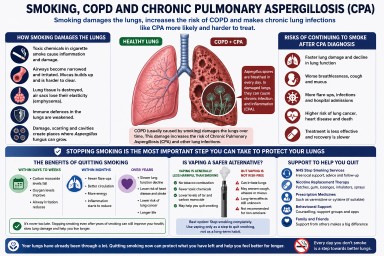
Smoking, COPD and Chronic Pulmonary Aspergillosis (CPA)
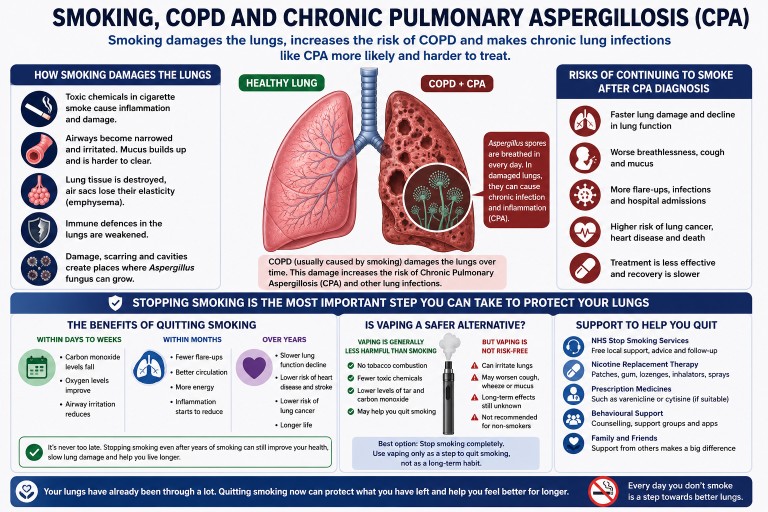
Many people diagnosed with Chronic Pulmonary Aspergillosis (CPA) also have Chronic Obstructive Pulmonary Disease (COPD). One of the strongest shared risk factors between the two conditions is cigarette smoking.
Smoking does not directly “cause” Aspergillus infection in the same way a virus or bacteria causes disease. However, it can create the lung damage and immune dysfunction that make CPA more likely to develop and harder to control.
Why smoking matters in CPA and COPD
Smoking damages the lungs over many years by:
- Destroying normal lung tissue and airways
- Causing chronic inflammation
- Reducing the lungs’ ability to clear mucus, dust and fungal spores
- Damaging the tiny hair-like structures called cilia that normally sweep organisms out of the airways
- Weakening local immune defence inside the lungs
- Increasing emphysema, cavities, scarring and bronchiectasis — all environments where Aspergillus can grow more easily
People breathe in Aspergillus spores every day. Healthy lungs usually remove them without difficulty. Damaged lungs are different. In COPD, especially severe COPD, spores can remain trapped in damaged airways and cavities, increasing the risk of long-term fungal colonisation or infection.
Is smoking causal?
The relationship is complex, but in many patients smoking is likely to be an important contributing cause.
Smoking contributes to:
- COPD development
- Structural lung damage
- Reduced immune clearance
- Increased infection risk
- Faster lung decline
All of these increase vulnerability to CPA.
Smoking is therefore not simply an associated factor. In many patients it is part of the chain of events that eventually leads to CPA developing.
Not every smoker develops CPA, and not every person with CPA has smoked. Some people develop CPA after tuberculosis, severe pneumonia, sarcoidosis, asthma, bronchiectasis, lung surgery or other lung diseases. However, smoking substantially increases risk because it accelerates lung injury and reduces the lungs’ resilience.
Why continuing to smoke after CPA diagnosis is dangerous
Once CPA is established, continuing to smoke can make management much harder.
Smoking may:
- Accelerate further lung destruction
- Worsen breathlessness and cough
- Increase mucus production
- Increase flare-ups and infections
- Reduce physical fitness and oxygen levels
- Reduce quality of life
- Increase hospital admissions
- Make COPD progression faster
- Increase risk of lung cancer alongside CPA
- Make recovery from infections slower
Many patients with CPA already have limited lung reserve. Continuing to smoke can progressively reduce the remaining healthy lung tissue.
“The damage is already done” — is stopping still worthwhile?
Yes. This is one of the commonest and most understandable feelings among long-term smokers with lung disease. However, stopping smoking can still help, even after significant lung damage has already occurred.
Within days to weeks
- Carbon monoxide levels fall
- Oxygen delivery improves
- Airways may become less irritated
- Some coughing and mucus clearance may improve
Within months
- COPD flare-ups may reduce
- Circulation improves
- Physical activity may become easier
- Inflammation begins to reduce
Over years
- Lung function decline slows
- Risk of heart disease and stroke falls
- Risk of lung cancer gradually decreases
- Survival improves compared with continued smoking
For people with CPA, preserving remaining lung function is critical. Slowing further structural damage may help stabilise disease, and antifungal treatment works best in lungs that are not being continually injured by smoke.
Is vaping a safer alternative?
Many patients ask whether vaping, or using e-cigarettes, is a safer option than smoking cigarettes.
Current evidence suggests that vaping is likely to be substantially less harmful than smoking tobacco cigarettes because vaping avoids the combustion process that produces tar, carbon monoxide and many toxic chemicals found in cigarette smoke.
For smokers who are unable to stop nicotine completely, switching entirely from smoking to vaping may reduce harm.
However, vaping is not risk-free.
Vaping aerosols can still irritate the lungs and may contain:
- Fine particles
- Flavouring chemicals
- Heating by-products
- Nicotine
- Other airway irritants
Some people with CPA, COPD, asthma or bronchiectasis report:
- Increased cough
- Throat irritation
- Chest tightness
- Wheeze
- Increased mucus symptoms
Long-term effects of vaping are still being studied.
For patients with CPA, the safest option for lung health is probably:
- Stop smoking cigarettes completely
- Use vaping only if it helps avoid returning to smoking
- Gradually reduce vaping if possible
A common problem is “dual use” — continuing to smoke while also vaping. This usually provides much less benefit than stopping cigarettes completely.
While nicotine itself is addictive, most of the major smoking-related lung damage comes from the toxic products created by burning tobacco.
For many patients, switching from smoking to vaping may still represent an important positive step if it helps them move away from cigarettes permanently.
Nicotine addiction is powerful
Many people with CPA and COPD have smoked for decades. Stopping is rarely simply a matter of willpower. Nicotine is strongly addictive, and smoking often becomes linked to stress relief, routine, anxiety management and social habits.
Patients should not feel ashamed if stopping is difficult. Repeated attempts are normal. Many successful quitters tried several times before succeeding permanently.
What can help?
Support works better than trying alone.
Options include:
- NHS stop smoking services
- Nicotine replacement therapy, such as patches, gum, sprays or lozenges
- Prescription medicines such as varenicline or cytisine, if suitable
- Behavioural support and counselling
- Gradual reduction plans
- Vape or e-cigarette transition strategies in selected patients
- Family and peer support
For many people with severe lung disease, stopping smoking is one of the most important treatments available — alongside inhalers, oxygen, physiotherapy and antifungal medication.
A realistic but important message
Patients with CPA often already live with fatigue, cough, breathlessness and anxiety about the future. Smoking may feel comforting in the short term, but it usually continues the cycle of lung injury that helped create the problem in the first place.
Stopping smoking cannot reverse established CPA, but it may:
- Slow further lung decline
- Improve day-to-day symptoms
- Improve response to treatment
- Preserve independence longer
- Reduce complications
- Improve long-term survival
Even small improvements in lung health can matter enormously when lung reserve is limited.
When to seek medical advice
Patients with CPA, COPD or other lung disease should seek medical advice if they:
- Become significantly more breathless
- Develop chest pain
- Cough up increasing amounts of blood
- Notice worsening wheeze or mucus production
- Develop fevers or signs of infection
- Feel unable to cope with smoking withdrawal symptoms alone
Stopping smoking is often difficult, but healthcare professionals can offer support and treatment options that improve the chances of success.
When Breathlessness Feels Severe — Even When Oxygen Levels Look “Normal”

Many people living with aspergillosis, severe asthma, bronchiectasis, or other chronic lung conditions describe a confusing and sometimes frightening experience:
“My oxygen saturations are normal, my peak flow is reasonable, there’s little wheeze, but I still feel like I’m drowning.”
This can be distressing for patients and frustrating for carers. Some people feel that because their oxygen levels or breathing tests appear “acceptable”, their symptoms are not fully understood.
Importantly, severe breathlessness can occur even when standard measurements such as oxygen saturations and peak flow readings appear relatively normal.
This does not mean the symptoms are imaginary or “all in the mind”. Breathlessness is complex and can have many different causes.
Why Breathlessness Is More Complicated Than Oxygen Levels
When doctors or nurses assess breathing problems, they often check:
- Oxygen saturation levels (sats)
- Peak flow readings
- Respiratory rate
- Presence of wheeze
- Chest sounds
These are all important. However, they do not always reflect how breathless a person feels.
Some people with chronic respiratory illness may have:
- Normal oxygen saturations
- Reasonable peak flow readings
- Little visible wheeze
- Minimal mucus production
…yet still experience intense sensations of:
- air hunger
- tight chest
- difficulty taking a satisfying breath
- feeling unable to “fill the lungs”
- panic associated with breathing
- extreme fatigue from breathing effort
What Can Cause This?
Breathlessness in aspergillosis and chronic lung disease is often caused by several factors happening together.
Inflammation and Airway Sensitivity
Conditions such as Allergic Bronchopulmonary Aspergillosis (ABPA), Severe Asthma with Fungal Sensitisation (SAFS), bronchiectasis, and Chronic Pulmonary Aspergillosis (CPA) can all cause inflamed and hypersensitive airways.
The lungs may feel irritated or tight even if oxygen exchange remains relatively preserved.
Small Airways Dysfunction
Some breathing problems occur in the smaller airways of the lungs and may not always show clearly on basic tests such as peak flow.
Patients can feel significant chest tightness or air trapping despite “good numbers”.
Muscle Fatigue
Breathing takes muscular effort. Chronic respiratory illness can place a long-term strain on the chest wall and breathing muscles, leading to exhaustion and increased awareness of breathing.
Mucus and Airflow Changes
Even relatively small amounts of mucus or airway narrowing can create sensations of chest heaviness or difficulty moving air.
Breathing Pattern Dysfunction (Dysfunctional Breathing)
This is increasingly recognised in people with chronic respiratory illness.
When breathing becomes difficult over months or years, people may unconsciously develop altered breathing patterns, including:
- rapid shallow breathing
- upper chest breathing
- frequent sighing
- over-breathing (hyperventilation)
- muscle tension around the chest and neck
This can worsen symptoms and create a vicious cycle where the sensation of breathlessness becomes amplified.
Symptoms may include:
- air hunger
- dizziness
- tingling
- tight chest
- panic sensations
- difficulty “switching off” breathing awareness
Importantly, this does not mean the illness is psychological or “not real”. Dysfunctional breathing can happen alongside genuine lung disease.
Why Inhalers Sometimes Seem to Help Less
Reliever inhalers such as salbutamol (Ventolin) are designed mainly to relax tightened airways.
If breathlessness is being driven partly by breathing pattern dysfunction, muscle fatigue, inflammation, hypersensitivity, or air trapping, inhalers may not always provide dramatic relief.
This can be confusing and upsetting for patients.
“But My Tests Are Normal…”
Many patients feel frustrated, frightened, or even dismissed when told that oxygen levels, peak flow readings, or chest examinations are “fine” despite severe breathlessness.
Normal oxygen saturations are reassuring in terms of immediate danger, but they do not always reflect the full experience of chronic respiratory illness.
Breathlessness is influenced by many factors including inflammation, airway sensitivity, breathing effort, muscle fatigue, anxiety associated with struggling to breathe, and altered breathing patterns.
Because of this, some people may feel profoundly breathless even when routine measurements appear relatively stable.
It can sometimes help to explain symptoms in practical, functional terms, such as:
- “I become breathless walking across the room.”
- “I recover much more slowly than usual.”
- “This feels much worse than my normal baseline.”
- “I feel exhausted by the effort of breathing.”
- “Breathing exercises seem to help settle things.”
These descriptions may help healthcare professionals understand how symptoms are affecting day-to-day life, rather than focusing only on oxygen levels or peak flow numbers.
Some patients also find it helpful to ask questions such as:
- Could breathing pattern dysfunction be contributing?
- Would respiratory physiotherapy help?
- Would pulmonary rehabilitation be appropriate?
- How should I judge when symptoms need urgent assessment?
Importantly, severe breathlessness should never simply be ignored. New, worsening, or unusual symptoms still require proper medical assessment.
At the same time, many people with chronic lung disease experience very real symptoms that are not always fully reflected by routine measurements alone.
Why Breathlessness Can Feel So Frightening
The sensation of breathlessness is created by the brain interpreting signals from the lungs, breathing muscles, chest wall, blood chemistry, and nervous system.
This means that the feeling of “not getting enough air” is not determined only by oxygen levels.
In chronic lung disease, several things can trigger the sensation of breathlessness, including:
- inflamed or sensitive airways
- extra effort needed to move air in and out
- air trapping in the lungs
- muscle fatigue
- rapid or shallow breathing
- stress hormones released during breathing distress
- heightened awareness of breathing sensations
When breathing becomes uncomfortable, the body naturally responds with anxiety and adrenaline. This is a protective survival response.
Unfortunately, this can sometimes create a cycle:
breathlessness → anxiety → faster breathing → more chest tightness → worse breathlessness
This does not mean symptoms are “psychological”. The physical sensation is real, but the body’s alarm systems can unintentionally amplify it.
What Can Help During an Episode of Breathlessness?
Different techniques help different people, and severe or rapidly worsening symptoms should always be medically assessed. However, some patients find the following approaches helpful during episodes of distressing breathlessness:
Slow the Breathing Rate
Trying to slow breathing gently can help reduce over-breathing and chest tightness.
Some people find it helpful to:
- breathe in gently through the nose
- breathe out slowly through pursed lips
- focus on making the out-breath longer than the in-breath
Use a Recovery Position
Sitting forward slightly with the arms supported on knees or a table can sometimes reduce the work of breathing.
Reduce Panic and “Air Hunger”
Trying to fight for bigger and bigger breaths can sometimes worsen symptoms.
Some patients find it more helpful to focus on:
- gentle breathing rhythm
- relaxing the shoulders and neck
- slowing breathing rather than deepening it
- focusing attention away from the chest where possible
Use Prescribed Treatments Appropriately
Follow the advice provided by your healthcare team regarding inhalers, nebulisers, airway clearance, or rescue medication.
If inhalers are not helping as expected, this should be discussed with a respiratory specialist rather than simply increasing use repeatedly.
Know Your “Usual” Pattern
Many patients find it useful to learn the difference between:
- their “usual” chronic breathlessness
- breathing pattern dysfunction or over-breathing episodes
- symptoms suggesting infection or acute deterioration
This can help patients feel more confident recognising when urgent medical assessment may be needed.
Can Breathing Retraining Help?
Some patients find breathing retraining exercises very helpful, especially when guided by:
- respiratory physiotherapists
- specialist breathing services
- pulmonary rehabilitation teams
- asthma nurse specialists
Breathing retraining may include:
- slowing breathing rate
- diaphragmatic (“belly”) breathing
- nasal breathing techniques
- recovery breathing positions
- relaxation techniques
- paced activity and pacing strategies
Some NHS respiratory teams recommend online breathing resources and guided exercises to help patients recognise and manage over-breathing patterns.
These approaches are usually intended to work alongside medical treatment — not instead of it.
Living With an “Invisible” Symptom
One of the hardest aspects of chronic breathlessness is that outward signs may not always match how severe symptoms feel internally.
Many patients report feeling dismissed when oxygen levels are normal or when tests appear “better than expected”.
The experience of breathlessness is real, even when routine measurements do not fully explain it.
This is one reason why specialist respiratory assessment can be important in complex conditions such as aspergillosis.
When to Seek Medical Help
You should seek urgent medical advice if breathlessness is:
- suddenly worsening
- associated with chest pain
- causing blue lips or fingertips
- associated with falling oxygen saturations
- accompanied by fever or signs of infection
- causing confusion or severe exhaustion
- significantly different from your usual symptoms
Even if previous episodes have been related to breathing pattern dysfunction, new or worsening symptoms should still be medically assessed.
Further Support
You may also find these resources helpful:
Last reviewed: May 2026
Produced by: National Aspergillosis Centre CARES Team / Aspergillosis Website
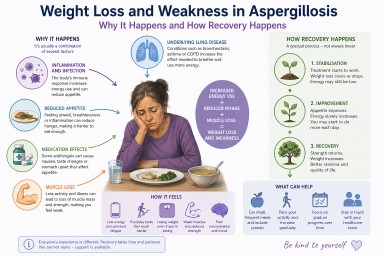
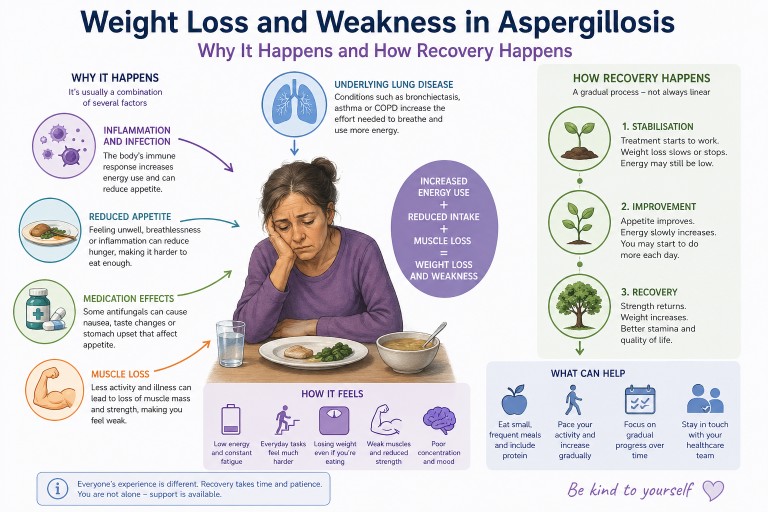
Weight Loss and Weakness in Aspergillosis: Why It Happens, How It Feels, and What Helps Recovery
Last reviewed: April 2026
Unexpected weight loss and severe weakness are among the most worrying symptoms people report after being diagnosed with aspergillosis. Many describe feeling unlike themselves—physically drained, thinner than they have ever been, and struggling with everyday activities.
This article explains why this happens, what is going on in the body, and what recovery typically looks like.
---
Key Points
- Weight loss and fatigue are common in aspergillosis, particularly early in the illness or during flare-ups.
- They are usually caused by a combination of inflammation, increased energy use, reduced appetite, and muscle loss.
- Medication side effects can contribute but are rarely the main cause.
- Many people improve over time, but recovery is usually gradual and can take weeks to months.
- Stabilising weight is often the first important step before regaining strength.
---
Contents
- Why does aspergillosis cause weight loss?
- What is happening inside the body?
- Which types of aspergillosis are affected?
- Why does it feel so severe?
- Does it get better?
- What can help day to day?
- Nutrition and rebuilding strength
- When to seek medical advice
- Common questions
---
Why does aspergillosis cause weight loss?
Weight loss in aspergillosis is rarely due to a single cause. Instead, it is usually the result of several overlapping processes.
1. Increased energy use (hypermetabolism)
When the body is dealing with infection or inflammation, it requires more energy. This is sometimes described as a hypermetabolic state.
- The immune system is active and consumes energy
- The body produces inflammatory signals
- Breathing effort may increase
This means you may be burning more calories than usual—even at rest.
2. Reduced appetite
Many people notice they are eating less, sometimes without realising it. This may be due to:
- Feeling unwell or fatigued
- Shortness of breath when eating
- Changes in appetite driven by inflammation
3. Medication effects
Some treatments can affect appetite or digestion. For example:
- Antifungal medications such as itraconazole or voriconazole may cause nausea or taste changes
- Steroids may increase appetite but can also contribute to muscle weakness over time
Medication effects vary widely and are usually only part of the overall picture.
4. Muscle breakdown
During illness, the body may break down muscle to meet energy needs. This can happen quickly, especially if activity levels fall.
This leads to:
- Loss of strength
- Reduced stamina
- A feeling of being “weak” rather than just lighter
5. Underlying lung disease
Many people with aspergillosis also have conditions such as bronchiectasis, asthma, or chronic obstructive pulmonary disease (COPD). These can increase the effort required for breathing and contribute to ongoing energy use.
---
What is happening inside the body?
Several biological processes contribute to weight loss and fatigue:
- Inflammatory signalling: The immune system releases chemical signals that affect metabolism and appetite
- Catabolism: The body breaks down tissues (including muscle) to release energy
- Energy imbalance: More energy is used than consumed
This combination can make weight loss feel rapid and difficult to control.
---
Which types of aspergillosis are affected?
These symptoms are most commonly seen in:
- Chronic Pulmonary Aspergillosis (CPA)
- Allergic Bronchopulmonary Aspergillosis (ABPA), particularly during flare-ups
However, not everyone experiences weight loss, and severity varies.
---
Why does it feel so severe?
Many people describe this stage as one of the most difficult parts of their illness. This is because several factors are happening at once:
- Physical energy is reduced
- Muscle strength has declined
- The body is under ongoing stress
- Recovery has not yet begun
This can make everyday activities—such as walking, cooking, or even eating—feel unusually difficult.
---
Does it get better?
In many cases, yes—there is gradual improvement over time, especially once treatment begins to control the condition.
Recovery often follows a pattern:
- Initial phase: weight loss and severe fatigue
- Stabilisation: weight loss slows or stops
- Recovery: gradual return of strength and energy
This process is usually slow and uneven, with good and bad days.
---
What can help day to day?
1. Focus on maintaining nutrition
- Eat small amounts regularly rather than large meals
- Choose foods that are easy to prepare and eat
- Include protein to support muscle maintenance
2. Pace activity carefully
- Gentle movement can help maintain strength
- Avoid pushing too hard, as this can worsen fatigue
- Increase activity gradually as energy improves
3. Look at trends over time
It can be helpful to focus on gradual changes such as:
- Weight stabilising
- Small improvements in energy
---
Nutrition and rebuilding strength
Recovery often happens in stages:
- Stage 1: Stabilising weight
- Stage 2: Gradually increasing intake
- Stage 3: Rebuilding muscle and strength
Regaining muscle mass takes time and usually follows once the underlying condition is better controlled.
---
When to seek medical advice
You should contact your healthcare team if you experience:
- Continued or rapid weight loss
- Increasing weakness
- Difficulty eating or swallowing
- New or worsening symptoms
This may indicate the need for additional support or adjustment of treatment.
---
Common questions
Is weight loss just due to poor appetite?
No. Reduced appetite is only one factor. Increased energy use and muscle loss are also important contributors.
Are medications the main cause?
Medications can contribute, but they are rarely the main reason for weight loss.
Will I regain my strength?
Many people do regain strength over time, although recovery is usually gradual.
Why does recovery take so long?
The body needs time to reduce inflammation, restore energy balance, and rebuild muscle.
---
Summary
Weight loss and weakness in aspergillosis are common and can feel severe, particularly early in the illness. They are usually caused by a combination of increased energy use, reduced appetite, muscle loss, and underlying lung disease.
Although recovery can take time, many people improve gradually as treatment takes effect.
---
Further Reading
- Chronic Pulmonary Aspergillosis (CPA)
- Allergic Bronchopulmonary Aspergillosis (ABPA)
- Treatment of Aspergillosis
---
Author & Review
Prepared for aspergillosis.org to support patient understanding. Content reflects current clinical knowledge and patient-reported experience.
Disclaimer
This page is for general information only and does not replace advice from your healthcare team.
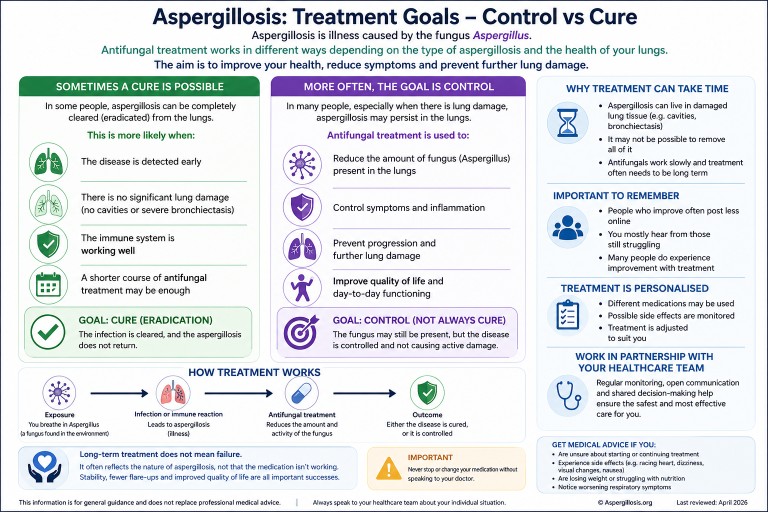
Can Aspergillosis Be Cured? Understanding Treatment, Control, and Long-Term Therapy
Last reviewed: April 2026
Key points
- Aspergillosis is caused by fungi from the Aspergillus group.
- Most people breathe in Aspergillus spores regularly without becoming ill.
- In some people, damaged airways, lung cavities, mucus plugs, or immune responses allow the fungus or fungal material to persist.
- Antifungal treatment may aim to cure, but in many cases the goal is long-term control.
- Steroids can reduce harmful inflammation in allergic disease, but they can also reduce the body’s ability to clear fungus.
Table of contents
- Overview
- Why breathing in spores does not usually cause disease
- Why aspergillosis can be hard to clear
- Infection and ABPA: different reasons for persistence
- Do steroids influence this?
- Control vs cure
- Common antifungal treatments
- Why you may not hear many success stories
- When to seek medical advice
- Common questions
Overview
It is very common for people diagnosed with aspergillosis to feel worried when they read that others have been taking antifungal medication for months or even years.
This can lead to an understandable question:
“Does treatment actually work, or will I have this forever?”
The answer is more nuanced than a simple yes or no. Different forms of aspergillosis behave differently, and treatment goals vary depending on the condition.
Two of the most common conditions are:
- Allergic Bronchopulmonary Aspergillosis (ABPA) – an allergic immune reaction to Aspergillus in the airways
- Chronic Pulmonary Aspergillosis (CPA) – a long-term fungal infection, usually in areas of damaged lung tissue
Understanding this difference is key to understanding why treatment may continue for a long time.
If you would like a more detailed explanation of how these conditions are diagnosed and managed, see our guides to chronic pulmonary aspergillosis (CPA) and allergic bronchopulmonary aspergillosis (ABPA).
Why breathing in spores does not usually cause disease
Aspergillus spores are present in the environment, including air, soil, compost, dust, and decaying vegetation. Most people breathe in small numbers of these spores regularly without becoming ill.
In healthy lungs, spores are usually cleared by the immune system and by the normal cleaning mechanisms of the airways.
This means:
- Exposure to Aspergillus is common
- Most exposure does not lead to disease
- Aspergillosis usually develops only when there are additional risk factors, such as lung damage, mucus trapping, or altered immune responses
So it is not accurate to think of most patients as being “constantly reinfected”. A better way to think about it is that some lungs provide conditions where Aspergillus, or fungal material, can persist and continue to cause problems.
Why aspergillosis can be hard to clear
Aspergillosis can be difficult to clear for several reasons. These include the structure of the lungs, the biology of the fungus, limitations of drug penetration, and the way the immune system responds.
1. Damaged lung tissue can provide protected spaces
In CPA, Aspergillus often grows in areas of abnormal lung, such as cavities, scarred tissue, or areas affected by bronchiectasis.
These areas can act as protected spaces where the fungus is harder for the immune system and antifungal medicines to reach.
2. Thick mucus can trap fungus and fungal material
In airway diseases such as asthma, bronchiectasis, and ABPA, thick mucus can trap spores, hyphae, and fungal fragments.
This trapped material can continue to stimulate inflammation even when the fungus is not invading lung tissue.
3. Antifungal medicines may suppress rather than sterilise
Antifungal medications can reduce fungal activity and help prevent progression, but they may not always remove every trace of fungus from damaged lung spaces or mucus-filled airways.
For this reason, success is often measured by:
- Improved symptoms
- Stabilised weight and energy
- Fewer flare-ups
- Stable or improved scans
- Prevention of further lung damage
Infection and ABPA: different reasons for persistence
Chronic pulmonary aspergillosis: persistence of infection
In chronic pulmonary aspergillosis, the problem is fungal growth in damaged lung tissue.
- Lung cavities provide spaces where fungus can grow
- Drug penetration may be limited
- The immune system may not fully clear infection
Allergic bronchopulmonary aspergillosis: persistence of reaction
In ABPA, the main issue is an exaggerated immune response.
- Mucus traps fungal material
- Small amounts can trigger strong reactions
- Inflammation leads to more mucus
Do steroids influence this?
Yes. Steroids can be helpful but must be used carefully.
In ABPA, they reduce inflammation but may also reduce fungal clearance.
In chronic infection, steroids can increase the risk of persistence or progression.
Monitoring and drug interactions are important during treatment.
Control vs cure: what is the goal?
For many people, the realistic goal is:
- Stability rather than eradication
- Reduced symptoms
- Prevention of progression
Common antifungal treatments
- Itraconazole
- Voriconazole
- Posaconazole
These treatments are selected based on individual factors and require monitoring.
Why don’t I hear many success stories?
People who improve often post less, while those still struggling are more visible in forums.
When to seek medical advice
- Uncertainty about treatment
- Side effects
- Weight loss
- Worsening symptoms
Common questions
Can aspergillosis be cured?
Sometimes, but often it is managed long-term.
Are people constantly reinfected?
No. Most people clear spores regularly without issue.
Why is ABPA difficult to treat?
Because of ongoing immune reactions and mucus trapping.
Further reading
- Chronic Pulmonary Aspergillosis (CPA)
- Allergic Bronchopulmonary Aspergillosis (ABPA)
- Antifungal medicines
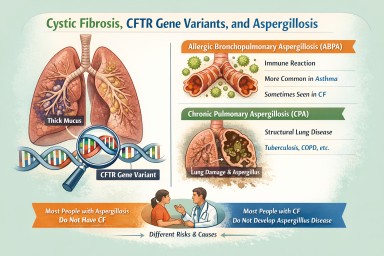
Cystic Fibrosis, CFTR Gene Variants, and Aspergillosis
Last reviewed: 8 April 2026
Some people with aspergillosis are told they have cystic fibrosis (CF), or that they carry a CFTR gene variant. This can be unexpected and may raise concerns about whether this explains their symptoms or diagnosis.
This article explains how cystic fibrosis and CFTR gene variants relate to Aspergillus-related lung disease, what current research shows, and—importantly—what conclusions should not be drawn.
Contents
- Key points
- Important reassurance
- What is cystic fibrosis?
- What is a CFTR gene variant?
- How CFTR affects the lungs
- How Aspergillus behaves in the lungs
- ABPA and cystic fibrosis
- CPA and cystic fibrosis
- Modern CF treatments and Aspergillus
- Does a CFTR variant explain symptoms?
- What should patients take from this?
- When to seek medical advice
- Conclusion
- References
Key points
- Most people with aspergillosis do not have cystic fibrosis.
- Most people with cystic fibrosis do not develop ABPA or CPA.
- ABPA is linked to mucus and immune responses, not just infection.
- CFTR variants may contribute to risk in some people, but are usually only one factor.
- CPA is mainly driven by structural lung damage, not CFTR genetics.
Important reassurance
Most people with aspergillosis do not have cystic fibrosis, and most people with cystic fibrosis do not develop Aspergillus-related disease.
Although these conditions can overlap, they are usually separate. Genetic findings such as CFTR variants should be interpreted carefully and in context.
What is cystic fibrosis?
Cystic fibrosis is a genetic condition caused by changes in the CFTR gene. This gene regulates salt and water movement across cells.
When CFTR function is reduced:
- mucus becomes thick and sticky
- airways are harder to clear
- microorganisms persist more easily
This creates an environment where bacteria and fungi can accumulate over time.
What is a CFTR gene variant?
CFTR variants range from severe mutations (causing cystic fibrosis) to mild or uncertain variants.
Carriers (with one variant):
- are common in the general population
- usually have no symptoms
- may have subtle effects in some cases
These subtle effects may include reduced mucus clearance or increased susceptibility to airway inflammation.
How CFTR affects the lungs
CFTR dysfunction affects the lungs in several key ways:
- Mucus dehydration: mucus becomes thick and difficult to clear
- Impaired clearance: particles and microbes remain in the airways
- Chronic inflammation: immune responses become exaggerated
This combination creates a “retention environment” where inhaled organisms—including Aspergillus—may persist.
How Aspergillus behaves in the lungs
Aspergillus is inhaled by everyone, but its effects vary depending on the lung environment.
- Healthy lungs: spores are cleared
- Impaired clearance: spores may persist
- Sensitive immune system: allergic reactions may develop
- Damaged lungs: chronic infection may develop
This explains why Aspergillus-related disease is diverse and depends heavily on underlying lung conditions.
ABPA and cystic fibrosis
ABPA is an allergic immune reaction to Aspergillus.
It is recognised in cystic fibrosis because:
- mucus retention increases exposure to Aspergillus
- immune responses can be exaggerated
However:
- Many CF patients never develop ABPA
- Most ABPA patients do not have CF
Some studies suggest CFTR variants may increase susceptibility, but this is not consistent across all research.
Key message: ABPA and CF can overlap, but one does not imply the other.
CPA and cystic fibrosis
CPA is a chronic fungal infection that develops in structurally damaged lungs.
The most important risk factor is:
pre-existing lung damage
This includes:
- bronchiectasis
- previous tuberculosis
- COPD
Cystic fibrosis can lead to bronchiectasis, and therefore indirectly increase CPA risk.
However:
- CPA is rarely driven directly by CFTR genetics
- most CPA patients do not have CF
Key message: CPA is primarily a disease of lung structure, not genetics.
Modern CF treatments and Aspergillus
CFTR modulators (such as elexacaftor/tezacaftor/ivacaftor) have transformed CF care.
They:
- improve CFTR function
- thin mucus
- improve clearance
Studies suggest:
- reduced Aspergillus detection in some patients
- fewer ABPA exacerbations in some cases
However:
- ABPA still occurs
- existing lung damage remains
- immune responses are not fully corrected
Overall: these therapies improve risk but do not eliminate Aspergillus-related disease.
Does a CFTR variant explain symptoms?
No single factor explains complex lung disease.
Symptoms may result from:
- underlying lung disease
- infection
- inflammation
- environmental exposure
A CFTR variant may contribute, but is rarely the sole cause.
What should patients take from this?
- CF and CFTR variants can sometimes contribute
- ABPA has the strongest connection
- CPA is mainly driven by lung damage
- Most patients with aspergillosis do not have CF
When to seek medical advice
Seek advice if symptoms worsen, change, or include coughing up blood, fever, or chest pain.
Conclusion
Cystic fibrosis and CFTR gene variants can play a role in some patients with Aspergillus-related lung disease, particularly where mucus clearance is affected. However, they should not be overemphasised. In most cases, they are just one part of a broader clinical picture involving lung structure, immune response, and environmental exposure.
References
- Miller PW et al. (1996)
- Marchand E et al. (2001)
- Eaton TE et al. (2002)
- Agarwal R et al. (2012)
- Chaudhary N et al. (2012)
- Warris A et al. (2019)
- Gamaletsou MN et al. (2018)
- Kosmidis C & Denning DW (2015)
- Moldoveanu B et al. (2021)
- Malik HS et al. (2025)
- Zeng C et al. (2025)
- Bendixen MP et al. (2025)
This article is for general information and does not replace advice from your clinical team.
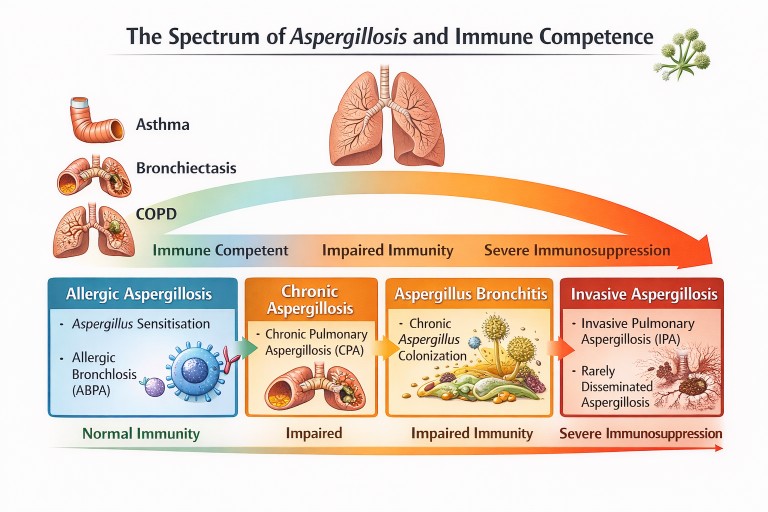
Asthma and Aspergillosis
How fungal spores interact with asthma and other lung diseases
Every day we inhale thousands of microscopic fungal spores from the environment. One of the most common fungi in the air is Aspergillus fumigatus. In healthy lungs these spores are removed quickly by the lungs’ natural defence systems and cause no illness.
However, in people with asthma—particularly severe asthma—the interaction between the lungs and Aspergillus can be very different. The fungus may trigger allergic inflammation, grow in mucus within the airways, or occasionally contribute to chronic lung disease.
Understanding this relationship helps explain several important conditions including:
-
Aspergillus sensitisation
-
Severe Asthma with Fungal Sensitisation (SAFS)
-
Allergic Bronchopulmonary Aspergillosis (ABPA)
-
Aspergillus bronchitis
-
Chronic Pulmonary Aspergillosis (CPA)
Although asthma is the most common condition linked to Aspergillus allergy, other lung diseases such as bronchiectasis, Chronic Obstructive Pulmonary Disease (COPD), and tuberculosis-related lung damage can also create environments where the fungus becomes important.
Why Asthma Creates a Favourable Environment for Aspergillus
Asthma is a disease of airway inflammation and hyper-reactivity. The bronchi narrow during attacks because the airway wall becomes swollen and the surrounding smooth muscle contracts.
Several features of asthma make it easier for Aspergillus spores to remain in the lungs.
Mucus production
Asthma often causes increased production of thick airway mucus.
Normally mucus traps inhaled particles and moves them upward toward the throat via the mucociliary escalator.
In asthma:
-
mucus becomes thicker
-
clearance becomes less efficient
-
spores remain trapped
This trapped environment allows fungal spores to persist in the airway mucus.
Allergic immune responses
Many asthma patients have Type-2 (T2) inflammation (50-70%), involving immune pathways driven by:
-
Immunoglobulin E (IgE)
-
Interleukin-4
-
Interleukin-5
-
Interleukin-13
-
eosinophils
These pathways respond strongly to fungal allergens. When the immune system recognises Aspergillus proteins it may trigger allergic inflammation in the airways.
Fungal sensitisation is increasingly recognised as an important contributor to severe asthma (PMID: 24735832).
Aspergillus Sensitisation
Many people with asthma develop allergic sensitisation to Aspergillus.
Sensitisation means the immune system produces antibodies against fungal proteins.
Features include:
-
positive Aspergillus skin test or IgE blood test
-
worsening asthma symptoms
-
increased exacerbations
Studies suggest 10–25% of patients attending severe asthma clinics show Aspergillus sensitisation (PMID: 24735832).
However, sensitisation alone does not necessarily cause lung damage.
Severe Asthma with Fungal Sensitisation (SAFS)
Some patients with severe asthma have fungal sensitisation but do not meet the criteria for ABPA.
This condition is known as Severe Asthma with Fungal Sensitisation (SAFS).
Typical features include:
-
severe or poorly controlled asthma
-
fungal allergy
-
moderate IgE elevation
A randomised controlled trial demonstrated that antifungal therapy may improve symptoms in some SAFS patients (PMID: 18948425).
Allergic Bronchopulmonary Aspergillosis (ABPA)
Allergic Bronchopulmonary Aspergillosis is the most important Aspergillus-related disease associated with asthma.
ABPA occurs when Aspergillus grows within airway mucus and triggers a strong allergic immune response.
Typical findings include:
-
very high total IgE levels
-
Aspergillus-specific IgE and IgG antibodies
-
eosinophilia
-
mucus plugs containing fungal hyphae
-
central bronchiectasis
ABPA occurs in approximately:
-
1–2% of all asthma patients
-
up to 10–15% of severe asthma patients
These figures come from global prevalence estimates of ABPA in asthma populations (PMID: 23210682/.
Modern diagnostic criteria for ABPA were updated by the International Society for Human and Animal Mycology (ISHAM) in 2024 (PMID: 38423624).
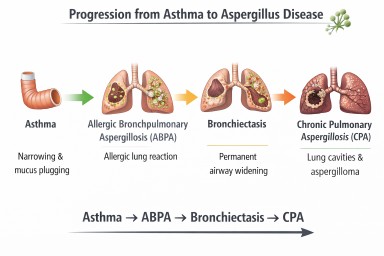
Asthma and Aspergillus Disease Pathway
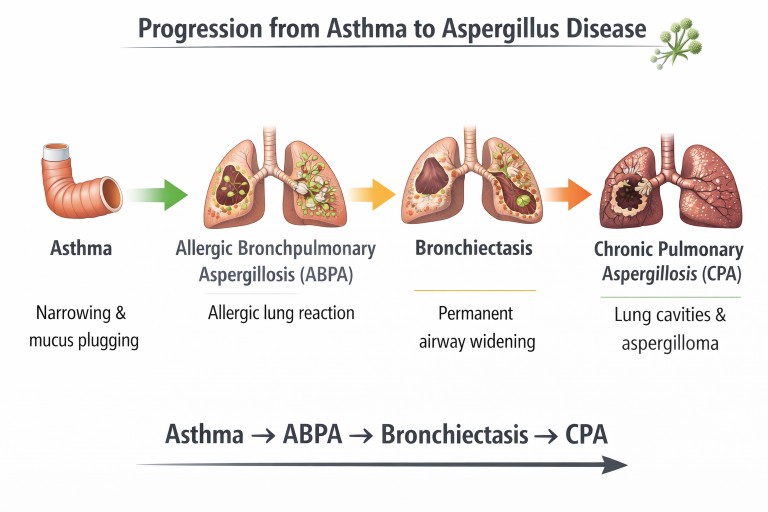
Possible interactions between asthma and Aspergillus. Some patients develop allergic disease (ABPA) which may lead to airway damage such as bronchiectasis (NB Progression to CPA is very rare).
When ABPA Causes Bronchiectasis
Repeated inflammation from ABPA may damage airway walls and lead to bronchiectasis.
Bronchiectasis occurs when airways become:
-
permanently widened
-
distorted
-
unable to clear mucus effectively
Instead of being cleared from the lungs, mucus pools in the airways.
This retained mucus creates an environment where microorganisms—including fungi—can grow.
Aspergillus Bronchitis
In some patients with bronchiectasis or chronic lung disease, Aspergillus may persist in airway mucus and cause chronic airway infection rather than allergy.
Symptoms may include:
-
chronic cough
-
sputum production
-
repeated positive Aspergillus cultures
IgE levels are usually lower than in ABPA.
Chronic Pulmonary Aspergillosis (CPA)
Chronic Pulmonary Aspergillosis is a slowly progressive fungal infection of damaged lung tissue.
CPA usually develops in lungs containing:
-
cavities
-
severe structural damage
Common underlying diseases include:
-
tuberculosis
-
sarcoidosis
-
severe COPD
Globally, the most common cause of CPA is previous tuberculosis infection (PMID: 22271943).
Asthma alone rarely causes CPA, but severe bronchiectasis or ABPA-related lung damage may occasionally lead to it.
Aspergillosis and Immune Competence
Different forms of aspergillosis occur depending on lung damage and immune function.
Other Lung Diseases Linked to Aspergillus
Although asthma is the most common condition associated with Aspergillus allergy, several other lung diseases can predispose to fungal disease.
Bronchiectasis
Dilated airways trap mucus, allowing fungi and bacteria to persist.
COPD
Chronic airway inflammation may lead to Aspergillus bronchitis or chronic pulmonary aspergillosis.
Tuberculosis
Post-tuberculosis lung cavities are the most common global cause of chronic pulmonary aspergillosis (PMID: 22271943).
Key Messages
-
Asthma is one of the most important diseases associated with Aspergillus-related lung conditions.
-
Many asthma patients develop fungal sensitisation.
-
A smaller proportion develop Allergic Bronchopulmonary Aspergillosis (ABPA).
-
Repeated inflammation from ABPA can lead to bronchiectasis.
-
Chronic pulmonary aspergillosis is rare in asthma alone but may occur if significant lung damage develops.
Understanding these interactions helps guide diagnosis and treatment for people living with asthma and Aspergillus-related disease.
Further reading
Agarwal R, Chakrabarti A, Shah A, Gupta D, Meis JF, Guleria R, Moss R, Denning DW; ABPA complicating asthma ISHAM working group. Allergic bronchopulmonary aspergillosis: review of literature and proposal of new diagnostic and classification criteria. Clin Exp Allergy. 2013 Aug;43(8):850-73. doi: 10.1111/cea.12141. PMID: 23889240.
Denning DW, Pleuvry A, Cole DC. Global burden of chronic pulmonary aspergillosis as a sequel to pulmonary tuberculosis. Bull World Health Organ. 2011 Dec 1;89(12):864-72. doi: 10.2471/BLT.11.089441. Epub 2011 Sep 27. PMID: 22271943; PMCID: PMC3260898.
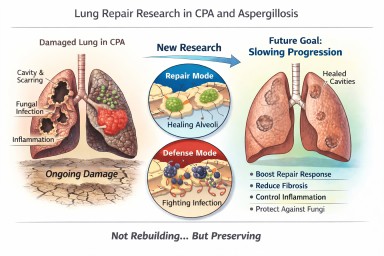
Looking further into the future - could we control lung damage, preserve healthy lung tissue better?
Can Lungs Repair Themselves?
What New Research Means for People with CPA (and Other Aspergillosis)
A recent scientific discovery has helped researchers understand how certain lung cells decide whether to focus on repairing damage or defending against infection. The work, highlighted by the Mayo Clinic and published in Nature Communications, describes a molecular “switch” inside specialised lung cells that influences this balance.
For people living with Chronic Pulmonary Aspergillosis (CPA) — and also those with Allergic Bronchopulmonary Aspergillosis (ABPA) — this kind of research is relevant. But it needs careful explanation.
This is not about rebuilding destroyed lungs.
It is about understanding how to better protect and preserve the lung tissue that remains.
The Discovery: A “Repair vs Defence” Switch
Researchers identified a regulatory circuit in alveolar type II (AT2) cells — specialised cells that:
-
Produce surfactant (which keeps air sacs open)
-
Act as a reserve “repair” population in the lung
-
Can regenerate other essential lung cells after injury
The study showed that these cells operate under tight control. When infection is present, they prioritise defence. When injury needs healing, they can switch into repair mode.
The key insight is that this switch is biologically regulated. It is not random. That means, in theory, it may one day be possible to influence it.
What “Repair” Means — and What It Does Not Mean
When we talk about lung repair in this context, we must be very clear.
It does not mean:
-
Lung cavities caused by CPA will close in the foreseeable future
-
Established fibrosis will melt away
-
Bronchiectasis will reverse
-
Severely distorted lung architecture will rebuild
CPA cavities represent major structural remodelling — destruction of alveoli, scarring, altered blood supply, and thickened pleura. Reconstructing that complex architecture is biologically extremely challenging and not currently realistic within the next decade.
What repair does realistically mean
In chronic lung disease, “repair” is more likely to mean:
-
Supporting survival of remaining alveoli
-
Preventing excessive fibrotic signalling
-
Helping lung lining cells recover more efficiently after inflammation
-
Reducing cumulative injury from repeated infection
-
Slowing progression of structural change
In other words:
Not rebuilding what is gone — but better protecting what remains.
For many people with CPA, this is a crucial distinction.
Why Preservation Is a Major Goal in CPA
CPA usually develops in lungs already weakened by conditions such as tuberculosis, non-tuberculous mycobacteria, chronic obstructive pulmonary disease, or severe pneumonia.
Over time, CPA can lead to:
-
Expanding cavities
-
Progressive scarring
-
Reduced gas exchange
-
Reduced exercise tolerance
Many patients have limited lung reserve. Even small additional losses of functioning lung tissue can significantly increase breathlessness or fatigue.
If future therapies could slow the rate of progression — even modestly — that would meaningfully affect long-term outcomes.
Flattening the decline curve is not trivial. It changes quality of life.
Why This Also Matters in ABPA
In ABPA, repeated inflammatory episodes can lead to:
-
Airway remodelling
-
Mucus plugging
-
Development or progression of bronchiectasis
Better control of inflammatory signalling — combined with improved epithelial recovery — could reduce long-term airway damage.
Again, this is about preservation rather than reversal.
Where Development Has Reached
The current research is still laboratory-based. It used advanced techniques such as:
-
Single-cell sequencing
-
Imaging of lung tissue
-
Preclinical models of injury
No human treatments based on this discovery are yet available.
However, the significance lies in identifying:
-
A defined molecular pathway
-
A controllable regulatory mechanism
-
A clearer understanding of why repair fails in chronic inflammation
That foundational knowledge is what eventually allows targeted drug development.
The Balance Challenge in Aspergillosis
There is an additional complexity in fungal lung disease.
Any attempt to promote repair must not weaken antifungal defence.
The immune system must:
-
Control Aspergillus
-
Avoid causing excessive inflammatory damage
Future therapies would need to strike that balance carefully.
What This Means for Patients Now
This discovery does not change current treatment.
The most effective preservation strategies today remain:
-
Consistent antifungal therapy when indicated
-
Careful inflammatory control
-
Biologic therapies where appropriate
-
Airway clearance
-
Vaccination and infection prevention
-
Avoiding damp and mould exposure
-
Pulmonary rehabilitation
These measures are already forms of lung preservation.
A Realistic and Hopeful Perspective
It is unlikely that cavities from CPA will be repaired in the near future.
It is realistic that within the next 5–10 years we may see improved strategies aimed at:
-
Slowing structural progression
-
Supporting endogenous repair cells
-
Reducing fibrotic signalling
-
Improving recovery after exacerbations
For people living long-term with CPA or ABPA, even incremental preservation could significantly affect independence and quality of life.
The science is still early — but understanding how the lung decides to repair itself is an important step forward.
Reference
Sawhney, A.S., Deskin, B.J., Cai, J. et al. A molecular circuit regulates fate plasticity in emerging and adult AT2 cells. Nat Commun 16, 8924 (2025). https://doi.org/10.1038/s41467-025-64224-1
Can blood tests help predict if chronic pulmonary aspergillosis will come back?
This study from the National Aspergillosis Centre (NAC) looked at people with chronic pulmonary aspergillosis (CPA) who had completed antifungal treatment and asked a simple question:
Can blood tests tell us who is more likely to relapse after treatment stops?
What the researchers did
Doctors reviewed patients with CPA who had:
-
Taken antifungal treatment for at least 6 months
-
Stopped treatment because they were clinically stable
They then followed these patients to see who stayed well and who relapsed, and compared this with their blood test results at the time treatment stopped.
What they found
-
About 1 in 4 patients had a relapse after stopping treatment
-
People whose Aspergillus IgG blood test was still high at the end of treatment were much more likely to relapse
-
Patients whose IgG level had fallen to a lower level did not relapse in this study
-
Signs of Aspergillus allergy or sensitisation also increased relapse risk
-
CT scan appearances and treatment length alone were not reliable predictors
Why this matters for patients
This means that:
-
Blood tests may help doctors decide when it is safe to stop treatment
-
Some people may need closer follow-up or longer treatment
-
Follow-up can be more personalised, rather than “one size fits all”
Importantly, a relapse does not mean treatment failed — it reflects how persistent this infection can be in damaged lungs.
Key takeaway
A simple blood test at the end of treatment may help predict who needs closer monitoring for CPA relapse.
This research supports a more individualised approach to long-term CPA care.
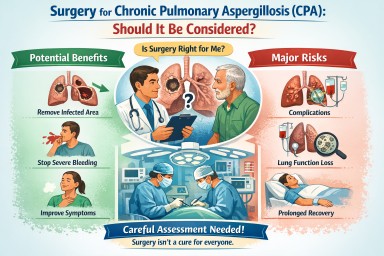
Surgery for Chronic Pulmonary Aspergillosis (CPA): why it is sometimes considered – and often not
For people living with chronic pulmonary aspergillosis (CPA), the idea of surgery can raise difficult questions. Some patients are told surgery might offer a chance of cure; others are advised very firmly against it. Both positions can be correct, depending on the individual situation.
This article explains when surgery may be considered, why it is often avoided, and what “success” or “cure” really means in CPA.
Why is surgery even considered in CPA
CPA usually develops in lungs that are already damaged (for example, by tuberculosis, chronic obstructive pulmonary disease, bronchiectasis, sarcoidosis, or prior infections). Antifungal medicines are therefore the mainstay of treatment.
However, surgery may be considered in a small and carefully selected group of patients, most commonly when:
1. Disease is localised to one area of the lung
If the aspergillus infection is confined to a single cavity or one lobe, and the rest of the lungs are relatively healthy, it may be technically possible to remove the affected area.
2. Recurrent or life-threatening haemoptysis (coughing up blood)
Large-volume or repeated bleeding is one of the strongest reasons surgery is considered. In some cases, surgery is viewed as a way to prevent catastrophic bleeding, rather than to eradicate infection.
3. A simple aspergilloma
Patients with a simple aspergilloma (a single fungal ball in a cavity, minimal surrounding disease, and preserved lung function) are the group most likely to benefit.
4. Failure or intolerance of antifungal therapy
If antifungal drugs cannot be taken long term due to side effects, drug resistance, or lack of response—and the disease remains localised—surgery may be discussed.
Why surgery is often not recommended
Although surgery can sound appealing, CPA surgery is high-risk and not suitable for most patients.
1. CPA is often widespread
Many patients have a disease affecting both lungs or multiple lobes. Removing one area does not treat the remaining infection.
2. Underlying lung reserve is limited
CPA commonly occurs in people with reduced lung function. Removing lung tissue can lead to:
-
Long-term breathlessness
-
Oxygen dependence
-
Reduced quality of life
Even if the operation itself is technically successful.
3. Surgery carries significant risks
Compared with many other lung operations, CPA surgery has higher complication rates, including:
-
Prolonged air leaks
-
Serious infections
-
Bleeding
-
Bronchopleural fistula (abnormal airway–pleural connection)
-
Need for prolonged hospitalisation or intensive care
4. Surgery does not address the underlying vulnerability
CPA reflects an ongoing susceptibility of the lung environment. Removing one fungal focus does not remove the underlying reason aspergillus was able to grow in the first place.
What is the “success rate” of surgery?
Success depends heavily on patient selection and surgical expertise.
In specialist centres:
-
Operative mortality (risk of death around the time of surgery):
Typically reported between 1–5%, but higher in complex diseases. -
Major complication rates:
Often 15–40%, depending on disease extent and lung health. -
Symptom improvement:
Many patients selected for surgery experience reduced haemoptysis and improved local control of disease.
These figures are why surgery is only offered after careful multidisciplinary discussion, usually involving respiratory physicians, infectious disease specialists, thoracic surgeons, and radiologists.
Is surgery a “cure” for CPA?
This is one of the most misunderstood points.
Short answer: sometimes, but often not in the long term
-
In a simple aspergilloma, surgery can be genuinely curative if:
-
The disease is completely removed
-
There is no other active CPA elsewhere
-
The patient’s lungs remain stable
-
-
In chronic cavitary or fibrosing CPA, surgery is rarely a true cure. Instead, it may:
-
Control bleeding
-
Remove a particularly problematic area
-
Reduce fungal burden
-
Even after apparently successful surgery, some patients still require:
-
Long-term antifungal therapy
-
Ongoing monitoring with scans and blood tests
Recurrence of aspergillus infection elsewhere in the lungs can occur months or years later.
Why are many patients managed medically instead
For most people with CPA, long-term antifungal therapy offers:
-
Disease stabilisation
-
Symptom control
-
Lower risk than surgery
While antifungals do not usually “cure” CPA either, they can:
-
Slow or halt progression
-
Reduce inflammation and symptoms
-
Improve quality of life
This is why surgery is best seen as a highly selective tool, not a standard treatment.
How decisions about surgery are made
If surgery is discussed, your team will usually consider:
-
Extent and pattern of CPA on imaging
-
Lung function tests
-
General fitness and other medical conditions
-
History of haemoptysis
-
Response and tolerance to antifungal treatment
-
Your own priorities and acceptable trade-offs
Importantly, being told surgery is not advised does not mean your care is being limited—it usually reflects a judgement that risks outweigh benefits in your specific case.
Key messages for patients
-
Surgery for CPA is uncommon and highly selective
-
It is most useful in localised disease or severe bleeding
-
Complication rates are significant
-
A guaranteed or permanent “cure” is not typical, except in carefully chosen cases
-
Long-term medical management remains the safest and most effective option for most patients
If surgery has been mentioned—or ruled out—in your case, it is reasonable to ask your team:
-
What specific problem would surgery aim to solve for me?
-
What risks apply to my lungs and overall health?
-
Would antifungal treatment still be needed afterwards?
These discussions are an important part of shared decision-making in CPA care.
⭐ Chronic Pulmonary Aspergillosis: Why Diagnosis Is Missed and Who Needs to Be More Aware
With estimated prevalence of 3–4 cases per 100,000 population, and far higher rates in high-risk groups.
Chronic Pulmonary Aspergillosis (CPA) is a slowly progressive fungal lung disease affecting an estimated 3–4 per 100,000 people in the UK, with higher estimates in global settings with greater TB prevalence. Despite this, many clinicians will go through entire careers without confidently recognising it — not because it is extremely rare, but because it almost always hides inside other long-term lung diseases.
The UK is unusual in having a nationally commissioned specialist service — the National Aspergillosis Centre (NAC), based at Wythenshawe Hospital, Manchester — offering funded diagnostics, multidisciplinary review, and long-term antifungal management. But only a fraction of expected CPA cases are ever referred. Most are simply never diagnosed.
This article explains why diagnoses are missed, who is at highest risk, which specialities need to be more alert, and the red flags that should trigger testing or referral.
⭐ How Common Is CPA? The Numbers Behind the Problem
The UK prevalence is estimated at 3–4 per 100,000 people — approximately 2,000–2,500 people with CPA at any given time.
But the risk is far higher in specific groups:
| Risk Group | Estimated CPA prevalence |
|---|---|
| Post-TB lung disease | 6–10% in those with residual cavities |
| Severe COPD (GOLD III–IV) | 1–3% |
| Bronchiectasis | 1–3% |
| NTM disease | 3–10% |
| Sarcoidosis with fibrosis | 1–2% |
| Immunosuppression (steroids/biologics) | Unknown, but rising |
Using these figures, the true UK caseload could exceed 4,000–6,000 individuals, yet NAC receives ~500–1,000 referrals, highlighting a large diagnostic gap.
⭐ Why CPA Is So Often Missed
1. Symptoms mimic common chronic lung diseases
CPA presents with:
-
Persistent cough
-
Breathlessness
-
Fatigue
-
Weight loss
-
Recurrent “chest infections”
-
Haemoptysis
These overlap almost perfectly with:
-
COPD
-
bronchiectasis
-
post-TB changes
-
long COVID
-
NTM infection
-
repeatedly “slow to clear” pneumonia
Because symptoms are non-specific, clinicians rarely think fungal.
2. Interpretation of imaging is inconsistent
CPA shows:
-
one or more cavities
-
pleural thickening
-
nodules
-
progressive changes over months
-
fungal balls
Common reporting pitfalls:
-
labelled “post-infective scarring”
-
misinterpreted as malignancy
-
seen but not compared longitudinally
-
incidental CT findings not acted upon
Radiology is one of the biggest missed opportunities for early detection.
3. IgG testing is not routinely requested
Aspergillus IgG is the key diagnostic biomarker — but it is:
-
often confused with IgE
-
not available in some hospitals
-
omitted from workups for recurrent infection
-
unfamiliar to non-respiratory clinicians
Without IgG, CPA is rarely diagnosed.
4. Short-term improvement with antibiotics is misleading
Patients with CPA may temporarily feel better after:
-
broad-spectrum antibiotics
-
steroids
-
physiotherapy
This transient improvement creates false reassurance.
5. CPA spans multiple specialisms — and no one owns it
Diagnosis requires combined expertise across:
-
respiratory medicine
-
infectious diseases
-
radiology
-
microbiology
-
immunology
When no one speciality takes responsibility, patients get lost.
⭐ Which Patients Are at High Risk?
CPA almost always develops on a background of existing lung damage.
1. Post-TB lung disease (PTLD)
Globally the largest CPA population.
Residual cavities are the strongest predictor.
Specialities needing awareness:
-
TB teams
-
ID physicians
-
Radiologists
-
Community TB nurses
-
Public health TB programmes
2. COPD (especially severe / emphysema)
Millions of people are potentially at risk.
Recurrent infections + bullae/cavities = fertile ground for CPA.
Specialities:
-
COPD clinics
-
Pulmonary rehab
-
Acute medicine (frequent admissions)
3. Bronchiectasis
Damaged airways enable persistent Aspergillus colonisation and inflammation.
Specialities:
-
Bronchiectasis MDTs
-
Severe asthma & NTM clinics
-
Respiratory physiotherapy
4. Sarcoidosis and ILD
Fibrosis and traction bronchiectasis develop cavities over time.
5. Post-COVID or post-influenza structural disease
Emerging risk group, especially in patients with:
-
ventilatory lung injury
-
persistent CT abnormalities
-
chronic steroid exposure
6. Chronic steroid or immunomodulator use
While invasive aspergillosis is linked to profound immunosuppression, CPA often affects those with milder, chronic immune dysfunction:
-
systemic steroids
-
high-dose inhaled steroids
-
biologics affecting eosinophils
-
poorly controlled diabetes
-
chronic kidney disease
-
malnutrition
⭐ Which Specialities Need to Be More Alert?
-
Respiratory Medicine – primary detection, but awareness varies greatly
-
Infectious Diseases – especially post-TB and persistent infection clinics
-
Radiology – key to spotting early changes
-
Primary Care – sees patients repeatedly with “ongoing chest infections”
-
Emergency & acute medicine – haemoptysis presentations
-
Bronchiectasis and NTM services – strong overlap
-
Severe asthma and biologics teams – ABPA → CPA evolution
-
TB clinics – highest prevalence globally, often least recognised
The National Aspergillosis Centre should be the referral point for any complex or uncertain case.
⭐ Red Flags: When to Suspect CPA
1. Cavities on CT (thin-, thick-walled, evolving, or multiple)
Especially with pleural thickening.
2. Haemoptysis
CPA is one of the most common causes of haemoptysis in people with cavities.
3. Symptoms lasting >3 months
Chronic cough, fatigue, weight loss, breathlessness.
4. “Recurrent infections” that never fully resolve
5. Post-TB patient with any new or worsening symptoms
6. Bronchiectasis patient with new cavity or Aspergillus culture
7. High or rising Aspergillus IgG
8. ABPA patient who deteriorates off antifungals
⭐ The Cost of Missed Diagnoses
When CPA is not recognised early, the consequences are severe:
-
irreversible lung damage
-
repeated hospitalisations
-
emergency haemoptysis events
-
prolonged antifungal therapy with more toxicity
-
reduced quality of life
-
avoidable deaths
For systems like the NHS, late diagnosis increases costs:
-
unplanned admissions
-
repeated CT imaging
-
prolonged antibiotics
-
intensive care during haemoptysis
-
complex surgery (lobectomy/pneumonectomy)
Early referral to specialist centres like the National Aspergillosis Centre prevents many of these harms.
⭐ Conclusion
CPA is not rare within the populations most likely to develop it.
Missed diagnoses are common, predictable, and preventable.
By increasing awareness across Respiratory, Infectious Diseases, Radiology, Primary Care, TB services, and severe asthma pathways — and by using simple tools such as Aspergillus IgG and careful CT interpretation — clinicians can dramatically reduce the diagnostic delay that damages lungs, quality of life, and survival.
The UK is fortunate to have the National Aspergillosis Centre as a nationally commissioned referral service. Recognising CPA early and referring appropriately has the power to save lives, reduce system costs, and improve long-term outcomes.