Genes and aspergillosis: why the same fungus causes different problems in different people
Why look at genes when talking about aspergillosis?
The theme of World Aspergillus Day 2026 was “How can the genomics revolution help patients with chronic aspergillosis?”
To answer that, we need to look briefly at genes and what they tell us about how the body resists infection.
Genes are the body’s instruction manual. They help control how our immune system works, how inflammation is managed, and how well we clear infections. Humans have around 25,000 genes, with two copies of each in almost every cell — and billions of cells using these instructions every day.
Small, natural differences in genes help explain why people respond differently to Aspergillus: some develop allergy, others chronic infection, and many clear it without any illness at all. Genes don’t determine outcomes, but they help us understand why the immune response differs between people.
Many people ask an understandable question:
“If we all breathe in Aspergillus spores, why do only some people get aspergillosis – and why does it look so different from person to person?”
Part of the answer lies in genes.
Genes do not cause aspergillosis on their own, but they can influence how the immune system responds once the fungus is encountered.
A simple way to think about genes
Genes act like settings, not switches.
They can influence:
-
how strongly your immune system reacts
-
whether that reaction is allergic, chronic, or weak
-
how well fungi are cleared from the lungs
Genes do not override:
-
lung damage (asthma, bronchiectasis, old infections)
-
steroid or immunosuppressive treatment
-
mould exposure levels
They help explain patterns of illness, not certainty.
Risk stacking: why combinations matter more than any single factor
Aspergillosis rarely develops because of one single cause. Instead, it usually arises through risk stacking, where several small risk factors overlap at the same time.
Each factor may add only a little vulnerability on its own, but together they can tip the balance from resistance to disease.
This helps explain why aspergillosis often appears after years of stability, or during periods of change such as illness, medication adjustment, or increased environmental exposure.
What does risk stacking look like in practice?
A person might have:
-
mild genetic tendencies toward allergic inflammation or reduced fungal clearance
-
asthma, bronchiectasis, or old lung damage
-
long-term inhaled or oral corticosteroid treatment
-
periods of higher mould exposure (for example, damp housing or renovation work)
None of these alone guarantees illness.
But stacked together, they increase the chance that Aspergillus:
-
is recognised as an allergen rather than ignored
-
is not cleared efficiently from the lungs
-
triggers ongoing inflammation or chronic infection
Where genes fit into risk stacking
Genes usually act as background modifiers, not primary causes.
In people with healthy lungs and normal immunity, genetic differences rarely matter.
In people who already have lung disease, immune suppression, or repeated exposure, those same genetic differences can add to the overall risk stack.
This also explains why there is no single genetic test that can predict aspergillosis — risk depends on combinations, not on one gene.
Just as risks can add up, risk reduction also adds up. Improvements in airway clearance, asthma control, steroid management, and home environment can all meaningfully reduce overall risk.
Why this matters in aspergillosis
Aspergillosis is not one condition. It includes:
-
fungal sensitisation and allergy
-
chronic pulmonary infection
-
invasive disease in people with weakened immunity
Different genes influence different stages of the immune response, which helps explain why people experience very different forms of disease.
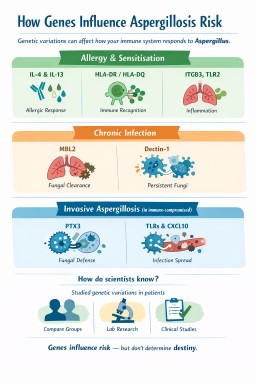
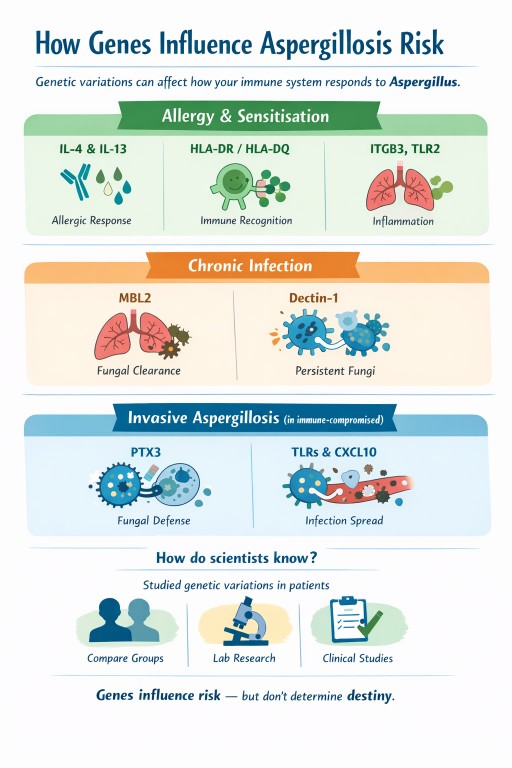
1. Genes linked to fungal allergy and sensitisation
These genes affect whether the immune system treats Aspergillus as a strong allergen.
IL-4, IL-13 and the IL-4 receptor
What they do
Control allergic inflammation, including:
-
immunoglobulin E (IgE)
-
eosinophils
-
mucus production
-
airway inflammation
What this means
Certain natural gene variants increase the likelihood of:
-
fungal sensitisation
-
asthma with fungal sensitisation
-
allergic bronchopulmonary aspergillosis (ABPA)
This fits closely with what patients experience clinically: high IgE, eosinophilia, steroid responsiveness, and response to biologic treatments.
HLA-DR and HLA-DQ
What they do
Help the immune system decide which proteins deserve attention.
What this means
Some HLA types present Aspergillus proteins in a way that:
-
encourages persistent allergic inflammation
-
increases the chance of ABPA
This helps explain why only a minority of people with asthma develop ABPA.
ITGB3 (integrin beta-3)
What it does
Helps airway and immune cells:
-
attach to surrounding tissue
-
communicate danger signals
-
interact with fungal-recognition pathways
What this means
Certain versions are linked to:
-
mould sensitisation
-
stronger immune signalling when fungal particles are present
This does not mean ITGB3 causes aspergillosis.
It helps explain why some people become sensitised more easily.
TLR2
What it does
Recognises fungal cell-wall components and triggers early immune responses.
What this means
Different versions can amplify or dampen inflammation, influencing sensitivity to fungi.
2. Genes linked to chronic pulmonary aspergillosis (CPA)
These genes influence how well fungi are cleared, especially in damaged lungs.
MBL2 (mannose-binding lectin)
What it does
Marks fungi so the immune system can remove them.
What this means
Low MBL activity may allow Aspergillus to persist once lung cavities or scarring exist.
Dectin-1 (CLEC7A)
What it does
Detects fungal cell-wall sugars and triggers antifungal responses.
What this means
Reduced detection can allow slow, long-term infection rather than allergy.
TLR4
What it does
Regulates inflammation in response to microbes.
What this means
Certain variants may influence how chronic inflammation and tissue damage evolve.
3. Genes linked to invasive aspergillosis
These matter most in people with weakened immune systems (for example, during chemotherapy or after transplant).
PTX3 (pentraxin-3)
What it does
Acts as an early fungal sensor and helps immune cells kill Aspergillus.
What this means
Reduced PTX3 activity is one of the strongest known genetic risk factors for invasive aspergillosis in high-risk medical settings.
TLR3 and interferon pathways (including CXCL10)
What they do
Coordinate immune communication and antifungal killing.
What this means
Impairment can delay fungal control and increase the risk of spread.
How do scientists know these genes are involved?
Researchers study natural genetic variations that:
-
are common in healthy people
-
are present from birth
-
usually cause small functional differences, not disease by themselves
They:
-
compare people with aspergillosis to similar people without it
-
identify gene variants linked to specific disease patterns
-
test how those genes affect fungal recognition, inflammation, or killing
-
confirm findings in laboratory and clinical studies
These are risk modifiers, not disease-causing genes.
Does this mean my family is at risk?
This is a very common concern. The reassuring answer for most people is:
No – aspergillosis does not usually run in families.
Why this is reassuring
-
These gene variants are common in the general population
-
Most people who carry them never develop aspergillosis
-
Aspergillosis requires other factors, such as lung disease, immune suppression, or heavy exposure
-
There is no consistent pattern of aspergillosis being passed from parent to child
Even strong genetic signals (such as PTX3) only increase risk in specific high-risk medical situations, not in healthy relatives.
Putting it all together
| Pattern of disease | Genes most often involved |
|---|---|
| Fungal sensitisation | IL-4, IL-13, IL-4 receptor, ITGB3, TLR2 |
| ABPA | IL-4/IL-13 pathway, HLA-DR/DQ, TLR3 |
| Chronic pulmonary aspergillosis | MBL2, Dectin-1, TLR4 |
| Invasive aspergillosis | PTX3, interferon pathways |
What this means for patients and families
-
Genetic testing is not routinely needed
-
These genes do not predict individual outcomes
-
Family members are not usually at increased risk
The most important factors remain:
-
good lung care
-
appropriate treatment
-
sensible mould exposure reduction
Genes influence risk — they do not determine destiny.