus, cough, reflux, nausea or fungal disease.
Examples include avoiding:
- dairy products
- sweet foods
- bread or dry foods
- meat
- acidic foods
- foods linked in the mind to a previous bad episode
Sometimes there is a genuine reason for avoiding a particular food. For example, reflux may make acidic or very fatty foods uncomfortable, and a dry crumbly food may clearly trigger coughing. The difficulty is that repeated bad experiences can also lead to over-restriction, where more and more foods are cut out than is really necessary.
That can leave the diet low in calories, low in protein and very repetitive. In practice, the aim is usually to adapt foods rather than cut out whole food groups unless there is a clear reason to avoid them.
Could the stomach or gut be part of the problem?
Yes. This is often overlooked.
Some patients with aspergillosis describe symptoms that sound mainly digestive rather than respiratory, for example:
- nausea
- heartburn or reflux
- bloating
- feeling full very quickly
- upper abdominal discomfort
- reduced appetite after starting or changing medication
- alternating diarrhoea and constipation
There are several possible reasons:
- Medicine side effects, including antifungals
- Gastro-oesophageal reflux disease (GORD), which can also worsen cough
- reduced activity levels and chronic illness
- constipation, especially when intake is poor or medicines contribute
- co-existing gastrointestinal disease that is separate from aspergillosis
If eating repeatedly causes upper abdominal or chest discomfort, or if reflux and nausea are prominent, it is reasonable to think of this as a symptom needing review rather than simply a “fussy eating” problem.
Practical ways to make eating easier
Different things help different people, but these approaches are often more realistic than trying to push through large meals.
1. Think “little and often”
Many people do better with five or six small eating opportunities through the day instead of three big meals. That may mean a small breakfast, a mid-morning snack, a light lunch, a nourishing drink, an evening meal and a supper snack.
2. Lower the effort of eating
Soft, moist foods are often easier than dry, chewy or crumbly foods. Examples include:
- porridge
- yoghurt
- custard or rice pudding
- mashed potato with added butter or cheese
- scrambled eggs
- soup with cream or grated cheese
- stews, casseroles or sauced dishes
3. Use drinks as nutrition
For some patients, drinks are easier to manage than food. Nourishing options can include:
- milky drinks
- smoothies
- milkshakes
- fortified hot drinks
- commercial oral nutritional supplements if prescribed or advised
4. Rest before eating
If fatigue or breathlessness are major barriers, it can help to eat after a rest rather than after exertion. Some people find breakfast or lunch easier than an evening meal.
5. Sit upright and stay upright afterwards
This can be especially helpful when reflux, coughing or chest discomfort are part of the picture.
6. Slow the pace
It is acceptable to eat slowly and pause often. Some patients benefit from smaller mouthfuls and short breathing pauses between them.
7. Look for manageable variety
If the diet has become very narrow, widening it gently may be more successful than trying to overhaul everything at once.
How to support weight maintenance
When keeping weight on is difficult, the most useful approach is often to increase the energy and protein content of what is already being tolerated.
Food-first ideas
- Add butter, cream, cheese, yoghurt, milk powder or olive oil to foods where suitable.
- Choose full-fat products rather than “diet” versions if weight loss is a concern.
- Add grated cheese to soup, mashed potato, scrambled eggs or vegetables.
- Make porridge with milk rather than water.
- Keep easy snacks available, such as yoghurts, cheese and crackers, peanut butter, hummus, custard, rice pudding or milky desserts.
Protein matters
Protein helps preserve muscle. Good sources include:
- milk, yoghurt and cheese
- eggs
- meat, fish and poultry if tolerated
- beans, lentils and other pulses
- nut butters where suitable
Oral nutritional supplements
When food alone is not enough, a doctor or dietitian may suggest oral nutritional supplements. These are often used between meals rather than instead of meals. They can be particularly helpful when appetite is low or meal size is very limited.
In general UK nutrition practice, a “food first” approach is usually tried first where appropriate, but oral nutritional supplements are commonly used when someone is at higher risk of malnutrition or is unable to meet needs from food alone.
Food and medicine issues to remember
Food and medicine can interact in two main ways.
1. Medicines can affect eating
Antifungal treatment and other medicines may contribute to:
- nausea
- indigestion
- altered taste
- poor appetite
- bowel upset
If these symptoms started after a medicine was introduced or changed, it is worth discussing that with the prescribing team.
2. Food can affect medicines
Some antifungal medicines have specific instructions about when to take them in relation to food. For example:
- Itraconazole capsules are generally taken with or just after food, while itraconazole liquid is generally taken on an empty stomach.
- Voriconazole is usually taken on an empty stomach.
- Some medicines also have important interactions with antacids or acid-suppressing medicines.
Because formulations differ, and because other medicines may also interact, patients should follow the instructions they have been given for their exact preparation and check with a pharmacist or clinical team if unsure.
Grapefruit and other food interactions: some medicines have clinically important food interactions. Patients should check current advice for each medicine rather than relying on memory or online generalisations.
Common diet myths
Dairy always makes mucus worse
This is a very common belief. Current evidence does not show that dairy routinely increases lung mucus production for most people. Some people do notice a thicker mouth or throat feeling after milk, which may relate to texture rather than extra mucus. If dairy is well tolerated, it can be a useful source of calories and protein.
Sugar “feeds” aspergillosis, so it should be cut out completely
Patients often hear this online, but strict self-imposed restriction can be more harmful than helpful when someone is already struggling to maintain intake. For many patients with weight loss, the immediate nutritional priority is adequate calories and protein, not aggressive dietary exclusion.
There is a special anti-aspergillosis diet
There is no widely accepted specialist diet that treats aspergillosis itself. In routine practice, nutrition advice usually focuses on preventing or treating malnutrition, easing symptoms and managing medicine-related issues.
If eating is difficult, I should just avoid more foods
Sometimes a food really is hard to tolerate, but repeated restriction can shrink the diet too far. Often it is more useful to ask, “Can this be made easier to eat?” rather than “Should I cut this out altogether?”
When to seek medical help
Patients should speak to their doctor, specialist team or another qualified healthcare professional if they have any of the following:
- ongoing unplanned weight loss
- clothes, rings or dentures becoming looser
- difficulty eating most days
- a very narrow diet with only a few “safe” foods
- persistent nausea, reflux, bloating or abdominal discomfort
- increasing weakness or fatigue
- concerns that medicines are worsening appetite or stomach symptoms
It may be appropriate to ask about a dietitian referral, especially if intake has been poor for some time or there are signs of malnutrition.
Seek urgent medical advice if:
- food or fluids are being kept down very poorly
- there are signs of dehydration
- weight loss is rapid or severe
- pain, vomiting, swallowing difficulty or other worrying symptoms are developing
Common questions
Should I force myself to eat full meals?
Usually not. If full meals are consistently overwhelming, smaller and more frequent intake is often more successful.
Are liquid calories “cheating”?
No. For some people, nourishing drinks are one of the most practical ways to protect weight and strength.
What if I only manage a few foods?
That is still worth discussing. A restricted diet may be understandable, but it can increase nutritional risk over time.
What if dairy feels unpleasant?
Individual experience matters. If a food clearly feels uncomfortable, it may help to try alternatives or use smaller amounts in different forms. But many people do not need to exclude dairy automatically.
Could reflux be making my cough worse?
Yes, it can in some people. Reflux can irritate the upper airway and may contribute to cough or discomfort around meals.
When to seek medical advice
Ask for medical advice if you are losing weight, struggling to eat most days, developing a very restricted diet, or think nausea, reflux or medication side effects are affecting your intake. Ask urgently if you are becoming dehydrated, vomiting repeatedly, or your intake has become extremely poor.
Author and review information
Prepared for: aspergillosis.org
Purpose: general educational information for patients and non-specialists
Review note: Because medicine instructions can change between formulations and brands, patients should always check the current advice supplied with their own prescription and confirm uncertainties with a pharmacist or clinical team.
References and further reading
- Carter C, Muldoon EG, Kosmidis C. Chronic pulmonary aspergillosis - a guide for the general physician. 2024.
PubMed - Tashiro M, Takazono T, Izumikawa K. Chronic pulmonary aspergillosis: comprehensive insights into epidemiology, diagnosis, treatment, and unresolved challenges. 2024.
Free full text - Roboubi A, et al. Allergic bronchopulmonary aspergillosis. 2023.
PubMed - Sunman B, et al. Current approach in the diagnosis and management of allergic bronchopulmonary aspergillosis in children with cystic fibrosis. 2020.
Free full text - Madhavan V, et al. Malnutrition in allergic bronchopulmonary aspergillosis complicating asthma. 2023.
Free full text - British Dietetic Association. Spotting and treating malnutrition.
BDA resource - BAPEN. Food first / food enrichment.
BAPEN resource - BAPEN / Malnutrition Pathway. Managing malnutrition in COPD.
PDF - NICE. Managing malnutrition in COPD, The Malnutrition Pathway.
NICE shared learning resource - NHS. Heartburn and acid reflux.
NHS advice - Cambridge University Hospitals NHS Foundation Trust. Dietary and lifestyle advice for adults with gastro-oesophageal reflux disease (GORD).
CUH advice - NICE BNF. Itraconazole.
BNF drug monograph - Manchester University NHS Foundation Trust, National Aspergillosis Centre. Patient Information: Itraconazole.
PDF - Manchester University NHS Foundation Trust, National Aspergillosis Centre. Patient Information: Voriconazole.
PDF - Oxford University Hospitals NHS Foundation Trust. Advice about antifungals.
PDF - Balfour-Lynn IM. Milk, mucus and myths. Archives of Disease in Childhood. 2019.
Article - Pinnock CB, Graham NM, Mylvaganam A. Relationship between milk intake and mucus production in adult volunteers challenged with rhinovirus-2. 1990.
PubMed - ASCIA. Milk, mucus and cough.
Patient resource
Diet and Aspergillosis: What Helps, What Doesn’t, and What Matters Most
Last reviewed: 8 April 2026
Many people living with aspergillosis ask whether diet can help “fight” the fungus, reduce symptoms, or improve recovery. This is completely understandable, particularly given the large amount of advice online suggesting that certain foods, supplements, or diets can control fungal disease.
This article explains what current evidence shows, what diet can and cannot do, and where it genuinely matters for people living with aspergillosis.
Core principle: Aspergillus-related disease is driven by what we breathe in and how the body responds — not by what we eat.
Contents
- Key points
- What do we mean by “no evidence”?
- How Aspergillus disease develops
- The role of airways, mucus and lung structure
- Immune response and inflammation
- Can food treat Aspergillus?
- Diet and ABPA (allergic disease)
- Allergy vs infection: why diet is often misunderstood
- Diet and CPA (chronic infection)
- Why nutrition still matters
- Steroids and diet
- Diet and antifungal medication
- Checking food–drug interactions
- Common diet myths (and why they persist)
- A practical, evidence-based approach
- When to seek help
- References
Key points
- There is no diet that treats aspergillosis.
- Aspergillus-related disease develops through inhalation of spores, changes in the lungs, and immune responses.
- Diet does not control Aspergillus growth in the lungs.
- Diet still matters for strength, weight, recovery, and treatment safety.
- Food can affect how medicines are absorbed and metabolised.
- Many popular online diet claims are based on misunderstood science, oversimplification, or marketing.
What do we mean by “no evidence”?
When this article says there is “no evidence” or “no strong evidence”, this does not mean that we are simply waiting for proof to arrive.
In most cases, it means one of two things:
- the idea has been studied and has not been shown to help real patients, or
- there is only laboratory or theoretical evidence, which does not translate into benefit in real-world disease
For example, fungi can grow in sugar-rich laboratory conditions. That does not mean eating sugar feeds Aspergillus in the lungs. The body tightly regulates blood glucose, and lung disease is far more complex than a laboratory culture dish.
Key message: when clinicians say there is “no evidence”, they usually mean an approach is unlikely to work in practice, not that it is a promising treatment that just has not been tested yet.
How Aspergillus disease develops
Aspergillus is a common environmental mould. People are exposed by breathing in microscopic spores from the air. Most people clear these spores without any problem.
Whether disease develops depends on the interaction between:
- the condition of the lungs
- how well mucus is cleared
- the immune response
In healthy lungs, inhaled spores are trapped in mucus, moved out of the airways, and removed by immune cells. When this system is disrupted, Aspergillus may persist or trigger inflammation. This is described in clinical reviews of pulmonary aspergillosis such as Kosmidis & Denning, 2015.
This process takes place in the respiratory system and is driven by inhalation — not diet.
The role of airways, mucus and lung structure
The lungs have several important defence systems. These include mucus, cilia (tiny hair-like structures that move mucus), and immune cells. Together, they help remove inhaled particles and organisms.
In conditions such as asthma, bronchiectasis, chronic obstructive pulmonary disease (COPD), or other chronic lung diseases:
- mucus may become thicker or harder to clear
- airways may be damaged or widened
- normal clearance may be less effective
This can make it easier for Aspergillus to remain in the lungs. In some people this contributes to allergic disease. In others, especially where there is structural damage, it can contribute to chronic infection.
These airway and lung-structure problems are not altered by avoiding particular foods.
Immune response and inflammation
In many people, particularly those with allergic forms of Aspergillus disease, symptoms are driven more by the immune system than by direct tissue invasion from the fungus.
For example, in allergic bronchopulmonary aspergillosis (ABPA), the body mounts an exaggerated allergic response to Aspergillus. This typically involves:
- raised IgE antibodies
- eosinophilic inflammation
- airway swelling and mucus production
Reviews of ABPA describe these immune processes in more detail, including the overlap with asthma and cystic fibrosis-related airway disease, for example Knutsen & Slavin, ABPA review.
These immune pathways are complex and are not controlled by specific foods.
Can food treat Aspergillus?
No specific food has been shown to treat Aspergillus-related lung disease.
You may come across claims about garlic, turmeric, coconut oil, probiotics, herbs, or “anti-fungal” foods. Some of these have shown antifungal effects in laboratory settings, but there is no reliable clinical evidence that eating them improves aspergillosis outcomes.
This is because:
- food is processed in the digestive system, not the lungs
- active compounds may not reach the lungs in useful amounts
- the biology of lung disease is much more complex than simple fungal growth in a dish
Diet can support the body, but it is not a treatment for Aspergillus disease.
Diet and ABPA (allergic disease)
In ABPA, the main problem is an allergic or immune reaction to Aspergillus. Diet does not switch that reaction on or off.
That means:
- food does not “feed” ABPA
- there is no evidence that an “anti-fungal diet” controls ABPA
- restrictive diets do not treat the underlying immune process
However, diet can become more important because many patients with ABPA are treated with prednisolone or other corticosteroids. These medicines can affect appetite, weight, blood sugar, and bone health. NHS information on prednisolone describes common effects such as weight gain, increased appetite, and longer-term bone risks: NHS Prednisolone guidance.
So in ABPA, diet often matters more in relation to treatment effects than in relation to the fungal trigger itself.
Allergy vs infection: why diet is often misunderstood
Many patients understandably ask whether a certain food might be “triggering” symptoms. This can be confusing because aspergillosis includes both allergic and infectious forms.
What matters here is the route of exposure:
- Aspergillus-related lung disease is driven by inhaled spores
- food enters the body through the digestive tract
Symptoms that seem to occur after eating may actually relate to:
- acid reflux
- throat irritation
- airway sensitivity
- coincidental fluctuation in symptoms
These may be real and troublesome, but they are not the same thing as diet directly driving Aspergillus disease.
Key message: Aspergillus-related lung symptoms are driven by what you breathe in and how your immune system responds — not by what you eat.
Diet and CPA (chronic infection)
Chronic pulmonary aspergillosis (CPA) is different from ABPA. CPA tends to occur in lungs that are already damaged or structurally abnormal, for example after tuberculosis, with bronchiectasis, or with COPD.
In CPA, the key issue is not food intake but the presence of abnormal lung tissue where Aspergillus can persist. This broader clinical picture is outlined in reviews such as Kosmidis & Denning, 2015.
Diet does not alter fungal growth directly, but it can matter because some people with CPA experience:
- weight loss
- fatigue
- low appetite
- reduced physical strength
In CPA, diet is therefore mainly about maintaining strength, resilience, and recovery — not about “starving” the fungus.
Why nutrition still matters
Although diet does not treat Aspergillus directly, nutrition still matters a great deal. Good nutrition supports the whole body, including the respiratory system.
Good nutrition can help support:
- muscle strength, including the muscles used for breathing
- energy levels
- general immune function
- recovery from illness and treatment
Poor nutrition, by contrast, can contribute to:
- tiredness
- lower resilience
- reduced strength
- slower recovery
This is one reason why very restrictive diets can be unhelpful, especially for patients already coping with chronic disease, breathlessness, or weight loss.
Steroids and diet
For patients taking corticosteroids such as prednisolone, diet becomes particularly relevant.
Important issues can include:
- increased appetite
- weight gain
- raised blood sugar
- fluid retention
- longer-term bone health
From a practical point of view, this is one of the strongest reasons to think carefully about diet in aspergillosis care. Here, diet is not being used to treat the fungus; it is helping patients cope with the effects of treatment and maintain overall health.
For longer-term steroid use, adequate calcium intake and attention to bone health may also be important. This is particularly relevant for people already at risk of osteoporosis.
Diet and antifungal medication
The clearest and most important direct link between diet and aspergillosis care is through medication.
Food can affect how medicines are absorbed, broken down, or cleared from the body.
Food–drug interactions
Some foods and drinks affect enzymes in the liver that metabolise drugs. A well-known example is grapefruit, which can interfere with CYP3A4 and change drug levels in the body.
Taking antifungals correctly
Some antifungal medicines are affected by food. For example, voriconazole is usually taken on an empty stomach so that absorption is more reliable. This is described in professional guidance such as the British National Formulary (BNF).
Supplements and herbal products
Supplements are often marketed as “natural”, but they can still interact with prescription medicines. Some herbal products may alter drug metabolism and therefore affect antifungal treatment.
Key message: diet rarely affects Aspergillus directly, but it can be very important in how your medicines work.
Checking food–drug interactions
Reliable sources for checking medicine and food interactions include:
- British National Formulary (BNF)
- patient information leaflets supplied with medicines
- Patient.info interaction checker
It is also important to understand the limits of specialist tools. Drug-interaction tools designed specifically for antifungals are very useful for drug–drug interactions, but they do not usually include food interactions in a comprehensive way.
The safest source of advice remains your pharmacist or clinical team.
Common diet myths (and why they persist)
Many people with aspergillosis come across strong claims online about diet and fungal disease. These often sound convincing, especially when symptoms are difficult to control and people understandably want something practical they can do.
However, most of these claims are based on misunderstandings of biology, laboratory research taken out of context, or commercial promotion.
Below are some of the most common myths, along with what current evidence suggests.
“Sugar feeds fungus”
This is one of the most common claims.
It comes from the fact that fungi can grow in sugar-rich laboratory conditions. However, this does not reflect what happens in the human body.
- blood sugar is tightly regulated
- Aspergillus in the lungs is not directly exposed to dietary sugar in the way people often imagine
- there are no clinical studies showing that reducing dietary sugar improves aspergillosis outcomes
Why it persists: it sounds intuitive, it is easy to repeat, and it fits with heavily marketed “anti-fungal” diet programmes.
“Milk and dairy increase mucus”
This is a very common concern in respiratory disease generally.
Research does not show that dairy increases mucus production in the lungs. Some people notice a thicker or coated feeling in the mouth and throat after milk, but that is different from producing more mucus in the airways.
- no good evidence of increased lung mucus
- no evidence that dairy worsens Aspergillus disease itself
Why it persists: the mouth and throat sensation after dairy can easily be mistaken for a lung effect.
“Yeast in food causes fungal infection”
Foods such as bread or fermented products may contain yeast, but yeast used in food is not the same thing as Aspergillus.
- food yeast and Aspergillus are different organisms
- Aspergillus-related lung disease is linked to inhalation of environmental spores, not eating yeast-containing foods
Why it persists: the word “fungus” is used broadly, which can blur important differences between very different organisms.
“Avoid foods made with mould”
Some patients are advised online to avoid blue cheese, mushrooms, or other foods associated with moulds.
There is no good evidence that eating these foods changes Aspergillosis in the lungs.
- the digestive and respiratory systems are separate
- food moulds are not the same thing as inhaled environmental Aspergillus exposure
Why it persists: when a disease involves mould, it feels logical to avoid all mould-associated foods, even though the biology does not support that approach.
“Low-carb or ketogenic diets can starve the fungus”
This idea grows out of the “sugar feeds fungus” claim.
However:
- the body keeps glucose within a narrow range
- lung infections are not directly altered by short-term dietary carbohydrate restriction
- there is no clinical evidence that low-carb or ketogenic diets improve aspergillosis outcomes
Why it persists: it sounds more scientific than it is, and it is frequently promoted in wellness and biohacking communities.
“Anti-fungal foods such as garlic, turmeric or coconut oil can treat aspergillosis”
Some of these substances show antifungal activity in laboratory experiments.
That is not the same as treating disease in people. The concentrations used in experiments are often very different from what is achievable through normal eating, and human lung disease is far more complex than a petri dish.
Why it persists: laboratory findings are often presented online as though they were proven clinical treatments.
“Detox diets or cleanses remove fungal infection”
There is no biological mechanism by which detox diets or juice cleanses remove Aspergillus from the lungs.
- the lungs are not “cleansed” through the digestive tract
- there is no clinical evidence supporting detox approaches in aspergillosis
Why it persists: detox language is emotionally appealing, especially when people feel unwell and want a sense of control.
“Candida overgrowth” diets apply to aspergillosis
Many patients come across “anti-Candida” diets and wonder whether the same advice applies to Aspergillus.
These diets often recommend:
- cutting out sugar
- avoiding carbohydrates
- removing yeast-containing foods
- following restrictive “anti-fungal” eating plans
However, these ideas are based on a different organism and a different part of the body.
Candida vs Aspergillus: important differences
- Candida is a yeast commonly found on the skin and in the gut
- Aspergillus is a mould in the environment that is inhaled into the lungs
Aspergillus-related disease such as ABPA or CPA affects the lungs and is driven by inhaled spores, not by changes in the gut.
Do “anti-Candida diets” affect Aspergillus?
There is no clinical evidence that diets designed to reduce Candida:
- affect Aspergillus in the lungs
- reduce allergic responses to Aspergillus
- improve outcomes in aspergillosis
These diets often rely on the same assumptions as other myths, especially the idea that “sugar feeds fungus”. Those assumptions do not fit how Aspergillus lung disease works.
What about the gut microbiome?
There is real scientific interest in the gut microbiome and its role in health. However, there is currently no evidence that changing diet to target gut fungi alters aspergillosis outcomes.
This is an area of research interest, but it is not a basis for dietary treatment at present.
Why these diets can be unhelpful
Restrictive anti-Candida or “anti-fungal” diets can sometimes lead to:
- reduced calorie intake
- weight loss
- nutritional imbalance
- anxiety around food
This can be particularly unhelpful in people with chronic lung disease who need to maintain strength and energy.
Key message: diets designed for “Candida overgrowth” are not relevant to aspergillosis and are not supported by evidence in this context.
“If symptoms improve after changing diet, the diet must be working”
This is a very understandable conclusion, but it can be misleading.
Symptoms in aspergillosis often fluctuate because of:
- natural variation in disease activity
- environmental exposure
- allergy activity
- medication changes
An improvement may happen at the same time as a dietary change without being caused by that change.
Advice from non-mainstream or alternative sources
Many people with long-term or difficult-to-control conditions look beyond standard medical care for additional answers. This is entirely understandable, especially when symptoms are persistent or uncertain.
You may come across advice from practitioners or online sources who describe themselves as offering “functional”, “integrative”, or “alternative” approaches. These often include:
- strict or highly restrictive diets
- “anti-fungal” or “detox” protocols
- long lists of supplements
- tests or diagnoses that are not widely used in NHS practice
Some of this advice may sound detailed or scientific. However, it is important to understand that:
- many of these approaches are not supported by clinical evidence in aspergillosis
- they may be based on theories that do not reflect how lung disease develops
- they are often not part of standard respiratory or infectious disease care
In some cases, following this advice can lead to:
- unnecessary dietary restriction
- weight loss or nutritional problems
- delays in receiving appropriate medical treatment
- confusion about symptoms and diagnosis
This does not mean that all non-mainstream approaches are harmful, but it does mean they should be approached with care.
Key message: if you are considering advice outside standard medical guidance, it is usually helpful to discuss it with your clinical team or pharmacist so it can be considered safely alongside your current treatment.
Overall message: many diet claims are based on ideas that sound plausible but do not reflect how aspergillosis works in the body.
A practical, evidence-based approach
For most people with aspergillosis, the most sensible and evidence-based approach is:
- eat a balanced diet
- maintain weight and strength
- include regular sources of protein
- avoid unnecessarily restrictive diets
- follow medicine-specific instructions carefully
- check food–drug interactions rather than relying on social media advice
Focus on supporting your body and treatment — not trying to treat Aspergillus through diet.
When to seek help
It may be worth asking for extra support if you are experiencing:
- unintentional weight loss
- poor appetite
- difficulty managing steroid-related appetite or weight changes
- concerns about blood sugar or bone health
- questions about food–drug interactions
Pharmacists, GPs, specialist teams, and where appropriate dietitians can all help with these issues.
References
- Kosmidis C, Denning DW. The clinical spectrum of pulmonary aspergillosis. Thorax. 2015.
- Warris A, Bercusson A, Armstrong-James D. Aspergillus colonization and antifungal immunity in cystic fibrosis patients. Med Mycol. 2019.
- Knutsen AP, Slavin RG. Reviews on allergic bronchopulmonary aspergillosis.
- NHS. Prednisolone guidance.
- British National Formulary (BNF).
- Patient.info interaction checker.
This article is for general information and should not replace advice from your own clinical team.
Cystic Fibrosis, CFTR Gene Variants, and Aspergillosis
Last reviewed: 8 April 2026
Some people with aspergillosis are told they have cystic fibrosis (CF), or that they carry a CFTR gene variant. This can be unexpected and may raise concerns about whether this explains their symptoms or diagnosis.
This article explains how cystic fibrosis and CFTR gene variants relate to Aspergillus-related lung disease, what current research shows, and—importantly—what conclusions should not be drawn.
Contents
- Key points
- Important reassurance
- What is cystic fibrosis?
- What is a CFTR gene variant?
- How CFTR affects the lungs
- How Aspergillus behaves in the lungs
- ABPA and cystic fibrosis
- CPA and cystic fibrosis
- Modern CF treatments and Aspergillus
- Does a CFTR variant explain symptoms?
- What should patients take from this?
- When to seek medical advice
- Conclusion
- References
Key points
- Most people with aspergillosis do not have cystic fibrosis.
- Most people with cystic fibrosis do not develop ABPA or CPA.
- ABPA is linked to mucus and immune responses, not just infection.
- CFTR variants may contribute to risk in some people, but are usually only one factor.
- CPA is mainly driven by structural lung damage, not CFTR genetics.
Important reassurance
Most people with aspergillosis do not have cystic fibrosis, and most people with cystic fibrosis do not develop Aspergillus-related disease.
Although these conditions can overlap, they are usually separate. Genetic findings such as CFTR variants should be interpreted carefully and in context.
What is cystic fibrosis?
Cystic fibrosis is a genetic condition caused by changes in the CFTR gene. This gene regulates salt and water movement across cells.
When CFTR function is reduced:
- mucus becomes thick and sticky
- airways are harder to clear
- microorganisms persist more easily
This creates an environment where bacteria and fungi can accumulate over time.
What is a CFTR gene variant?
CFTR variants range from severe mutations (causing cystic fibrosis) to mild or uncertain variants.
Carriers (with one variant):
- are common in the general population
- usually have no symptoms
- may have subtle effects in some cases
These subtle effects may include reduced mucus clearance or increased susceptibility to airway inflammation.
How CFTR affects the lungs
CFTR dysfunction affects the lungs in several key ways:
- Mucus dehydration: mucus becomes thick and difficult to clear
- Impaired clearance: particles and microbes remain in the airways
- Chronic inflammation: immune responses become exaggerated
This combination creates a “retention environment” where inhaled organisms—including Aspergillus—may persist.
How Aspergillus behaves in the lungs
Aspergillus is inhaled by everyone, but its effects vary depending on the lung environment.
- Healthy lungs: spores are cleared
- Impaired clearance: spores may persist
- Sensitive immune system: allergic reactions may develop
- Damaged lungs: chronic infection may develop
This explains why Aspergillus-related disease is diverse and depends heavily on underlying lung conditions.
ABPA and cystic fibrosis
ABPA is an allergic immune reaction to Aspergillus.
It is recognised in cystic fibrosis because:
- mucus retention increases exposure to Aspergillus
- immune responses can be exaggerated
However:
- Many CF patients never develop ABPA
- Most ABPA patients do not have CF
Some studies suggest CFTR variants may increase susceptibility, but this is not consistent across all research.
Key message: ABPA and CF can overlap, but one does not imply the other.
CPA and cystic fibrosis
CPA is a chronic fungal infection that develops in structurally damaged lungs.
The most important risk factor is:
pre-existing lung damage
This includes:
- bronchiectasis
- previous tuberculosis
- COPD
Cystic fibrosis can lead to bronchiectasis, and therefore indirectly increase CPA risk.
However:
- CPA is rarely driven directly by CFTR genetics
- most CPA patients do not have CF
Key message: CPA is primarily a disease of lung structure, not genetics.
Modern CF treatments and Aspergillus
CFTR modulators (such as elexacaftor/tezacaftor/ivacaftor) have transformed CF care.
They:
- improve CFTR function
- thin mucus
- improve clearance
Studies suggest:
- reduced Aspergillus detection in some patients
- fewer ABPA exacerbations in some cases
However:
- ABPA still occurs
- existing lung damage remains
- immune responses are not fully corrected
Overall: these therapies improve risk but do not eliminate Aspergillus-related disease.
Does a CFTR variant explain symptoms?
No single factor explains complex lung disease.
Symptoms may result from:
- underlying lung disease
- infection
- inflammation
- environmental exposure
A CFTR variant may contribute, but is rarely the sole cause.
What should patients take from this?
- CF and CFTR variants can sometimes contribute
- ABPA has the strongest connection
- CPA is mainly driven by lung damage
- Most patients with aspergillosis do not have CF
When to seek medical advice
Seek advice if symptoms worsen, change, or include coughing up blood, fever, or chest pain.
Conclusion
Cystic fibrosis and CFTR gene variants can play a role in some patients with Aspergillus-related lung disease, particularly where mucus clearance is affected. However, they should not be overemphasised. In most cases, they are just one part of a broader clinical picture involving lung structure, immune response, and environmental exposure.
References
- Miller PW et al. (1996)
- Marchand E et al. (2001)
- Eaton TE et al. (2002)
- Agarwal R et al. (2012)
- Chaudhary N et al. (2012)
- Warris A et al. (2019)
- Gamaletsou MN et al. (2018)
- Kosmidis C & Denning DW (2015)
- Moldoveanu B et al. (2021)
- Malik HS et al. (2025)
- Zeng C et al. (2025)
- Bendixen MP et al. (2025)
This article is for general information and does not replace advice from your clinical team.