Managing Life with Haemoptysis
Managing life with haemoptysis — especially when it's recurrent, low-volume, or threatening to recur — can be physically and emotionally exhausting. Whether due to CPA, ABPA, bronchiectasis, aspergillus bronchitis, or other underlying lung conditions, the goal is to minimise triggers, support healing, and maintain safety without living in constant fear.
Here’s a comprehensive, practical guide to managing haemoptysis during recovery or periods of fragility:
🔴 Understanding the Risk
Haemoptysis (coughing up blood) can range from:
-
Minor (streaks in mucus)
-
Moderate (5–50ml)
-
Massive or life-threatening (>200–600ml in 24h — a medical emergency)
If you're in a recovery phase, you may be:
-
Post-bleed but still inflamed
-
Dealing with recurrent trickles
-
Worried about provoking a bleed due to fragile blood vessels or fungal activity
✅ Core Management Goals
| Goal | How to Achieve It |
|---|---|
| Prevent rebleeding | Avoid straining, irritating airways, or increasing pressure |
| Allow fragile vessels to heal | Stay well-hydrated, avoid airway trauma, reduce inflammation or infection |
| Identify and treat causes | Maintain antifungal, antibiotic or anti-inflammatory treatment as prescribed |
| Stay calm during symptoms | Know how to position yourself and who to contact |
| Keep life going gently | Pace activity, prioritise rest, manage anxiety without isolation |
🔹 1. Activity & Positioning: How to Move Safely
-
Avoid intense exercise, heavy lifting, straining (including on the toilet).
-
Keep your head elevated when sleeping (2 pillows or wedge).
-
If coughing blood:
-
Sit upright or lean slightly forward (don’t lie flat).
-
Lie on the side that’s bleeding (if known) — this protects the better lung.
-
🔹 2. Breath & Cough Management
-
Cough suppression may help reduce vessel trauma:
-
Use warm steam or gentle hydration first.
-
Use prescribed suppressants only if safe (some conditions need mucus clearance).
-
-
Huffing can be gentler than coughing.
-
Avoid dry air — use a humidifier, nasal rinses, or saltwater gargles.
🔹 3. Medication Adherence
-
Antifungals (e.g., voriconazole, itraconazole): Maintain strict levels.
-
Steroids (if prescribed): Taper cautiously under supervision.
-
Antibiotics or macrolides: Prevent secondary infection.
-
Tranexamic acid: Sometimes used short-term to reduce bleeding risk (under guidance).
🔹 4. Environmental & Lifestyle Support
-
Avoid dust, smoke, aerosols, strong odours, and temperature extremes.
-
Wear a mask when gardening, cleaning, or in crowded spaces.
-
Avoid alcohol and anti-inflammatory meds (NSAIDs) unless cleared.
-
Maintain gentle hydration, warm teas, and soothing soups to support healing.
🔹 5. Psychological Support: Managing Fear and Anxiety
-
It’s normal to fear rebleeding — but hypervigilance can increase stress and airway irritation.
-
Use mindfulness or grounding during panic (see above).
-
Talk with a support group or therapist if fear is affecting sleep or daily life.
-
Ask for a written plan from your medical team: “What to do if it happens again.”
🔹 6. When to Seek Help
Call your medical team or go to A&E if:
-
Bleeding increases or becomes bright red and continuous
-
You cough up >1 tablespoon of blood
-
You feel faint, breathless, or distressed
-
Blood is mixed with froth, or you feel it rising in your throat
📦 Preparedness Tips
-
Keep a rescue plan printed or saved on your phone.
-
Store clean cloths or tissues, bottled water, and calming items near where you rest.
-
Carry a medical ID or information card if you’re going out alone.
🧘♀️ Living Well While Letting It Heal
-
Maintain light, slow routines – a little movement, some fresh air, safe distraction.
-
Avoid cough triggers like strong smells or cold air.
-
Use the time to focus on recovery, build strength gradually, and connect with others.
🧠 What Is Mindfulness? Can it Help Living With Aspergillosis?
Mindfulness can be a powerful support tool for people living with aspergillosis, whether it's CPA, ABPA, SAFS, or related conditions like bronchiectasis or asthma. It doesn’t replace medical treatment, but it can significantly help with coping, symptom awareness, and emotional wellbeing.
Below is a breakdown of how mindfulness practices may benefit people with aspergillosis:
Mindfulness means paying attention, on purpose, in the present moment, without judgment. It involves noticing your thoughts, feelings, and physical sensations — without getting caught up in them or trying to change them immediately.
This can be done through:
-
Breath awareness
-
Body scans
-
Guided meditation
-
Mindful movement (e.g. yoga, tai chi)
-
Mindful walking or eating
🌿 How Can Mindfulness Help with Aspergillosis?
| Challenge | How Mindfulness Helps |
|---|---|
| Breathlessness / coughing fits | Helps calm panic and reduce over-breathing; improves awareness of breath patterns |
| Medication side effects (e.g. voriconazole) | Reduces anxiety and fear responses; helps observe sensations without spiralling |
| Chronic fatigue / brain fog | Supports pacing and acceptance; improves focus and attention |
| Pain or chest discomfort | Helps reduce distress by observing pain without fighting it (used in pain management) |
| Anxiety or health fear | Teaches how to sit with fear without letting it dominate thinking |
| Frustration with slow progress | Encourages self-kindness and reduces reactivity to setbacks |
| Isolation / low mood | Builds resilience and reduces rumination by shifting focus to what is happening now |
| Poor sleep | Calms a racing mind before bedtime |
🧘♀️ Simple Mindfulness Practices for Patients
1. Breath Awareness (1–2 minutes)
Focus gently on the in-breath and out-breath. When your mind wanders (and it will), gently bring it back to the breath.
Use when: You feel overwhelmed, panicked, or need to slow down.
2. Body Scan (5–10 minutes)
Lie or sit down, and slowly move your attention through your body from feet to head. Notice sensations — tension, warmth, numbness — without judgment.
Use when: Resting, falling asleep, or managing pain or fatigue.
3. Mindfulness in Everyday Tasks
Choose a task (e.g., washing up, brushing teeth, folding clothes). Do it slowly and notice the sensations, smells, sounds — staying fully present.
Use when: You feel disconnected or anxious during the day.
4. RAIN Method for Anxiety
-
Recognise what’s happening
-
Allow the experience to be there
-
Investigate with curiosity (Where in my body do I feel this?)
-
Nurture (What do I need right now?)
Use when: You’re feeling a strong emotional response (fear, frustration, sadness).
💬 What Patients Have Said
“It doesn’t stop the coughing, but I panic less when I feel it coming on.”
“I used to fear every twinge in my chest. Mindfulness gave me space before I reacted.”
“When I feel the visual side effects starting, I breathe and tell myself: just observe, it will pass.”
📱 Resources and Apps (Many Are Free)
-
Headspace (good for beginners)
-
Insight Timer (lots of free guided meditations)
-
Calm (for sleep and anxiety)
-
Breathworks (mindfulness for health conditions)
-
Smiling Mind (free and evidence-based)
⚠️ Important Notes
-
Mindfulness is a skill — it takes time and practice. Start small.
-
It can feel uncomfortable at first, especially if you're used to avoiding difficult thoughts or sensations.
-
If you’ve experienced trauma or distressing symptoms, guided mindfulness with a therapist may be safer.
Grounding Techniques for Aspergillosis: Coping with Panic, Anxiety, and Sensory Overload
Living with aspergillosis — whether it’s ABPA, CPA, SAFS, or another form — can be physically and emotionally overwhelming. From managing symptoms like breathlessness or cough, to coping with powerful medications (like voriconazole), or dealing with uncertainty and isolation, it’s not unusual to feel anxious, panicked, or disconnected.
Grounding techniques are simple, effective strategies that help you regain a sense of calm, control, and presence, even in the middle of a crisis.
🧭 What Are Grounding Techniques?
Grounding techniques are ways to anchor yourself in the present moment, especially when your mind or body feels out of control. They use your senses, movement, and breath to shift focus away from distressing thoughts, frightening physical sensations, or overwhelming emotion.
They are widely used by people living with:
-
Chronic illness
-
Anxiety or panic
-
Medication side effects (e.g. visual disturbances from voriconazole)
-
Post-traumatic stress
-
Neurological symptoms or dissociation
🩺 When Might a Person with Aspergillosis Use Grounding?
| Situation | How Grounding Can Help |
|---|---|
| Visual disturbances from medications like voriconazole | Reduce fear, reconnect with reality |
| Panic or anxiety about symptoms or breathlessness | Calm racing thoughts and slow the body’s stress response |
| Overstimulation in hospitals or crowded places | Help regulate sensory overload |
| Before or after medical appointments | Reduce anticipatory anxiety or recover afterward |
| During flare-ups, pain, or coughing episodes | Stay focused, steady, and more in control |
| While trying to sleep | Settle a restless mind or racing heart |
| When feeling isolated, overwhelmed, or low | Feel more present and safe in the body and environment |
✅ Simple Grounding Techniques to Try
🔹 1. The 5-4-3-2-1 Technique (Sensory Anchoring)
Name:
5 things you can see
4 things you can touch
3 things you can hear
2 things you can smell
1 thing you can taste
When to use: During a visual disturbance, panic, or if you feel disconnected.
🔹 2. Box Breathing
Inhale – 4 seconds
Hold – 4 seconds
Exhale – 4 seconds
Hold – 4 seconds
When to use: During a flare-up, medication side effect, or while waiting in a stressful situation (e.g., hospital, travel, scans).
🔹 3. Cold Sensation Reset
-
Hold an ice cube
-
Splash cold water on your face
-
Use a cooling gel pack on your hands or wrists
When to use: To snap out of panic or rapid heart rate, especially if you feel faint or detached.
🔹 4. Movement and Pressure
-
Press your feet firmly into the floor
-
Grip the arm of a chair or ball your fists and release
-
Walk slowly, focusing on each step
When to use: When anxious, overwhelmed, or before a test, clinic visit, or treatment.
🔹 5. Safe Words & Orientation
Say aloud:
“I’m [your name].
I’m in [location].
It’s [day/time].
I feel this way because [e.g. the medication is affecting me], but it will pass.”
When to use: After waking up anxious, during episodes of confusion, visual disturbance, or feeling “spaced out.”
🔹 6. Grounding Object or Support Contact
-
Carry a smooth stone, photo, textured card, or essential oil roller.
-
Call or message someone you trust.
-
Look at a comforting image or mantra you keep nearby.
When to use: When you need emotional reassurance or support — especially after a scary experience.
💡 A Note on Voriconazole Visual Effects
Visual side effects from voriconazole (e.g. flashing lights, color changes, or visual trails) usually:
-
Start within 30 min to 2 hours of a dose
-
Last 2 to 6 hours
-
Fade over time or after stopping the drug
Grounding techniques can’t stop the visual changes but can help you stay calm and reduce the fear or panic response they often trigger.
If the symptoms become unmanageable, let your clinical team know — they may adjust your dose or change the medication.
💬 Final Thoughts
Grounding techniques are safe, non-invasive, and free. They don’t cure symptoms but can help you cope more confidently, especially when dealing with the emotional and sensory impact of life with aspergillosis.
Many patients find it helpful to practice them regularly, even when they feel fine — so they’re easier to use in a crisis.
Visual Disturbances on Voriconazole: What’s Happening and Why It Can Feel So Scary
If you're experiencing frightening or panicked feelings due to visual disturbances while taking voriconazole, you're not alone — and there are reasons behind both the symptoms and your emotional response.
🧠 What Causes Visual Disturbances with Voriconazole?
-
Voriconazole crosses the blood–brain barrier and affects the central nervous system (CNS).
-
It interacts with retinal photoreceptors (especially rods), which may explain:
-
Lights seeming too bright or flickering.
-
Halos, flashes, or color distortions.
-
Visual “trails” following movement.
-
-
Although not fully understood, it may involve:
-
Temporary interference with visual signal processing in the brain and retina.
-
Altered neurotransmitter activity or enhanced retinal light sensitivity.
-
⏱️ When Do These Disturbances Happen and How Long Do They Last?
-
Onset: Symptoms typically begin 30 minutes to 2 hours after a dose.
-
Duration: They usually last 2 to 6 hours, fading as the drug is processed.
-
Resolution: Most people find the effects wear off before the next dose.
-
Over time, even if you stay on the drug, your body often adapts, and the symptoms lessen or disappear entirely within a few days to weeks.
😰 Why Do They Feel So Scary?
-
The effects are sudden and intense, often catching people off guard.
-
Our brains rely on visual input for safety — when this gets disrupted, it can trigger anxiety or panic.
-
If you live with anxiety or take other medications, your brain may amplify the sense of threat, even though the effect is temporary.
-
Descriptions like “psychedelic” or “derealised” are common — which understandably causes distress.
🩺 What Can You Do?
-
Talk to your medical team. They may:
-
Check voriconazole blood levels.
-
Reduce the dose or change timing.
-
Switch to an alternative antifungal if needed.
-
-
Avoid driving at night or doing visually demanding tasks until things settle.
-
If anxiety is a problem:
-
Try grounding techniques (e.g. breath control, sound orientation).
-
Ask your doctor whether temporary support (like anti-anxiety medication or CBT) might help.
-
-
Some patients find taking voriconazole in the evening lets them sleep through the worst of it — but check this with your prescriber first.
🔔 When to Get Urgent Help
If symptoms last unusually long, worsen over time, or include confusion, agitation, or hallucinations, contact your healthcare provider. These may be signs of CNS toxicity, especially if blood levels are high.
🧠 Why Some Medications Can't Be Prescribed by GPs

In the UK, the NHS uses a tiered prescribing system that sometimes prevents GPs from prescribing certain medications, even if those medicines are available elsewhere in the NHS.
Here’s a clear explanation of how and why this happens:
🔒 1. Shared Care or Specialist-Only Medications
Some medicines are designated as “specialist-only” or “shared care” treatments. This means:
-
GPs are not authorised to initiate them.
-
In some cases, they can continue a prescription once a specialist starts it — but only if a formal shared care agreement is in place.
Examples include:
-
Biologics for asthma, ABPA, or autoimmune disease
-
High-risk antifungals like voriconazole or posaconazole
-
Certain cancer, transplant, or hormone drugs
This system ensures that:
-
The medication is closely monitored by someone with specialist knowledge
-
Risks like interactions, side effects, and required blood tests are safely managed
📜 2. Local Prescribing Formularies
Each NHS Integrated Care Board (ICB) or local NHS Trust maintains a formulary — a list of medicines approved for use in that area.
-
If a medicine isn't on the local formulary, the GP may be unable to prescribe it, even if NICE (the National Institute for Health and Care Excellence) says it's effective.
-
These decisions are based on local budget priorities, agreements with hospitals, and clinical capacity.
💷 3. Cost Controls and Prior Approvals
Some medications are expensive or highly specialised, and require:
-
Prior approval by a funding panel
-
A hospital-based consultant to apply for and justify the treatment
GPs usually cannot access these approval pathways directly.
⚠️ 4. Liability and Risk
Even if a GP understands the condition, they may not have:
-
Access to monitoring protocols
-
Up-to-date knowledge of rare drug interactions or side effects
-
The ability to interpret complex blood results needed for safe prescribing
For legal and safety reasons, GPs must follow guidance from their local ICB or NHS England on what they can and can’t prescribe.
✅ What Patients Can Do
-
Ask the hospital team if the medication can be prescribed under shared care, and whether your GP has agreed to it.
-
Ask your GP to request guidance from the local medicines management team.
-
Request a hospital prescription if urgent — but note this often requires collection from hospital pharmacies.
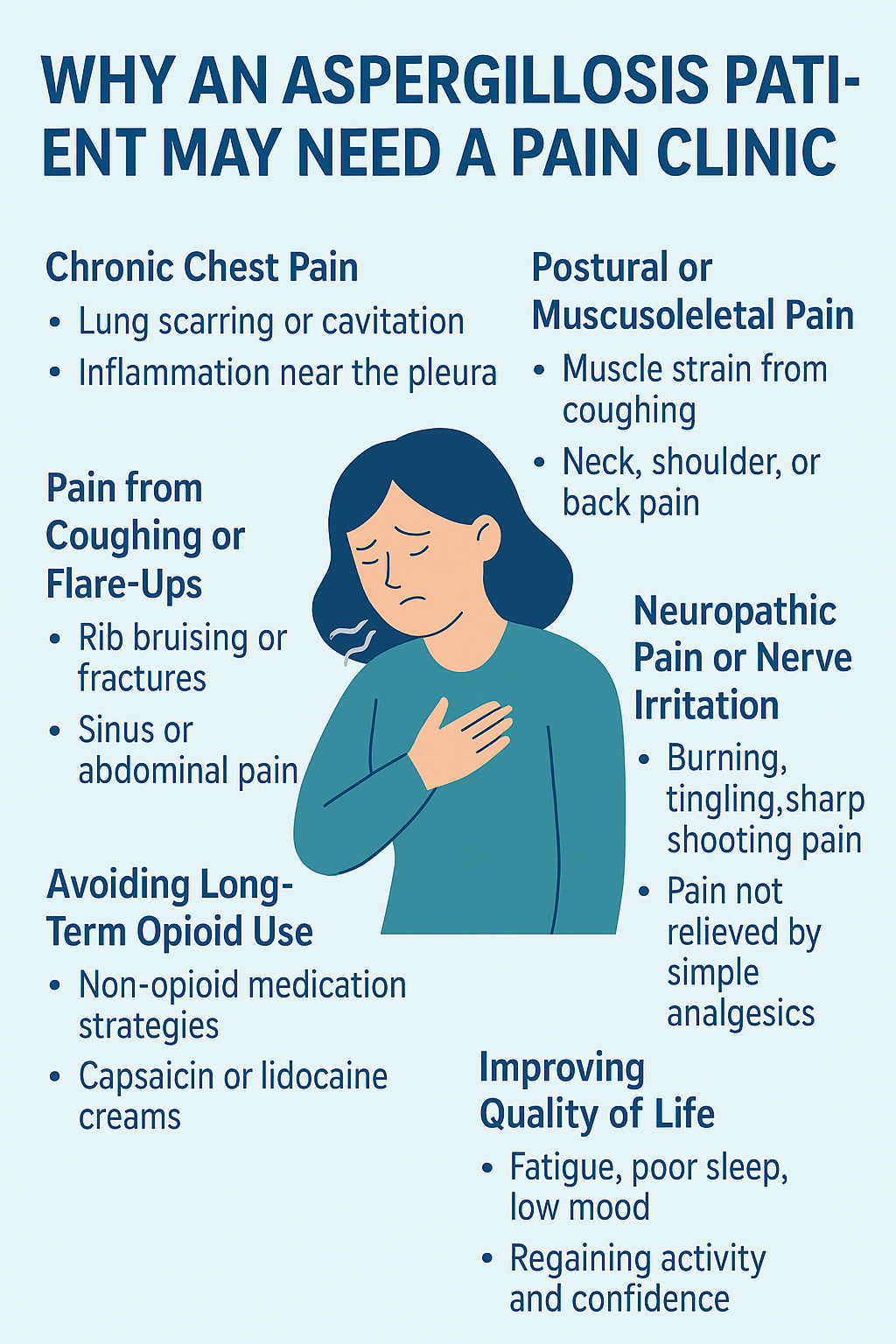
🩺 Why an Aspergillosis Patient May Need a Pain Clinic
Chronic Chest Pain
-
Some patients with CPA, especially those with lung scarring, cavitation, or aspergillomas, develop persistent chest wall pain.
-
This can be caused by:
-
Chronic inflammation or infection near the pleura (lining of the lungs)
-
Pressure or stretching from damaged lung tissue
-
Muscle strain from chronic coughing
-
-
A pain clinic can assess neuropathic pain and offer non-opioid or low-dose medication strategies.
2. Postural or Musculoskeletal Pain
-
Long-term breathing difficulties can change how patients sit, stand, or move, leading to:
-
Neck, shoulder, or back pain
-
Costochondritis (pain around rib joints)
-
-
Pain clinics often include physiotherapists and occupational therapists who can help correct posture and reduce strain.
3. Pain from Coughing or Flare-Ups
-
Recurrent coughing fits in ABPA or CPA can lead to:
-
Rib bruising or even fractures
-
Abdominal muscle pain
-
Headaches and facial pain from sinus pressure
-
-
Clinics can help identify safer ways to manage flare-ups, including breathing strategies and adjunct treatments.
4. Neuropathic Pain or Nerve Irritation
-
Invasive aspergillosis (or surgical interventions for aspergilloma) can affect surrounding nerves, leading to:
-
Burning, tingling, or sharp shooting pain
-
Pain unresponsive to simple analgesics
-
-
Pain specialists may use gabapentinoids, tricyclics, or even nerve blocks to reduce symptoms.
5. Avoiding Long-Term Opioid Use
-
Pain clinics can help avoid or reduce opioid dependence by offering:
-
Safer, evidence-based medication options
-
Capsaicin creams, lidocaine patches, or infusions
-
Psychological support for the emotional toll of chronic pain
-
6. Improving Quality of Life
-
Chronic pain can lead to fatigue, poor sleep, low mood, and reduced activity.
-
Pain clinics work holistically, often involving:
-
Clinical psychologists for CBT or ACT (pain-focused therapy)
-
Personalised pacing plans
-
Tools for managing flare-ups and regaining confidence
-
✅ Summary:
Pain is real and common in aspergillosis — but often under-recognised. A referral to a specialist pain clinic offers a structured, supportive, and multidisciplinary approach to relief.
Attending an NHS pain clinic can offer several important advantages for people living with aspergillosis-related pain, as well as other long-term or complex pain conditions. GPs often have limited tools, time, and prescribing freedom — but pain clinics bring together specialist teams and a much broader range of options.
✅ Advantages of Attending an NHS Pain Clinic
1. Multidisciplinary Care
Pain clinics usually involve:
-
Pain consultants (anaesthetists or neurologists)
-
Specialist nurses
-
Physiotherapists
-
Clinical psychologists
This team approach helps tackle pain from multiple angles — physical, emotional, and functional.
2. Access to a Wider Range of Treatments
Pain clinics can offer things your GP usually can’t prescribe or organise, such as:
-
Topical capsaicin or lidocaine plasters
-
Nerve blocks or steroid injections
-
Low-dose ketamine or lidocaine infusions (in hospital setting)
-
Medications like duloxetine, amitriptyline, or low-dose opioids used carefully
-
Neuromodulation techniques (e.g. TENS, PENS, spinal cord stimulators in some cases)
3. Safer Use of Medication
Specialists understand how to balance benefits and risks of pain medications — including when to use opioids, and how to minimise side effects or dependence.
They can also help you taper safely if you've been on pain meds long-term.
4. Improved Quality of Life
Pain clinics often focus on function over cure — helping you:
-
Sleep better
-
Move more confidently
-
Reduce pain flare-ups
-
Return to work or hobbies
-
Break the cycle of stress, pain, and fatigue
5. Psychological Support (Optional, but Useful)
Chronic pain is exhausting — emotionally as well as physically. Pain clinics may offer:
-
CBT for pain
-
Mindfulness or ACT (Acceptance and Commitment Therapy)
-
Support with coping, pacing, and flare-up planning
This isn’t about “it’s all in your head” — it’s about helping your brain and body deal with ongoing pain more effectively.
6. Validation and Advocacy
Just being believed and supported by a pain specialist can be a huge relief. They can also:
-
Write to your GP with specialist recommendations
-
Support benefits applications or workplace adjustments
-
Help you navigate complex diagnoses like aspergillosis + fibromyalgia, or lung disease + neuropathic pain
📍 When Should You Ask for a Referral?
-
If pain is lasting more than 3 months
-
If it’s affecting your daily function or mental wellbeing
-
If you're relying on medications that aren’t working or cause side effects
-
If you want to explore non-drug options
🩸 Understanding Blood Tests for Aspergillosis: A Patient Guide

If you’ve been told you have ABPA, CPA, or another form of aspergillosis, your doctors may run several blood tests. These tests help confirm the diagnosis, guide treatment, and monitor your progress.
Please remember that blood tests only form part of the process of diagnosing and managing aspergillosis - scans, case history, symptoms and more are also essential parts of a doctor's reasoning. To get a complete picture of your diagnosis, we need all of the parts.
Here’s a breakdown of what each test is, why it’s done, and what it means:
1. Total IgE (Immunoglobulin E)
🧪 What it is: A measure of all the allergy-related antibodies in your blood.
📌 Why it's used:
-
In ABPA, total IgE is usually very high — often above 1,000 IU/mL.
-
Doctors use it to help diagnose ABPA and then to monitor flare-ups or improvements.
💡 What it tells you:
-
High IgE suggests an allergic response, often to Aspergillus.
-
A fall in IgE after treatment often shows you're getting better.
-
A sudden rise might mean a flare-up.
2. Aspergillus-specific IgE
🧪 What it is: A test that looks for allergy antibodies targeting Aspergillus fumigatus.
📌 Why it's used:
-
Helps confirm whether your immune system is reacting to Aspergillus.
-
It’s part of the diagnosis for ABPA and SAFS (Severe Asthma with Fungal Sensitisation).
💡 What it tells you:
-
A positive result means you are sensitised (allergic) to Aspergillus.
-
It doesn’t prove infection — just allergy.
3. Aspergillus-specific IgG
🧪 What it is: A test for long-term antibody response to Aspergillus.
📌 Why it's used:
-
Important in diagnosing Chronic Pulmonary Aspergillosis (CPA).
-
Also used in Aspergillus bronchitis.
💡 What it tells you:
-
High IgG means your immune system has been exposed to Aspergillus over time, likely indicating long-term infection.
-
It’s not an allergy test — it looks for signs of chronic infection or colonisation.
4. Aspergillus precipitins (Immunodiffusion or counter-immunoelectrophoresis)
🧪 What it is: An older test to detect antibodies to Aspergillus proteins.
📌 Why it's used:
-
Sometimes used in CPA or fungal ball (aspergilloma) diagnosis.
-
Less sensitive than IgG ELISA but still used in some labs.
💡 What it tells you:
-
A positive test supports the diagnosis of chronic infection.
5. Eosinophil Count
🧪 What it is: A blood count of a type of white cell linked to allergy and inflammation.
📌 Why it's used:
-
In ABPA, eosinophils are often elevated, especially during flares.
-
It helps show how much inflammation is present.
💡 What it tells you:
-
High eosinophils support a diagnosis of allergic inflammation.
6. Galactomannan (in blood or BAL fluid)
🧪 What it is: A test for fungal cell wall fragments released by Aspergillus, useful when detecting the patient's immune response to infection is limited.
📌 Why it's used:
-
Mainly used in hospitals to detect invasive aspergillosis, especially in people with weakened immune systems.
💡 What it tells you:
-
A positive result may suggest active infection — but false positives can occur.
7. Beta-D-Glucan (BDG)
🧪 What it is: A general marker of fungal infection in the bloodstream.
📌 Why it's used:
-
Used to detect invasive fungal infections, especially in ICU patients.
💡 What it tells you:
-
Not specific to Aspergillus, but may support the diagnosis of serious fungal disease.
🧭 Putting It All Together
Different types of aspergillosis need different combinations of tests:
| Condition | Useful Tests |
|---|---|
| ABPA | Total IgE, Aspergillus-specific IgE, eosinophils |
| SAFS | Aspergillus-specific IgE only |
| CPA | Aspergillus-specific IgG, precipitins, imaging |
| Aspergillus bronchitis | Aspergillus IgG, culture, sometimes IgE |
| Invasive Aspergillosis | Galactomannan, Beta-D-Glucan, CT scan, biopsy (in hospital settings) |
🗨️ Questions to Ask Your Doctor
-
What type of aspergillosis do I have?
-
Which tests are being used to monitor my condition?
-
Should I expect these results to go up and down?
-
What symptoms should I report if things change?
Climate Change: What it Means for People with Aspergillosis.
The recent study here in Manchester and elsewhere suggested that as the climate warms, there is evidence that fungal pathogens will be able to set up home in new areas of the world, increasing the risk of, eg, aspergillosis. Naturally, there has been some alarm at this news from current aspergillosis patients. Are they more at risk and what can be done to protect them?
🌍 Climate Change and Fungal Risk in the UK: What You Need to Know
The study looked at how fungal pathogens like Aspergillus fumigatus may spread over the next 70 years due to climate change. While this sounds alarming, let’s break it down — especially in terms of what it means for those of us in the UK with ABPA, asthma, CPA, or bronchiectasis.
✅ Key Facts
-
Aspergillus fumigatus is already widespread in the UK — in compost, garden soil, air, and dust.
-
The study doesn’t mean the UK will suddenly become “at risk” — rather, the risk may increase due to warmer, drier weather allowing spores to thrive for more of the year.
-
It’s about slow change over decades, not sudden danger.
🌦️ What Might Happen in the UK?
-
More months per year with high airborne spore levels
-
Higher overall concentrations of spores during dry, hot periods
-
Wider spread of antifungal resistance, already being found in urban soil and compost
💚 What We’re Already Doing to Stay Safe
Many in our community are already taking excellent steps to reduce risk, and these are even more important going forward:
🛡️ Wear an FFP2/FFP3 mask when gardening, composting, or in dusty environments
🌬️ Use HEPA air purifiers indoors
🚿 Shower and change clothes after outdoor work
🌡️ Track weather conditions – avoid dusty or windy days when spores are highest
🧪 Ask your doctor about resistance testing if symptoms flare up
🌱 We Can Also Make a Difference
While these changes are long-term, they remind us how connected our health is to our environment. By supporting efforts to cut emissions and reduce global warming, we can help limit the spread of harmful fungi for ourselves and future generations.
If you're seeking reliable resources on current UK efforts to combat climate change, here are some key organisations and initiatives:
🇬🇧 UK Government Initiatives
-
Net Zero by 2050: The UK has a legally binding commitment to achieve net-zero greenhouse gas emissions by 2050. Interim targets include a 68% reduction by 2030 and an 81% reduction by 2035, compared to 1990 levels. Le Monde.fr
-
Department for Energy Security and Net Zero (DESNZ): This department oversees the UK's energy policy and climate change initiatives, including the implementation of the Net Zero Strategy. Wikipedia
-
Public Building Energy Upgrades: The UK government has announced a £630 million investment to improve energy efficiency in public buildings, such as schools and hospitals, by installing solar panels and heat pumps. Reuters
🧭 Independent Oversight and Analysis
-
Climate Change Committee (CCC): An independent body that advises the UK government on emissions targets and reports on progress. The CCC monitors the UK's adaptation to climate change and provides policy recommendations. London.gov.uk
-
UK Parliament Research Briefings: Provides detailed analyses of the UK's climate policies, progress towards net-zero, and sector-specific strategies. House of Commons Library
🌿 Non-Governmental Organizations
-
Greenpeace UK: Offers insights into the UK's climate actions and advocates for stronger environmental policies.
-
Energy Saving Trust: Provides advice and support for individuals and organizations to reduce energy consumption and carbon emissions, including information on grants and energy-saving technologies. Wikipedia
-
UK Green Building Council (UKGBC): Focuses on reducing carbon emissions in the built environment and promotes sustainable construction practices. UKGBC
🏙️ Local and Regional Initiatives
-
Greater London Authority's Climate Action Plan: Outlines strategies for London to become a zero-carbon city, including measures across energy, transport, and waste sectors. London.gov.uk
- Zero Carbon Manchester Manchester.gov.uk
These resources offer comprehensive information on the UK's multifaceted approach to addressing climate change.
Diet Help for Patients with ABPA, Bronchiectasis & Asthma
Living with Allergic Bronchopulmonary Aspergillosis (ABPA), bronchiectasis, and asthma means managing chronic lung inflammation, mucus production, and allergies. While no diet can cure these, the right food choices can help support the lungs, reduce flare-ups, and boost immunity.
It is worth noting that a good balanced diet is important. The foods suggested below are to be included in addition to a good diet, not instead of one.
✅ What to Include
1. Anti-inflammatory foods
- 🍇 Berries, cherries, grapes
- 🐟 Oily fish (salmon, sardines, mackerel – omega-3)
- 🫒 Olive oil, avocado, flaxseed
- 🍵 Green tea and turmeric (with black pepper for absorption - remember to mention that you are taking any food supplement to your doctor )
2. High-antioxidant foods
- 🥦 Broccoli, spinach, kale, sweet potatoes
- 🍅 Tomatoes (rich in lycopene for lung health)
- 🧄 Garlic and onions (natural anti-inflammatories)
3. Good hydration
- 💧 Plenty of water and herbal teas to loosen mucus
- 🍲 Soups and broths can help soothe airways
4. Foods rich in vitamin D, C and zinc
- 🥚 Eggs, fortified cereals, mushrooms (vitamin D)
- 🍊 Oranges, bell peppers, kiwi (vitamin C)
- 🥜 Nuts, seeds, legumes (zinc)
❌ Foods to Avoid or Limit
1. 'Mucus-thickening' foods
- 🧀 Excess dairy (cheese, cream, full-fat milk) may worsen the sensation of mucus for some
- 🍬 Processed sugar (cakes, sweets, fizzy drinks) triggers inflammation
2. Common allergens
- 🌾 Wheat/gluten or dairy can worsen symptoms if you're intolerant
- 🥜 Nuts or soy – avoid if known allergens
3. Pro-inflammatory foods
- 🍟 Fried foods, processed meats (bacon, sausages)
- 🥤 Artificial additives and preservatives
4. Alcohol and caffeine (in excess)
- Can dehydrate and irritate airways
🚫 Watch Out For:
- Mouldy or fermented foods (blue cheese, kimchi, kombucha) can contain fungi and may trigger ABPA if spores are inhaled.
- Compost or mouldy food in the kitchen – avoid exposure due to risk of inhaling fungal spores.
🔁 Bonus Tips
- Eat small meals if large ones trigger breathlessness
- Keep a food-symptom diary to spot personal triggers
- Work with a dietitian if weight loss, fatigue, or food intolerance is an issue
🚫 Foods to Avoid or Limit While Taking Aspergillosis Medications
| ⚠️ Food or Drink | ❓ Why Avoid It |
|---|---|
| Grapefruit and grapefruit juice | Blocks liver enzymes (CYP3A4), increasing drug levels dangerously (especially itraconazole, voriconazole) |
| Seville oranges (marmalade) | Same enzyme-blocking effect as grapefruit |
| High-fat meals (with voriconazole) | May reduce absorption – best taken on an empty stomach |
| Very low-acid foods (with itraconazole capsules) | Needs stomach acid to absorb – avoid taking with antacids, PPIs (e.g. omeprazole), or alkaline meals |
| Alcohol | Increases the risk of liver toxicity, especially with long-term antifungal use |
| Liquorice root (in large amounts) | May raise blood pressure and interact with the metabolism of antifungals |
| St John’s Wort (herbal) | Dramatically reduces antifungal effectiveness by speeding up liver metabolism |
| Supplements with high calcium or magnesium | Can interfere with some oral suspensions or acid levels, depending on timing |
💊 Drug-Specific Tips
| Antifungal | Take With Food? | Notes |
|---|---|---|
| Itraconazole capsules | ✅ Yes – needs acid and fat for absorption | |
| Itraconazole solution | ❌ No – better on empty stomach | |
| Voriconazole | ❌ No – take 1 hour before or 1–2 hours after food | |
| Posaconazole tablets | ✅ Yes – improved absorption with food | |
| Isavuconazole | ✅ Can be taken with or without food |
✅ General Diet Tips During Treatment
-
Stay well hydrated
-
Eat a liver-friendly diet (low alcohol, reduced processed food, good hydration)
-
Focus on whole foods – vegetables, fruits (except grapefruit), whole grains, lean protein
-
Keep your pharmacist or consultant informed of any supplements or dietary changes
📌 Summary
Avoid:
-
Grapefruit, Seville oranges
-
Alcohol
-
Mouldy/fermented foods (for ABPA patients)
-
Herbal products like St John’s Wort
-
Antacids/PPIs without timing advice
Eat:
-
As recommended for your specific antifungal (some require food, others don’t)
-
A balanced, anti-inflammatory diet supportive of liver and immune health
🌦️ How Weather Affects Respiratory Symptoms
Weather has a well-documented impact on respiratory symptoms, especially in people with asthma, bronchiectasis, ABPA, CPA, COPD, and allergic lung diseases. The effects are complex and vary by individual, but here’s a clear, structured overview of what we know:
1. Cold Weather
❄️ Effects:
-
Constricts airways (bronchoconstriction), especially in asthma
-
Increases mucus production
-
Triggers coughing and breathlessness
-
Dries out nasal passages, increasing infection risk
🔍 At Risk:
-
Asthma, COPD, ABPA, bronchiectasis, CPA
✅ What Helps:
-
Wear a scarf or heat-exchange mask to warm inhaled air
-
Breathe through your nose, not your mouth
-
Use bronchodilators 15–30 mins before going out
2. Hot Weather & Heatwaves
☀️ Effects:
-
Causes airway irritation and inflammation
-
Worsens dehydration and mucus thickening
-
Triggers fatigue and breathlessness
-
Can increase ozone and air pollution levels
🔍 At Risk:
-
People on long-term corticosteroids or antifungals (e.g. risk of electrolyte imbalance)
-
CPA and bronchiectasis patients who already struggle with mucus clearance
✅ What Helps:
-
Stay indoors during the hottest part of the day
-
Keep well hydrated to thin secretions
-
Use a fan, shade, or cooling cloths — but avoid blowing dust directly into the face
3. Humid Weather
💧 Effects:
-
Promotes fungal and mould spore growth (e.g. Aspergillus)
-
Feels harder to breathe due to reduced air density
-
May worsen allergic responses in ABPA or SAFS
-
Can lead to damp indoor environments
✅ What Helps:
-
Dehumidifiers indoors (aim for 40–60% humidity)
-
FFP2/FFP3 masks during gardening or compost use
-
Avoid indoor drying of clothes if dampness is a problem
4. Sudden Weather Changes (e.g. Pressure Drops, Storms)
⛈️ Effects:
-
Trigger asthma and ABPA flares
-
Can cause “thunderstorm asthma” due to pollen breakdown and airborne allergen spikes
-
Barometric pressure changes may affect sinus and airway pressures
✅ What Helps:
-
Monitor air quality and pollen forecasts
-
Stay indoors with windows closed during storms
-
Use antihistamines or inhalers preventatively if needed
🌬️ Air Pollution and Weather
Weather also affects how pollutants (e.g. nitrogen dioxide, ozone, particulate matter) accumulate:
| Condition | Effect |
|---|---|
| Sunny + still air | Ozone builds up – irritates lungs |
| Cold + still air | Traps pollutants close to ground (inversion) |
| Windy or rainy | Cleans the air – often improves symptoms short term |
🧪 Evidence Highlights
-
Cold, dry air has been shown to trigger bronchospasm in asthmatic and bronchiectatic patients (European Respiratory Journal, 2020)
-
Thunderstorm asthma events have caused hospital surges (e.g. Melbourne, 2016)
-
High humidity increases airborne fungal spore concentrations, including Aspergillus fumigatus
-
Pollution and weather combinations increase hospital admissions for respiratory disease (Lancet Planetary Health, 2019)
✅ General Tips for Respiratory Patients
-
Track air quality, pollen, and weather using apps (e.g. Breezometer, AirVisual, Met Office)
-
Plan medication use around forecasted triggers (e.g. pre-treat with reliever inhaler)
-
Use air purifiers or dehumidifiers at home if needed
-
Layer clothing to control temperature and humidity exposure