When Breathlessness Feels Severe — Even When Oxygen Levels Look “Normal”

Many people living with aspergillosis, severe asthma, bronchiectasis, or other chronic lung conditions describe a confusing and sometimes frightening experience:
“My oxygen saturations are normal, my peak flow is reasonable, there’s little wheeze, but I still feel like I’m drowning.”
This can be distressing for patients and frustrating for carers. Some people feel that because their oxygen levels or breathing tests appear “acceptable”, their symptoms are not fully understood.
Importantly, severe breathlessness can occur even when standard measurements such as oxygen saturations and peak flow readings appear relatively normal.
This does not mean the symptoms are imaginary or “all in the mind”. Breathlessness is complex and can have many different causes.
Why Breathlessness Is More Complicated Than Oxygen Levels
When doctors or nurses assess breathing problems, they often check:
- Oxygen saturation levels (sats)
- Peak flow readings
- Respiratory rate
- Presence of wheeze
- Chest sounds
These are all important. However, they do not always reflect how breathless a person feels.
Some people with chronic respiratory illness may have:
- Normal oxygen saturations
- Reasonable peak flow readings
- Little visible wheeze
- Minimal mucus production
…yet still experience intense sensations of:
- air hunger
- tight chest
- difficulty taking a satisfying breath
- feeling unable to “fill the lungs”
- panic associated with breathing
- extreme fatigue from breathing effort
What Can Cause This?
Breathlessness in aspergillosis and chronic lung disease is often caused by several factors happening together.
Inflammation and Airway Sensitivity
Conditions such as Allergic Bronchopulmonary Aspergillosis (ABPA), Severe Asthma with Fungal Sensitisation (SAFS), bronchiectasis, and Chronic Pulmonary Aspergillosis (CPA) can all cause inflamed and hypersensitive airways.
The lungs may feel irritated or tight even if oxygen exchange remains relatively preserved.
Small Airways Dysfunction
Some breathing problems occur in the smaller airways of the lungs and may not always show clearly on basic tests such as peak flow.
Patients can feel significant chest tightness or air trapping despite “good numbers”.
Muscle Fatigue
Breathing takes muscular effort. Chronic respiratory illness can place a long-term strain on the chest wall and breathing muscles, leading to exhaustion and increased awareness of breathing.
Mucus and Airflow Changes
Even relatively small amounts of mucus or airway narrowing can create sensations of chest heaviness or difficulty moving air.
Breathing Pattern Dysfunction (Dysfunctional Breathing)
This is increasingly recognised in people with chronic respiratory illness.
When breathing becomes difficult over months or years, people may unconsciously develop altered breathing patterns, including:
- rapid shallow breathing
- upper chest breathing
- frequent sighing
- over-breathing (hyperventilation)
- muscle tension around the chest and neck
This can worsen symptoms and create a vicious cycle where the sensation of breathlessness becomes amplified.
Symptoms may include:
- air hunger
- dizziness
- tingling
- tight chest
- panic sensations
- difficulty “switching off” breathing awareness
Importantly, this does not mean the illness is psychological or “not real”. Dysfunctional breathing can happen alongside genuine lung disease.
Why Inhalers Sometimes Seem to Help Less
Reliever inhalers such as salbutamol (Ventolin) are designed mainly to relax tightened airways.
If breathlessness is being driven partly by breathing pattern dysfunction, muscle fatigue, inflammation, hypersensitivity, or air trapping, inhalers may not always provide dramatic relief.
This can be confusing and upsetting for patients.
“But My Tests Are Normal…”
Many patients feel frustrated, frightened, or even dismissed when told that oxygen levels, peak flow readings, or chest examinations are “fine” despite severe breathlessness.
Normal oxygen saturations are reassuring in terms of immediate danger, but they do not always reflect the full experience of chronic respiratory illness.
Breathlessness is influenced by many factors including inflammation, airway sensitivity, breathing effort, muscle fatigue, anxiety associated with struggling to breathe, and altered breathing patterns.
Because of this, some people may feel profoundly breathless even when routine measurements appear relatively stable.
It can sometimes help to explain symptoms in practical, functional terms, such as:
- “I become breathless walking across the room.”
- “I recover much more slowly than usual.”
- “This feels much worse than my normal baseline.”
- “I feel exhausted by the effort of breathing.”
- “Breathing exercises seem to help settle things.”
These descriptions may help healthcare professionals understand how symptoms are affecting day-to-day life, rather than focusing only on oxygen levels or peak flow numbers.
Some patients also find it helpful to ask questions such as:
- Could breathing pattern dysfunction be contributing?
- Would respiratory physiotherapy help?
- Would pulmonary rehabilitation be appropriate?
- How should I judge when symptoms need urgent assessment?
Importantly, severe breathlessness should never simply be ignored. New, worsening, or unusual symptoms still require proper medical assessment.
At the same time, many people with chronic lung disease experience very real symptoms that are not always fully reflected by routine measurements alone.
Why Breathlessness Can Feel So Frightening
The sensation of breathlessness is created by the brain interpreting signals from the lungs, breathing muscles, chest wall, blood chemistry, and nervous system.
This means that the feeling of “not getting enough air” is not determined only by oxygen levels.
In chronic lung disease, several things can trigger the sensation of breathlessness, including:
- inflamed or sensitive airways
- extra effort needed to move air in and out
- air trapping in the lungs
- muscle fatigue
- rapid or shallow breathing
- stress hormones released during breathing distress
- heightened awareness of breathing sensations
When breathing becomes uncomfortable, the body naturally responds with anxiety and adrenaline. This is a protective survival response.
Unfortunately, this can sometimes create a cycle:
breathlessness → anxiety → faster breathing → more chest tightness → worse breathlessness
This does not mean symptoms are “psychological”. The physical sensation is real, but the body’s alarm systems can unintentionally amplify it.
What Can Help During an Episode of Breathlessness?
Different techniques help different people, and severe or rapidly worsening symptoms should always be medically assessed. However, some patients find the following approaches helpful during episodes of distressing breathlessness:
Slow the Breathing Rate
Trying to slow breathing gently can help reduce over-breathing and chest tightness.
Some people find it helpful to:
- breathe in gently through the nose
- breathe out slowly through pursed lips
- focus on making the out-breath longer than the in-breath
Use a Recovery Position
Sitting forward slightly with the arms supported on knees or a table can sometimes reduce the work of breathing.
Reduce Panic and “Air Hunger”
Trying to fight for bigger and bigger breaths can sometimes worsen symptoms.
Some patients find it more helpful to focus on:
- gentle breathing rhythm
- relaxing the shoulders and neck
- slowing breathing rather than deepening it
- focusing attention away from the chest where possible
Use Prescribed Treatments Appropriately
Follow the advice provided by your healthcare team regarding inhalers, nebulisers, airway clearance, or rescue medication.
If inhalers are not helping as expected, this should be discussed with a respiratory specialist rather than simply increasing use repeatedly.
Know Your “Usual” Pattern
Many patients find it useful to learn the difference between:
- their “usual” chronic breathlessness
- breathing pattern dysfunction or over-breathing episodes
- symptoms suggesting infection or acute deterioration
This can help patients feel more confident recognising when urgent medical assessment may be needed.
Can Breathing Retraining Help?
Some patients find breathing retraining exercises very helpful, especially when guided by:
- respiratory physiotherapists
- specialist breathing services
- pulmonary rehabilitation teams
- asthma nurse specialists
Breathing retraining may include:
- slowing breathing rate
- diaphragmatic (“belly”) breathing
- nasal breathing techniques
- recovery breathing positions
- relaxation techniques
- paced activity and pacing strategies
Some NHS respiratory teams recommend online breathing resources and guided exercises to help patients recognise and manage over-breathing patterns.
These approaches are usually intended to work alongside medical treatment — not instead of it.
Living With an “Invisible” Symptom
One of the hardest aspects of chronic breathlessness is that outward signs may not always match how severe symptoms feel internally.
Many patients report feeling dismissed when oxygen levels are normal or when tests appear “better than expected”.
The experience of breathlessness is real, even when routine measurements do not fully explain it.
This is one reason why specialist respiratory assessment can be important in complex conditions such as aspergillosis.
When to Seek Medical Help
You should seek urgent medical advice if breathlessness is:
- suddenly worsening
- associated with chest pain
- causing blue lips or fingertips
- associated with falling oxygen saturations
- accompanied by fever or signs of infection
- causing confusion or severe exhaustion
- significantly different from your usual symptoms
Even if previous episodes have been related to breathing pattern dysfunction, new or worsening symptoms should still be medically assessed.
Further Support
You may also find these resources helpful:
Last reviewed: May 2026
Produced by: National Aspergillosis Centre CARES Team / Aspergillosis Website
Asthma and Aspergillosis
How fungal spores interact with asthma and other lung diseases
Every day we inhale thousands of microscopic fungal spores from the environment. One of the most common fungi in the air is Aspergillus fumigatus. In healthy lungs these spores are removed quickly by the lungs’ natural defence systems and cause no illness.
However, in people with asthma—particularly severe asthma—the interaction between the lungs and Aspergillus can be very different. The fungus may trigger allergic inflammation, grow in mucus within the airways, or occasionally contribute to chronic lung disease.
Understanding this relationship helps explain several important conditions including:
-
Aspergillus sensitisation
-
Severe Asthma with Fungal Sensitisation (SAFS)
-
Allergic Bronchopulmonary Aspergillosis (ABPA)
-
Aspergillus bronchitis
-
Chronic Pulmonary Aspergillosis (CPA)
Although asthma is the most common condition linked to Aspergillus allergy, other lung diseases such as bronchiectasis, Chronic Obstructive Pulmonary Disease (COPD), and tuberculosis-related lung damage can also create environments where the fungus becomes important.
Why Asthma Creates a Favourable Environment for Aspergillus
Asthma is a disease of airway inflammation and hyper-reactivity. The bronchi narrow during attacks because the airway wall becomes swollen and the surrounding smooth muscle contracts.
Several features of asthma make it easier for Aspergillus spores to remain in the lungs.
Mucus production
Asthma often causes increased production of thick airway mucus.
Normally mucus traps inhaled particles and moves them upward toward the throat via the mucociliary escalator.
In asthma:
-
mucus becomes thicker
-
clearance becomes less efficient
-
spores remain trapped
This trapped environment allows fungal spores to persist in the airway mucus.
Allergic immune responses
Many asthma patients have Type-2 (T2) inflammation (50-70%), involving immune pathways driven by:
-
Immunoglobulin E (IgE)
-
Interleukin-4
-
Interleukin-5
-
Interleukin-13
-
eosinophils
These pathways respond strongly to fungal allergens. When the immune system recognises Aspergillus proteins it may trigger allergic inflammation in the airways.
Fungal sensitisation is increasingly recognised as an important contributor to severe asthma (PMID: 24735832).
Aspergillus Sensitisation
Many people with asthma develop allergic sensitisation to Aspergillus.
Sensitisation means the immune system produces antibodies against fungal proteins.
Features include:
-
positive Aspergillus skin test or IgE blood test
-
worsening asthma symptoms
-
increased exacerbations
Studies suggest 10–25% of patients attending severe asthma clinics show Aspergillus sensitisation (PMID: 24735832).
However, sensitisation alone does not necessarily cause lung damage.
Severe Asthma with Fungal Sensitisation (SAFS)
Some patients with severe asthma have fungal sensitisation but do not meet the criteria for ABPA.
This condition is known as Severe Asthma with Fungal Sensitisation (SAFS).
Typical features include:
-
severe or poorly controlled asthma
-
fungal allergy
-
moderate IgE elevation
A randomised controlled trial demonstrated that antifungal therapy may improve symptoms in some SAFS patients (PMID: 18948425).
Allergic Bronchopulmonary Aspergillosis (ABPA)
Allergic Bronchopulmonary Aspergillosis is the most important Aspergillus-related disease associated with asthma.
ABPA occurs when Aspergillus grows within airway mucus and triggers a strong allergic immune response.
Typical findings include:
-
very high total IgE levels
-
Aspergillus-specific IgE and IgG antibodies
-
eosinophilia
-
mucus plugs containing fungal hyphae
-
central bronchiectasis
ABPA occurs in approximately:
-
1–2% of all asthma patients
-
up to 10–15% of severe asthma patients
These figures come from global prevalence estimates of ABPA in asthma populations (PMID: 23210682/.
Modern diagnostic criteria for ABPA were updated by the International Society for Human and Animal Mycology (ISHAM) in 2024 (PMID: 38423624).
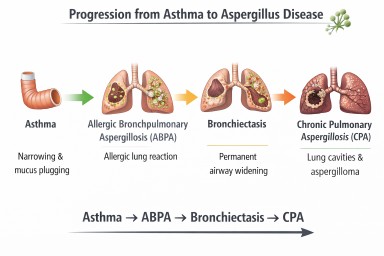
Asthma and Aspergillus Disease Pathway
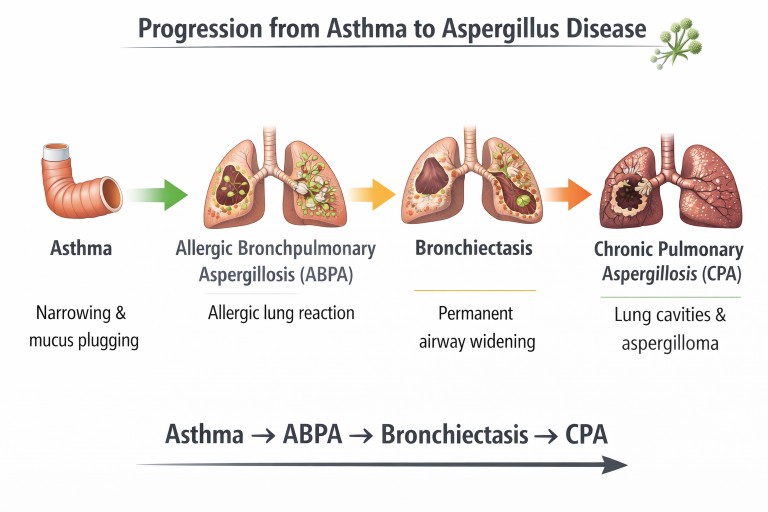
Possible interactions between asthma and Aspergillus. Some patients develop allergic disease (ABPA) which may lead to airway damage such as bronchiectasis (NB Progression to CPA is very rare).
When ABPA Causes Bronchiectasis
Repeated inflammation from ABPA may damage airway walls and lead to bronchiectasis.
Bronchiectasis occurs when airways become:
-
permanently widened
-
distorted
-
unable to clear mucus effectively
Instead of being cleared from the lungs, mucus pools in the airways.
This retained mucus creates an environment where microorganisms—including fungi—can grow.
Aspergillus Bronchitis
In some patients with bronchiectasis or chronic lung disease, Aspergillus may persist in airway mucus and cause chronic airway infection rather than allergy.
Symptoms may include:
-
chronic cough
-
sputum production
-
repeated positive Aspergillus cultures
IgE levels are usually lower than in ABPA.
Chronic Pulmonary Aspergillosis (CPA)
Chronic Pulmonary Aspergillosis is a slowly progressive fungal infection of damaged lung tissue.
CPA usually develops in lungs containing:
-
cavities
-
severe structural damage
Common underlying diseases include:
-
tuberculosis
-
sarcoidosis
-
severe COPD
Globally, the most common cause of CPA is previous tuberculosis infection (PMID: 22271943).
Asthma alone rarely causes CPA, but severe bronchiectasis or ABPA-related lung damage may occasionally lead to it.
Aspergillosis and Immune Competence
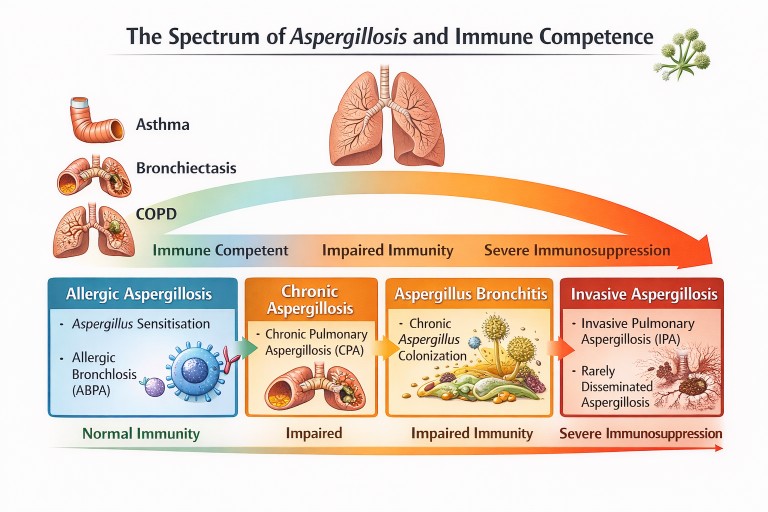
Different forms of aspergillosis occur depending on lung damage and immune function.
Other Lung Diseases Linked to Aspergillus
Although asthma is the most common condition associated with Aspergillus allergy, several other lung diseases can predispose to fungal disease.
Bronchiectasis
Dilated airways trap mucus, allowing fungi and bacteria to persist.
COPD
Chronic airway inflammation may lead to Aspergillus bronchitis or chronic pulmonary aspergillosis.
Tuberculosis
Post-tuberculosis lung cavities are the most common global cause of chronic pulmonary aspergillosis (PMID: 22271943).
Key Messages
-
Asthma is one of the most important diseases associated with Aspergillus-related lung conditions.
-
Many asthma patients develop fungal sensitisation.
-
A smaller proportion develop Allergic Bronchopulmonary Aspergillosis (ABPA).
-
Repeated inflammation from ABPA can lead to bronchiectasis.
-
Chronic pulmonary aspergillosis is rare in asthma alone but may occur if significant lung damage develops.
Understanding these interactions helps guide diagnosis and treatment for people living with asthma and Aspergillus-related disease.
Further reading
Agarwal R, Chakrabarti A, Shah A, Gupta D, Meis JF, Guleria R, Moss R, Denning DW; ABPA complicating asthma ISHAM working group. Allergic bronchopulmonary aspergillosis: review of literature and proposal of new diagnostic and classification criteria. Clin Exp Allergy. 2013 Aug;43(8):850-73. doi: 10.1111/cea.12141. PMID: 23889240.
Denning DW, Pleuvry A, Cole DC. Global burden of chronic pulmonary aspergillosis as a sequel to pulmonary tuberculosis. Bull World Health Organ. 2011 Dec 1;89(12):864-72. doi: 10.2471/BLT.11.089441. Epub 2011 Sep 27. PMID: 22271943; PMCID: PMC3260898.
Looking further into the future - could we control lung damage, preserve healthy lung tissue better?
Can Lungs Repair Themselves?
What New Research Means for People with CPA (and Other Aspergillosis)
A recent scientific discovery has helped researchers understand how certain lung cells decide whether to focus on repairing damage or defending against infection. The work, highlighted by the Mayo Clinic and published in Nature Communications, describes a molecular “switch” inside specialised lung cells that influences this balance.
For people living with Chronic Pulmonary Aspergillosis (CPA) — and also those with Allergic Bronchopulmonary Aspergillosis (ABPA) — this kind of research is relevant. But it needs careful explanation.
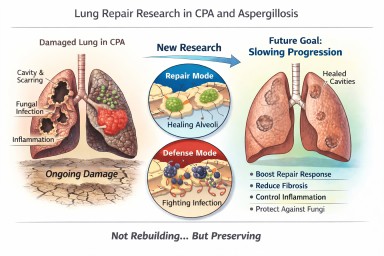
This is not about rebuilding destroyed lungs.
It is about understanding how to better protect and preserve the lung tissue that remains.
The Discovery: A “Repair vs Defence” Switch
Researchers identified a regulatory circuit in alveolar type II (AT2) cells — specialised cells that:
-
Produce surfactant (which keeps air sacs open)
-
Act as a reserve “repair” population in the lung
-
Can regenerate other essential lung cells after injury
The study showed that these cells operate under tight control. When infection is present, they prioritise defence. When injury needs healing, they can switch into repair mode.
The key insight is that this switch is biologically regulated. It is not random. That means, in theory, it may one day be possible to influence it.
What “Repair” Means — and What It Does Not Mean
When we talk about lung repair in this context, we must be very clear.
It does not mean:
-
Lung cavities caused by CPA will close in the foreseeable future
-
Established fibrosis will melt away
-
Bronchiectasis will reverse
-
Severely distorted lung architecture will rebuild
CPA cavities represent major structural remodelling — destruction of alveoli, scarring, altered blood supply, and thickened pleura. Reconstructing that complex architecture is biologically extremely challenging and not currently realistic within the next decade.
What repair does realistically mean
In chronic lung disease, “repair” is more likely to mean:
-
Supporting survival of remaining alveoli
-
Preventing excessive fibrotic signalling
-
Helping lung lining cells recover more efficiently after inflammation
-
Reducing cumulative injury from repeated infection
-
Slowing progression of structural change
In other words:
Not rebuilding what is gone — but better protecting what remains.
For many people with CPA, this is a crucial distinction.
Why Preservation Is a Major Goal in CPA
CPA usually develops in lungs already weakened by conditions such as tuberculosis, non-tuberculous mycobacteria, chronic obstructive pulmonary disease, or severe pneumonia.
Over time, CPA can lead to:
-
Expanding cavities
-
Progressive scarring
-
Reduced gas exchange
-
Reduced exercise tolerance
Many patients have limited lung reserve. Even small additional losses of functioning lung tissue can significantly increase breathlessness or fatigue.
If future therapies could slow the rate of progression — even modestly — that would meaningfully affect long-term outcomes.
Flattening the decline curve is not trivial. It changes quality of life.
Why This Also Matters in ABPA
In ABPA, repeated inflammatory episodes can lead to:
-
Airway remodelling
-
Mucus plugging
-
Development or progression of bronchiectasis
Better control of inflammatory signalling — combined with improved epithelial recovery — could reduce long-term airway damage.
Again, this is about preservation rather than reversal.
Where Development Has Reached
The current research is still laboratory-based. It used advanced techniques such as:
-
Single-cell sequencing
-
Imaging of lung tissue
-
Preclinical models of injury
No human treatments based on this discovery are yet available.
However, the significance lies in identifying:
-
A defined molecular pathway
-
A controllable regulatory mechanism
-
A clearer understanding of why repair fails in chronic inflammation
That foundational knowledge is what eventually allows targeted drug development.
The Balance Challenge in Aspergillosis
There is an additional complexity in fungal lung disease.
Any attempt to promote repair must not weaken antifungal defence.
The immune system must:
-
Control Aspergillus
-
Avoid causing excessive inflammatory damage
Future therapies would need to strike that balance carefully.
What This Means for Patients Now
This discovery does not change current treatment.
The most effective preservation strategies today remain:
-
Consistent antifungal therapy when indicated
-
Careful inflammatory control
-
Biologic therapies where appropriate
-
Airway clearance
-
Vaccination and infection prevention
-
Avoiding damp and mould exposure
-
Pulmonary rehabilitation
These measures are already forms of lung preservation.
A Realistic and Hopeful Perspective
It is unlikely that cavities from CPA will be repaired in the near future.
It is realistic that within the next 5–10 years we may see improved strategies aimed at:
-
Slowing structural progression
-
Supporting endogenous repair cells
-
Reducing fibrotic signalling
-
Improving recovery after exacerbations
For people living long-term with CPA or ABPA, even incremental preservation could significantly affect independence and quality of life.
The science is still early — but understanding how the lung decides to repair itself is an important step forward.
Reference
Sawhney, A.S., Deskin, B.J., Cai, J. et al. A molecular circuit regulates fate plasticity in emerging and adult AT2 cells. Nat Commun 16, 8924 (2025). https://doi.org/10.1038/s41467-025-64224-1
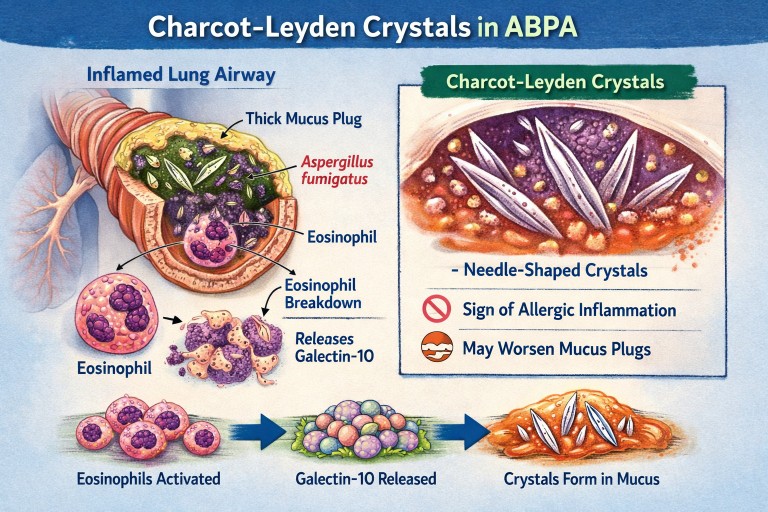
🧬 Could Antibody-Driven Dissolving of Charcot–Leyden Crystals Help ABPA?
Researchers have recently discovered that Charcot–Leyden crystals (CLCs) — the needle-shaped structures formed from the eosinophil protein galectin-10 — are not just debris.
In laboratory studies, specially designed antibodies can dissolve these crystals.
This has raised two important questions:
-
Could dissolving the crystals reduce airway inflammation?
-
Could dissolving them make mucus plugs easier to clear?
Here is what we currently know.
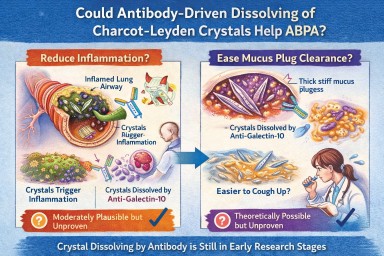
1️⃣ Could dissolving crystals reduce airway inflammation?
What we know
Laboratory and animal studies have shown:
-
Charcot–Leyden crystals can activate immune cells (especially macrophages).
-
They can stimulate inflammatory pathways (including inflammasome signalling).
-
In mouse models, antibodies targeting galectin-10 dissolved the crystals.
-
When crystals were dissolved, airway inflammation decreased.
This suggests that the crystals themselves may amplify inflammation, rather than simply mark it.
What this means biologically
In ABPA and eosinophilic asthma:
-
Eosinophils release galectin-10.
-
Galectin-10 crystallises.
-
Crystals may trigger further immune activation.
-
That leads to more inflammation → more eosinophils → more crystals.
Dissolving the crystals could theoretically interrupt this feedback loop.
How likely is this to help inflammation in humans?
Moderately plausible, but not yet proven.
The biological mechanism is strong.
The animal data are encouraging.
But no human clinical trials have yet shown reduced inflammation through crystal dissolution.
If developed successfully, this approach could:
-
Reduce airway immune activation
-
Lower exacerbation risk
-
Potentially reduce steroid dependence
But at present, it remains investigational.
2️⃣ Could dissolving crystals make mucus plugs easier to cough up?
This is more speculative — but still biologically reasonable.
Why mucus plugs are so thick in ABPA
ABPA mucus plugs contain:
-
Gel-forming mucins
-
DNA from inflammatory cells
-
Dead cells
-
Fungal fragments
-
Eosinophil proteins
-
Charcot–Leyden crystals
The crystals are:
-
Rigid
-
Needle-shaped
-
Structurally stable
When embedded in mucus, they likely increase:
-
Mechanical stiffness
-
Plug density
-
Resistance to deformation
From a physics perspective:
Removing rigid crystalline structures from a gel should reduce stiffness and improve flow.
Do we have direct evidence?
No.
There are currently:
-
No human studies measuring mucus clearance after crystal dissolution
-
No trials showing improved plug expectoration from crystal-targeting therapy
So while it is plausible that dissolving crystals could soften plugs, this has not yet been demonstrated in patients.
3️⃣ How strong is the overall case?
| Outcome | Evidence strength | Likelihood |
|---|---|---|
| Reduced inflammation | Strong biological rationale + animal data | Moderately promising |
| Easier mucus clearance | Biophysical plausibility only | Possible but unproven |
Inflammation reduction is the more evidence-supported target.
Improved plug clearance is plausible but currently theoretical.
4️⃣ How does this compare to existing treatments?
Current therapies (e.g., anti-IL-5 biologics) reduce eosinophils upstream.
That leads to:
-
Less galectin-10 release
-
Fewer crystals forming
-
Reduced inflammation
-
Often improved mucus plugging
So biologics already indirectly reduce crystal burden.
A crystal-dissolving antibody would act downstream, targeting the structural product directly.
This could theoretically:
-
Accelerate resolution of existing plugs
-
Reduce residual inflammatory signalling
But again, this remains in early research stages.
5️⃣ Practical take-home message
At present:
-
Dissolving Charcot–Leyden crystals reduces inflammation in animal models.
-
It is biologically plausible that this could also soften mucus plugs.
-
There is no human clinical proof yet.
-
No approved therapy currently targets the crystals directly.
The concept is scientifically credible — but still under development.
🔭 The Bigger Picture
ABPA is increasingly understood as a condition driven by:
-
Eosinophils
-
Allergic immune signalling
-
Abnormal mucus biology
-
Structural plug formation
Crystal-targeting therapies may eventually become part of a more precise approach to treating eosinophilic airway disease.
But for now, they remain a promising research direction rather than a clinical option.
🔬 Charcot–Leyden Crystals in ABPA and Asthma
What are they? Why do they form? Do they matter?
If you live with Allergic Bronchopulmonary Aspergillosis (ABPA) or severe asthma, you may see the term Charcot–Leyden crystals in a sputum or pathology report.
They can sound worrying.
They are:
-
Not fungus
-
Not infection
-
Not cancer
They are a sign of a particular type of allergic inflammation in the airways.
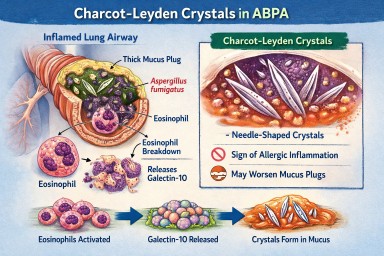
🧬 What Are Charcot–Leyden Crystals?
Charcot–Leyden crystals are microscopic, needle-shaped structures found in mucus.
They are made from a protein called galectin-10, which is stored inside a type of white blood cell called an eosinophil.
Eosinophils are immune cells involved in:
-
Allergic asthma
-
ABPA
-
Severe asthma with fungal sensitisation
-
Parasitic infections
When eosinophils are activated and break down, they release galectin-10.
If enough of this protein accumulates in thick airway mucus, it crystallises into visible crystals.
So the crystals are made from your immune cells, not from Aspergillus.
🫁 Why Do They Appear in ABPA?
In ABPA:
-
The immune system overreacts to Aspergillus fumigatus.
-
This triggers a strong allergic (Type 2) immune response.
-
Large numbers of eosinophils move into the airways.
-
Eosinophils break down and release galectin-10.
-
The protein crystallises inside mucus plugs.
The crystals are therefore a footprint of intense allergic inflammation, not fungal invasion.
🌡 Is Most ABPA Eosinophilic?
Yes — almost all classical ABPA is eosinophilic.
ABPA is fundamentally a Type 2 allergic condition, driven by immune pathways involving:
-
IL-4
-
IL-5
-
IL-13
-
IgE
-
Eosinophils
IL-5 in particular stimulates eosinophil production and survival.
Because of this, eosinophils are central to the disease process.
Historically, raised blood eosinophils have been part of diagnostic criteria.
However:
-
Eosinophil counts can fluctuate
-
Steroids can suppress blood levels
-
Eosinophils may still be present in airway mucus even if blood counts appear normal
So ABPA is biologically eosinophilic — even if a single blood test does not show a high count.
True non-eosinophilic ABPA would be unusual and would prompt clinicians to reconsider the diagnosis.
❓ Are Crystals Caused by Aspergillus Infection?
No.
They are caused by the immune reaction to Aspergillus — not by the fungus itself.
They can also be seen in:
-
Severe eosinophilic asthma
-
Parasitic infections
-
Other allergic lung conditions
They reflect eosinophil activity, not fungal growth.
🧠 Why Don’t All People with Asthma Develop These Crystals?
Asthma is not one single disease. It has different inflammatory patterns.
Type 2 (Eosinophilic) Asthma
This involves high eosinophils and allergic pathways.
Common in:
-
Allergic asthma
-
ABPA
-
Severe eosinophilic asthma
These patients can develop Charcot–Leyden crystals.
Non–Type 2 (Non-Eosinophilic) Asthma
This includes:
Neutrophilic asthma
Driven by neutrophils rather than eosinophils.
Paucigranulocytic asthma
Very few inflammatory cells present.
In these forms:
-
Eosinophils are low
-
Galectin-10 is not released in large amounts
-
Crystals are unlikely to form
🧱 Do Charcot–Leyden Crystals Make Mucus Plugs Worse?
Possibly.
Research suggests they may:
-
Increase mucus thickness
-
Contribute mechanically to airway blockage
-
Stimulate further inflammation
For many years they were thought to be harmless debris.
Modern studies suggest they may actively amplify inflammation when present in large amounts.
🎯 Do They Have a Purpose?
Eosinophils evolved mainly to help fight parasitic infections.
Galectin-10 probably has immune signalling roles inside cells.
However, when large amounts are released into thick airway mucus, crystallisation appears to be a by-product of excessive immune activity rather than a useful defence.
In ABPA and allergic asthma, they are more likely part of the problem than part of the solution.
💧 Can Their Formation Be Reduced?
Hydration alone does not stop them forming.
Drinking fluids helps:
-
Keep mucus less sticky
-
Support airway clearance
But it does not prevent eosinophils releasing galectin-10.
What reduces crystal formation?
Reducing eosinophilic inflammation:
-
Corticosteroids
-
Anti-IL-5 biologics
-
Anti-IL-4/IL-13 biologics
When eosinophil numbers fall:
→ Less galectin-10 is released
→ Fewer crystals form
Antifungal treatment in ABPA may indirectly help by reducing allergic stimulation, but the main driver is the immune response.
📊 Do They Change Treatment?
Not directly.
Doctors base treatment on:
-
Symptoms
-
Blood eosinophils
-
Total IgE
-
Imaging
-
Lung function
-
Exacerbation history
Crystals support the diagnosis of eosinophilic inflammation but do not determine treatment alone.
🔎 What Do They Tell Us?
Charcot–Leyden crystals tell us:
-
The airway inflammation is eosinophilic.
-
The immune response is strongly allergic.
-
Mucus plugging risk may be higher.
They are a marker of immune overreaction, not infection severity.
🧠 Key Points to Remember
-
They are made from proteins released by eosinophils.
-
They are not Aspergillus.
-
They do not mean invasive fungal infection.
-
Most classical ABPA is eosinophilic.
-
They are unlikely in non-eosinophilic asthma.
-
Reducing eosinophils reduces their formation.
-
Hydration helps clearance but does not prevent formation.
In simple terms:
Charcot–Leyden crystals are microscopic signs that the immune system is working too hard in the airways.
Airways mucus and aspergillosis
A clear, patient-friendly explainer
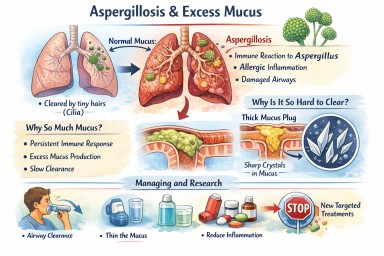
People living with aspergillosis often say that mucus is one of the hardest symptoms to manage — thick sputum, coughing fits, plugs that feel “stuck”, and flare-ups that seem to come out of nowhere. This explainer brings everything together in one place: what mucus is for, why aspergillosis causes so much of it, why it becomes abnormal, and what current and future treatments aim to do.
1. What is airway mucus and why do we need it?
Mucus is normal, healthy, and essential. Everyone produces it all the time.
Its main roles are to:
-
Trap inhaled particles (dust, spores, bacteria, pollution)
-
Protect the airway lining from drying and irritation
-
Support the immune system
-
Clear the lungs, using tiny moving hairs (cilia) that sweep mucus upwards so it can be swallowed or coughed out
(this clearance system is called the mucociliary escalator)
In healthy lungs:
-
Mucus is thin
-
Produced in small amounts
-
Cleared without you noticing it
2. Why aspergillosis causes excessive mucus
In aspergillosis, the lungs are under ongoing stress. Several factors combine:
Persistent immune activation
The immune system keeps reacting to Aspergillus material in the airways. Even when the fungus is controlled, inflammation can persist.
Allergic-type inflammation (especially in ABPA)
Allergic immune responses strongly stimulate mucus-producing cells, leading to:
-
Large volumes of mucus
-
Very sticky or rubbery sputum
Airway damage
Conditions commonly associated with aspergillosis (such as bronchiectasis or long-standing asthma) cause:
-
Widened or damaged airways
-
Poor mucus clearance
-
Pools of mucus that are hard to shift
Slowed clearance
Inflammation and infection impair cilia, so mucus:
-
Moves more slowly
-
Sits in the lungs longer
-
Becomes thicker and harder to clear
➡️ What starts as a protective response becomes a self-perpetuating problem.
3. Why thick mucus causes symptoms
Excess or abnormal mucus can:
-
Block airways → breathlessness and wheeze
-
Trigger coughing → especially overnight or on waking
-
Trap infection → repeated flare-ups
-
Reduce oxygen exchange
-
Increase fatigue and chest discomfort
Many patients describe it as:
“Glue-like”, “stringy”, “rubbery”, or “impossible to move”
4. Mucus plugs and crystals – why some mucus is so hard to clear
Mucus plugs
When mucus becomes very thick, it can:
-
Form plugs that partially or completely block airways
-
Show up on CT scans
-
Worsen breathlessness suddenly
Charcot–Leyden crystals
In allergic and eosinophilic airway disease (including allergic bronchopulmonary aspergillosis):
-
Breakdown products of allergic immune cells can form microscopic crystals
-
These crystals make mucus:
-
Stiffer
-
More irritating
-
Harder to clear
-
Their presence is a sign of ongoing allergic inflammation, not infection alone.
5. Why managing mucus really matters
Mucus is not just an inconvenience. Poor mucus control can:
-
Increase infection risk
-
Drive repeated exacerbations
-
Worsen lung damage over time
-
Reduce quality of life and sleep
-
Increase hospital admissions
For aspergillosis, mucus management is core treatment, not optional.
6. What helps now (current approaches)
A. Thin the mucus
-
Good hydration
-
Nebulised saline (normal or hypertonic)
-
Selected mucolytic medicines (used carefully)
B. Move it out
-
Regular airway clearance physiotherapy
-
Breathing techniques (e.g. active cycle breathing)
-
Oscillating devices (flutter, Acapella, Aerobika)
-
Gentle, regular physical activity where possible
C. Reduce inflammation
-
Inhaled corticosteroids (when appropriate)
-
Oral steroids (used cautiously)
-
Biologic therapies for selected allergic or eosinophilic disease
-
Antifungal treatment when fungal burden is contributing
D. Treat infections early
-
Bacterial infections thicken mucus further
-
Prompt treatment reduces long-term damage
7. What research is doing differently (emerging therapies)
Research is moving beyond simply “loosening mucus”.
1. Reducing mucus production at source
Scientists are developing drugs that aim to:
-
Switch off excessive mucus secretion
-
Preserve normal protective mucus
This targets the mucus-producing cells directly.
2. Blocking the signals that drive over-production
Inflammation sends chemical signals telling airways to make more mucus. New treatments aim to:
-
Calm allergic and immune pathways
-
Prevent expansion of mucus-producing cells
Some current biologic therapies already reduce mucus indirectly; future drugs will be more precise.
3. Changing mucus structure
Instead of thinning everything, researchers are studying ways to:
-
Loosen the internal “mesh” of mucus
-
Prevent dense plugs from forming
-
Restore normal movement by cilia
4. Targeting mucus crystals
In allergic aspergillosis, research is exploring how to:
-
Reduce crystal formation
-
Calm the specific immune responses that create them
5. New inhaled and physical approaches
Early trials are testing:
-
Inhaled therapies designed to mobilise secretions
-
Treatments that improve airflow behind mucus plugs
6. Precision medicine
Future mucus treatments are likely to be:
-
Personalised
-
Based on inflammation type, fungal involvement, airway damage, and immune markers
Two people with aspergillosis may have very different mucus drivers — and need different solutions.
8. What this means for patients today
-
There is no single “anti-mucus cure” yet
-
Promising therapies are in research and early trials
-
Safety and long-term effects must be proven first
For now:
-
Regular airway clearance remains essential
-
Treating inflammation and infection promptly is crucial
-
Understanding why your mucus behaves as it does helps guide treatment
Key messages to remember
-
Mucus is normally protective
-
Aspergillosis turns a helpful system into a problem
-
Thick, sticky mucus reflects ongoing inflammation and airway damage
-
Crystals signal allergic involvement, not just infection
-
Research is moving toward preventing abnormal mucus formation, not just thinning it
When discharge from a specialist service is being discussed
A reassuring explanation for people with long-standing Aspergillus bronchitis
This page is for people who have lived for many years with a diagnosis of Aspergillus bronchitis, and who are now hearing that discharge from a specialist or tertiary service may be discussed, or is being gently considered.
Many patients tell us this brings up worries such as:
-
“Does this mean I’m less safe?”
-
“Does this mean they’re not sure anymore?”
-
“What if things get worse later?”
These feelings are very common, and they make sense.
First — nothing has been decided yet
If discharge is being discussed, it usually means:
-
Your team is reviewing your care carefully
-
They are looking at whether regular specialist follow-up is still helping right now
-
They are not withdrawing care, and not closing doors
Discussion is part of good medicine — especially with conditions that can change slowly over time.
Why might discharge even come up after many years?
This can feel surprising, but it is usually because:
-
Your condition has been stable for a long time
-
There has been no clear progression
-
Specialist treatments are not currently being changed
-
Ongoing follow-up may not be adding extra benefit at this stage
This is often a sign of relative stability, not doubt or disbelief.
Does this mean they think you never had Aspergillus bronchitis?
No — not at all.
What it usually means is:
-
Aspergillus bronchitis was a reasonable and helpful way to understand your symptoms at the time
-
Over time, the balance has shifted
-
Aspergillus may now be less active or less central to how you are feeling
Medical understanding evolves, and long-term conditions often change their shape rather than disappear or suddenly become “wrong”.
Does discharge mean Aspergillus is no longer important?
Not exactly.
It usually means:
“We don’t think Aspergillus is the main thing driving your symptoms right now.”
It does not mean:
“Aspergillus will never matter again.”
Your specialists know that Aspergillus-related problems can:
-
Fluctuate
-
Become more relevant during periods of illness or change
-
Need revisiting later on
That possibility is built into discharge planning, even if it is not always said clearly.
Why does this still feel unsettling?
Because specialist care often feels like a safety net.
You may have felt:
-
Known and understood by the team
-
Reassured by specialist oversight
-
Protected by regular review
Thinking about discharge can feel like losing that protection — even when nothing is actually changing day to day.
That emotional response is completely understandable.
What discharge from a specialist service usually does mean
If discharge does happen, it usually means:
-
Your care continues with your GP or respiratory team
-
Your history does not disappear
-
You are not starting from scratch
-
Re-referral is expected if things change
Specialist teams rarely intend discharge to be permanent or final.
What about being re-referred if things worsen?
This is one of the most important points — and a reassuring one.
In most cases:
-
Re-referral is anticipated
-
Patients previously known to the service are often reviewed more quickly
-
You do not need to “prove” everything again
Discharge usually comes with an open door, even if that door is not labelled as such.
What helps patients feel safer at this stage
It is reasonable to want:
-
A clear explanation of why discharge is being discussed
-
Reassurance that this is about now, not forever
-
Clarity about what would prompt a return
-
Confidence that your GP knows your history
These are normal needs — not demands.
What you might gently ask your team
You could ask:
-
“If my symptoms change, would re-referral be straightforward?”
-
“What sort of changes should prompt a review?”
-
“Will my GP have clear guidance from you?”
-
“Is discharge something we can review over time?”
These questions often help turn uncertainty into reassurance.
Key things to hold onto
-
Discussion of discharge usually reflects stability, not dismissal
-
It does not mean your past diagnosis was wrong
-
It does not mean you are being left unsupported
-
Re-referral is part of good planning, not failure
-
Feeling unsure or vulnerable at this point is very common
In gentle terms
Talking about discharge usually means “you are doing well enough not to need us right now” — not “you never needed us” and not “you’re on your own”.
⭐ Aspergillus Bronchitis: The Overlooked Condition Hiding in Plain Sight
Estimated prevalence 1–2% in bronchiectasis and chronic airway disease clinics.
Aspergillus Bronchitis (AB) is a chronic, symptomatic infection of the airways caused by Aspergillus species in people with underlying lung disease. It sits between simple colonisation and chronic pulmonary aspergillosis (CPA), and is frequently overlooked or mislabelled as “recurrent infection,” “post-viral symptoms,” or uncontrolled bronchiectasis.
Unlike CPA, Aspergillus Bronchitis does not require cavities or major structural destruction — which makes it both easier to miss and surprisingly common among people with chronic airway disease.
When recognised and treated (usually with antifungal therapy for several months), symptoms often improve significantly. But because awareness remains low, most patients cycle through unnecessary antibiotics, repeated exacerbations, and worsening airway disease before the real cause is identified.
⭐ What Exactly Is Aspergillus Bronchitis?
Aspergillus Bronchitis is defined by:
-
chronic productive cough
-
sputum growing Aspergillus species repeatedly
-
airway inflammation
-
symptoms lasting over 3 months
-
underlying airway disease (bronchiectasis, CF, COPD, prior TB, ABPA)
-
response to antifungal therapy
Unlike ABPA:
-
there is no allergic response,
-
IgE is usually normal,
-
eosinophils are normal or mildly elevated.
Unlike CPA:
-
there are no cavities on imaging,
-
IgG may be normal or only slightly elevated,
-
disease is confined to the airways, not lung tissue.
This places AB in a “grey zone” — often invisible unless specifically looked for.
⭐ Why Aspergillus Bronchitis Is Missed
1. Symptoms mimic common chronic airway disease
Typical AB symptoms include:
-
daily productive cough
-
worsening sputum thickness
-
breathlessness
-
fatigue
-
repeated “chest infections”
-
slow-to-clear mucus
-
crackles or wheeze
These resemble:
-
bronchiectasis exacerbations
-
COPD flare-ups
-
chronic infection with Pseudomonas or NTM
-
post-viral cough
-
uncontrolled asthma
Without fungal awareness, clinicians default to bacterial explanations.
2. Sputum grows multiple organisms — Aspergillus is dismissed
In bronchiectasis, sputum frequently grows:
-
Haemophilus
-
Pseudomonas
-
Staphylococcus
-
Streptococcus
-
NTM
When Aspergillus appears, it’s often labelled:
-
“colonisation”
-
“contaminant”
-
“not clinically relevant”
But repeated isolation with persistent symptoms is highly suggestive of AB.
3. IgE/IgG results may be normal
Many clinicians expect high IgE or IgG to “confirm Aspergillus disease.”
But in Aspergillus Bronchitis:
-
IgE is usually normal
-
IgG can be normal or borderline
This leads to false reassurance.
4. Radiology rarely shows overt features
CT scans in AB may show:
-
mucus plugging
-
mild bronchial wall thickening
-
small nodules
-
progression of bronchiectasis
But they do not show the cavities of CPA or classic features of ABPA.
Therefore radiologists often report scans as “no significant change” or “stable bronchiectasis.”
5. Antibiotics appear to help — temporarily
Patients often improve slightly with:
-
amoxicillin
-
doxycycline
-
macrolides
-
ciprofloxacin
This gives clinicians the impression of bacterial disease, but symptoms soon return.
6. Lack of awareness
Many specialists (even in respiratory clinics) are unaware that Aspergillus Bronchitis:
-
exists as a distinct clinical entity
-
can be disabling
-
responds to antifungals
-
predicts progression to CPA if untreated
This leads to significant diagnostic delay.
⭐ Who Is at Highest Risk?
1. Bronchiectasis
The largest risk group.
Aspergillus Bronchitis may account for 1–2% of all bronchiectasis patients, and up to 5–10% in severe or frequent exacerbator groups.
2. Cystic Fibrosis (CF)
These patients frequently grow Aspergillus but not all have ABPA — some have Aspergillus Bronchitis.
3. COPD and chronic productive cough
Especially those with:
-
frequent mucus plugging
-
repeated “infective exacerbations”
-
progressive sputum production
4. Post-TB airway damage
Chronic airway deformity, scarring, and bronchiectasis from old TB predispose to fungal infection.
5. Post-COVID structural disease
A new and growing risk group, especially after prolonged ICU ventilation.
6. ABPA patients
Some patients develop Aspergillus Bronchitis during steroid-dominated treatment or after stopping antifungals.
⭐ Which Specialities Need Greater Awareness?
-
Respiratory medicine
(especially bronchiectasis clinicians and severe asthma teams) -
Infectious Diseases
(frequent respiratory presentations with chronic airway infection) -
Radiology
(to recognise subtle but progressive airway changes) -
Primary care
(“recurrent chest infection” or “persistent cough” patients) -
Physiotherapy & airway clearance teams
(excessive sputum with fungal elements) -
Cystic Fibrosis services
The National Aspergillosis Centre is the ideal referral destination when diagnosis is uncertain or symptoms persist despite typical management.
⭐ Red Flags Suggesting Aspergillus Bronchitis
1. Chronic (>3 months) productive cough + repeated Aspergillus in sputum
Even 2 positive sputums in the right clinical context should raise suspicion.
2. Bronchiectasis patient not improving on repeated antibiotics
3. Thick, tenacious mucus with black, grey, or brown plugs
4. Worsening CT bronchiectasis or mucus plugging
5. Absence of features typical for ABPA (normal IgE, no fleeting infiltrates)
6. Asthma or COPD patient with new persistent sputum
7. Partial response to antibiotics but rapid relapse
8. Unexplained fatigue and breathlessness in someone with airway disease
⭐ The Cost of Missed Aspergillus Bronchitis
If AB is not recognised early, consequences include:
-
repeated exacerbations
-
accelerating bronchiectasis
-
long-term airway damage
-
chronic inflammation
-
steroid overuse
-
unnecessary antibiotics
-
repeated hospitalisations
-
progression to CPA in some patients
For health systems, missed diagnosis leads to:
-
higher admission rates
-
inappropriate long-term antibiotic use
-
avoidable CT scans and investigations
-
greater long-term burden of CPA
But antifungal therapy — when appropriately used — can offer marked symptom improvement and reduce exacerbation frequency.
⭐ Conclusion
Aspergillus Bronchitis is a distinct, treatable form of chronic airway disease seen in people with bronchiectasis, asthma, COPD, CF, and post-TB lung damage. Yet lack of awareness means many patients are repeatedly misdiagnosed with bacterial infections or unexplained chronic cough.
Recognising red flags, reviewing sputum results carefully, and considering antifungal therapy can dramatically improve outcomes. Early referral to specialist centres such as the National Aspergillosis Centre is recommended for complex cases or uncertain diagnosis.
Early identification prevents airway deterioration — and reduces the likelihood of progression to CPA.
🟣 Aspergillus Bronchitis: A Patient Guide
If you've been diagnosed with bronchiectasis, COPD, or other chronic lung problems, and keep getting infections or mucus that tests positive for Aspergillus, you might be told you have Aspergillus bronchitis. But what does that mean? And how is it different from ABPA (Allergic Bronchopulmonary Aspergillosis)?
This guide explains what Aspergillus bronchitis is, how it’s diagnosed, how common it is, and how it differs from ABPA.
🌬️ What Is Aspergillus Bronchitis?
Aspergillus bronchitis is a chronic fungal infection of the airways caused by Aspergillus fumigatus. It happens in people with damaged or scarred airways, such as:
-
Bronchiectasis
-
COPD
-
Cystic fibrosis
-
Occasionally asthma (if structural damage exists)
It’s a low-grade infection, not an allergy and not an invasive disease. The fungus lives in the mucus lining the airways, causing persistent inflammation, infection, and symptoms.
🔍 What Are the Symptoms?
| Common symptoms | What to know |
|---|---|
| Persistent cough | Often brings up sticky or brown mucus |
| Worsening breathlessness | Not always explained by asthma or infection alone |
| Fatigue or low energy | Common in chronic fungal infections |
| Frequent infections | May keep returning despite antibiotics |
| Wheeze | Sometimes, especially in asthma or ABPA overlap |
| Weight loss or low-grade fever | Possible in long-standing infection |
📊 How Common Is It?
Aspergillus bronchitis is underdiagnosed but increasingly recognised — especially in patients referred to specialist centres.
Estimated frequency in different conditions:
| Underlying condition | Estimated rate of Aspergillus bronchitis |
|---|---|
| Bronchiectasis | ~5–15%, higher in referral centres |
| COPD | ~1–5%, especially with steroid use |
| Cystic fibrosis | 5–10% (non-allergic cases) |
| Asthma (alone) | Rare unless bronchiectasis is also present |
It may be mistaken for a flare-up or chronic bacterial infection. Many people are told it’s “just colonisation” — but if symptoms persist and Aspergillus keeps growing in sputum, Aspergillus bronchitis should be considered.
🧪 How Is It Diagnosed?
There’s no single test. Diagnosis is based on clinical features plus evidence of fungal growth and a non-allergic immune pattern.
| Test | What it shows |
|---|---|
| Sputum culture / PCR | Repeated detection of Aspergillus |
| Aspergillus IgG (blood) | Often raised, shows chronic exposure |
| Aspergillus IgE & eosinophils | Usually normal — helps exclude ABPA |
| CT scan | Shows bronchiectasis, mucus plugging, but no cavitation |
| Response to antifungals | Clinical improvement supports diagnosis |
🔄 How Is It Different from ABPA?
ABPA is an allergic reaction to Aspergillus that mainly affects people with asthma or cystic fibrosis.
Aspergillus bronchitis, on the other hand, is a fungal infection in damaged airways, not an allergy.
| Feature | Aspergillus Bronchitis | ABPA |
|---|---|---|
| Type of disease | Chronic fungal infection | Allergic lung disease |
| Immune markers | High IgG, normal IgE | High IgE and eosinophils |
| Sputum | Repeated growth of Aspergillus | May or may not grow |
| CT findings | Bronchiectasis, mucus | Central bronchiectasis, mucus plugs |
| Treatment | Antifungals only | Steroids ± antifungals |
| Typical patient | Bronchiectasis, COPD | Asthma (often severe), sometimes CF |
Some patients can have both conditions at once, especially those with asthma and bronchiectasis — so testing is important.
💊 Treatment Options
| Treatment | Purpose |
|---|---|
| Oral antifungals (itraconazole, voriconazole) | Main treatment — often for several months |
| Inhaled antifungals (e.g. nebulised amphotericin) | Alternative if oral drugs not tolerated |
| Airway clearance physiotherapy | Helps remove mucus and fungal load |
| Regular sputum testing | To monitor treatment response |
| Steroids | Not used unless there’s overlapping ABPA or asthma |
🧠 Summary
| Question | Answer |
|---|---|
| Is it an infection? | ✅ Yes — fungal infection in the airways |
| Is it an allergy? | ❌ No — that’s ABPA |
| Can it coexist with ABPA? | ✅ Yes, in some cases |
| How is it diagnosed? | Repeated Aspergillus in sputum + high IgG + symptoms |
| How is it treated? | Antifungal medication (oral or nebulised) |
| Will it go away? | Often improves with treatment, but monitoring is essential |
💬 What to Ask Your Doctor
-
Could my symptoms be from Aspergillus bronchitis?
-
Have I had sputum cultures and Aspergillus blood tests (IgG, IgE)?
-
Would antifungal treatment help me?
-
Should I be referred to a specialist centre (e.g. for CPA, ABPA, bronchiectasis)?
-
Am I on the best airway clearance and physiotherapy plan?
Aspergillus Tracheobronchitis (ATB) for Expert patients and non-Specialist Clinicians
Expert Information for Patients, GPs, and Specialist Nurses
🔎 What Is Aspergillus Tracheobronchitis?
Aspergillus tracheobronchitis (ATB) is a rare but serious form of airway-invasive aspergillosis that primarily affects the trachea and large bronchi, rather than the lung parenchyma. It occurs predominantly in immunocompromised patients and may present with obstructive airway symptoms or respiratory failure.
ATB can exist on a spectrum from superficial colonisation to ulcerative or pseudomembranous invasion of the bronchial wall.
🧬 Pathophysiology
-
Inhaled Aspergillus spores adhere to and invade damaged airway mucosa.
-
Occurs more commonly when local airway immunity is impaired (e.g. in transplant recipients or critical illness).
-
May co-exist with invasive pulmonary aspergillosis (IPA) or appear in isolation.
👥 Who Is at Risk?
High-risk groups include:
-
Lung transplant recipients
-
Hematopoietic stem cell transplant patients
-
Severe COPD or structural airway disease
-
Patients with prolonged corticosteroid use
-
Critically ill or mechanically ventilated patients
-
COVID-19 or influenza patients (sometimes overlapping with CAPA/IAPA)
⚠️ Clinical Presentation
Symptoms depend on the degree of airway obstruction and depth of invasion:
-
Cough (dry or productive)
-
Worsening breathlessness
-
Stridor or wheeze
-
Hoarseness or vocal changes
-
Fever unresponsive to antibiotics
-
Haemoptysis (may be life-threatening)
-
Airway obstruction or collapse in advanced cases
ATB may be mistaken for tracheobronchial malignancy, infection, or stenosis.
🧪 Diagnosis
Bronchoscopy is essential for diagnosis:
-
Direct visualisation of:
-
Ulceration
-
Pseudomembranes
-
Plaques
-
Necrotic debris
-
-
Biopsies may reveal fungal hyphae invading mucosa.
Microbiological Investigations:
-
Culture and PCR for Aspergillus from BAL or brushings
-
BAL galactomannan
-
Serum galactomannan or β-D-glucan may be supportive
-
CT chest may be normal or show airway thickening, bronchial wall invasion, or tree-in-bud opacities
💊 Treatment
Systemic Antifungals:
-
Voriconazole is first-line
-
Isavuconazole or liposomal amphotericin B if azole intolerant or resistant
Airway Management:
-
Debridement or bronchoscopic removal of pseudomembranes in severe obstruction
-
Airway stenting in refractory strictures
-
Nebulised antifungals (e.g. amphotericin B) may be used as adjunct in selected cases
Prompt initiation of antifungal therapy is vital. Delays can lead to respiratory failure or death.
🧾 Monitoring
-
Clinical response: breathlessness, cough, fever
-
Repeat bronchoscopy in some cases
-
CT imaging of airways
-
Antifungal drug levels
-
Liver and renal function
📚 More Information
-
ATB is under-recognised, especially in non-neutropenic or critically ill patients.
-
Should be considered in transplant recipients or ICU patients with persistent respiratory symptoms and negative bacterial cultures.
-
Referral to respiratory, infectious diseases, and ICU teams is essential.
-
Resources: aspergillosis.org ; BTS Statement on aspergillosis