How gut and lung microbiota are transforming the way we diagnose, treat and understand infection in aspergillosis

For decades, infection was seen through a simple lens:
Find the organism → treat the organism → infection cured.
But modern microbiome research has shown that this view is too narrow—especially for chronic lung diseases such as aspergillosis, bronchiectasis, ABPA, SAFS and CPA.
We now understand that the:
-
lungs,
-
gut,
-
sinuses,
-
skin, and even
-
CPA cavities
contain complex microbial ecosystems (bacteria, fungi, viruses, archaea) that interact dynamically with each other and with your immune system.
Rather than being passive passengers, these microbes shape inflammation, immunity, symptoms, resistance, treatment response and overall wellbeing.
This is why microbiome science is truly revolutionising how clinicians think about infection.
🧬 1. What is a microbiome?
A microbiome is the entire community of microorganisms living in a particular environment, plus all the genes, chemicals, signals and interactions that exist between them.
Healthy microbiomes are:
-
diverse
-
stable
-
environmentally balanced
-
dominated by harmless or beneficial species
Disease-associated microbiomes are:
-
less diverse
-
unstable
-
dominated by a few harmful organisms
-
deeply involved in inflammation
This imbalance is called dysbiosis.
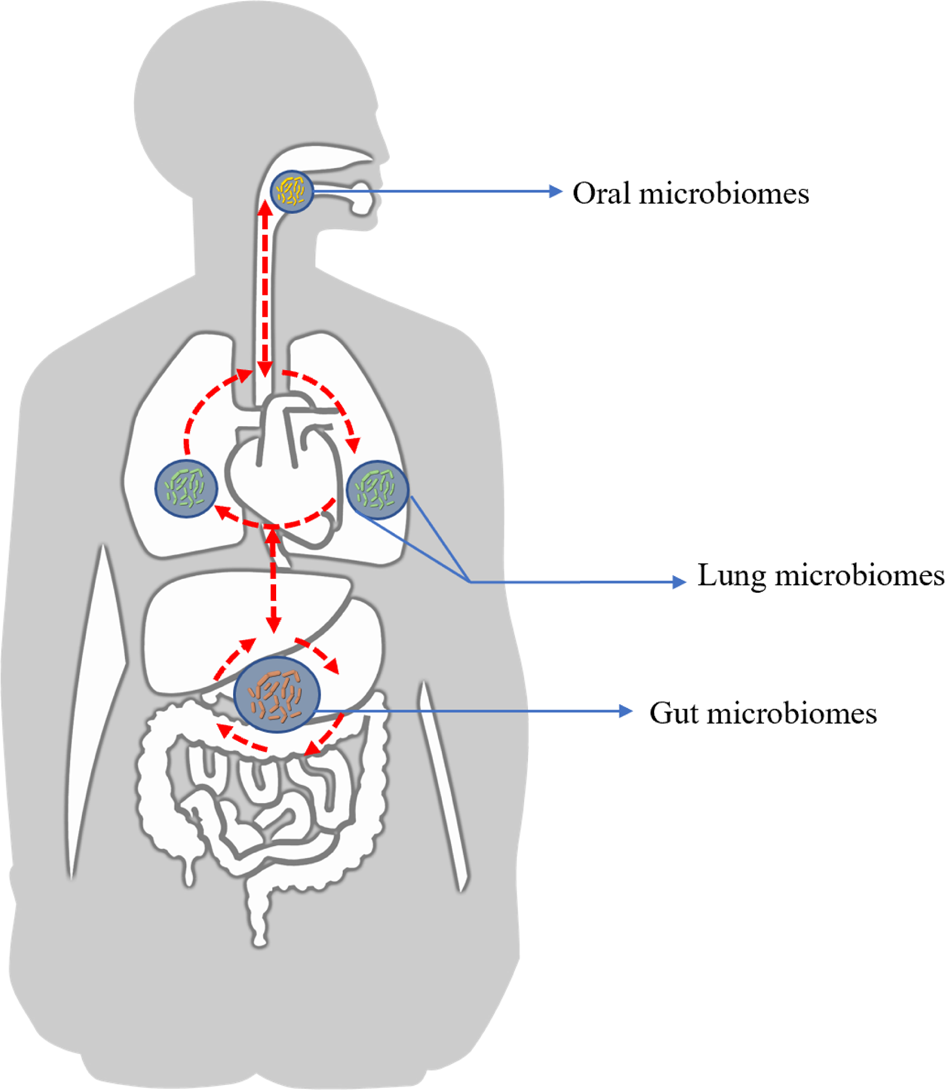
🫁 2. The lung microbiome: complex, dynamic, and vital
The lungs are not sterile—they contain a delicate, low-density microbiome.
In health, microbes drift in and out through:
-
breathing
-
micro-aspiration
-
mucociliary clearance
The “healthy lung microbiota” remains balanced because airflow and immune regulation prevent any single species from dominating.
In disease (aspergillosis, bronchiectasis, ABPA, SAFS, CPA), the situation changes:
-
thick mucus traps microbes
-
reduced airflow produces stagnant zones
-
inflammation increases microbial stickiness
-
biofilms form
-
pathogens dominate
-
microbial diversity drops
-
chronic inflammation becomes self-sustaining
This drives persistent symptoms even when cultures appear negative.
🍽️ 3. The gut microbiome: our “second immune system”

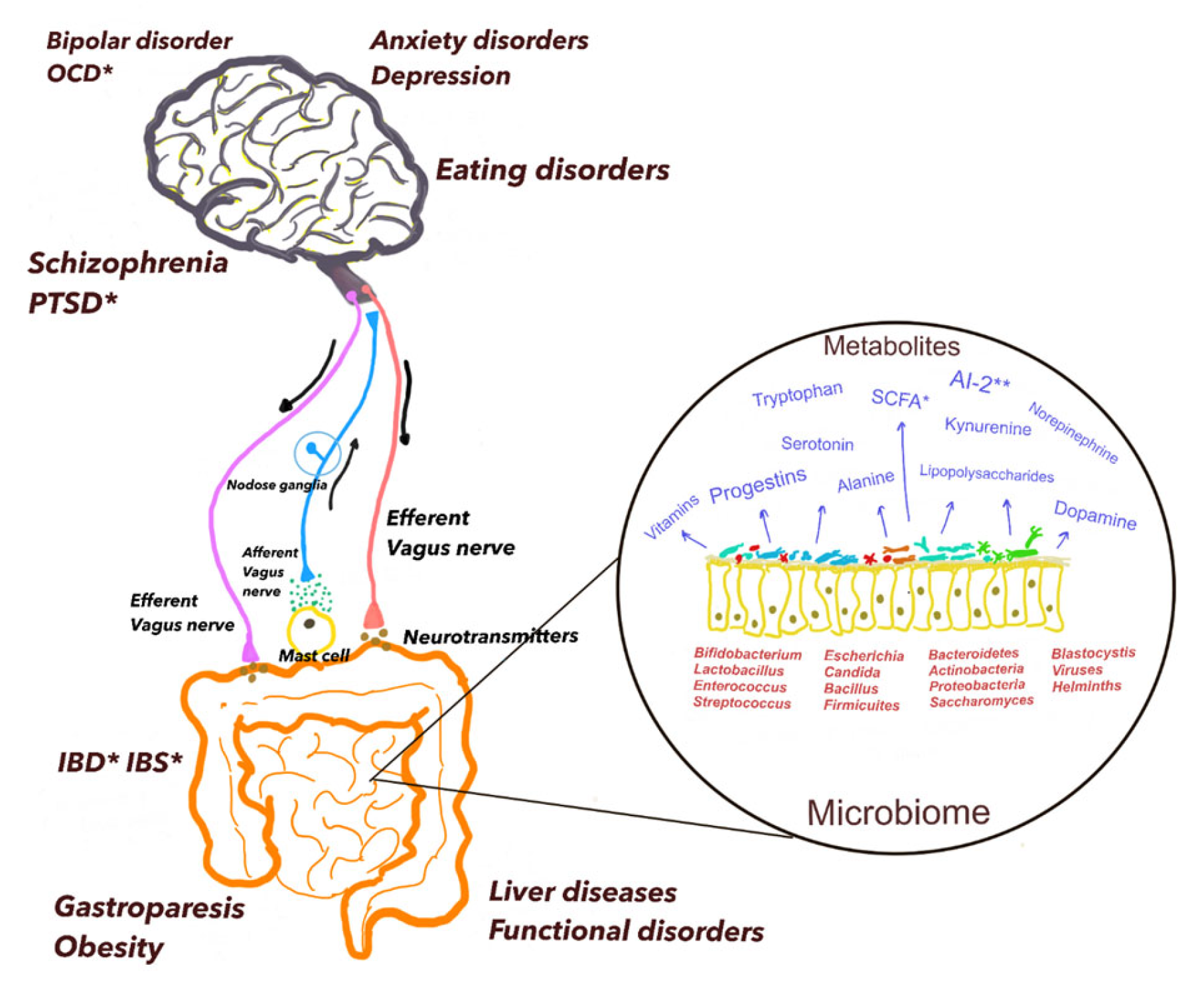
The gut contains trillions of bacteria, fungi and viruses.
Far from being limited to digestion, the gut microbiome influences:
-
immune development
-
inflammation control
-
IgE responses
-
eosinophils
-
energy levels
-
weight regulation
-
steroid responsiveness
-
susceptibility to infection
-
mental wellbeing
Up to 70% of your immune system is shaped by gut microbes.
This means:
Gut health directly affects lung health, including risk and severity of aspergillosis-related disease.
🔄 4. The Gut–Lung Axis: how the two microbiomes talk to each other
The gut and lungs are connected through a biochemical “highway” known as the gut–lung axis.
How the gut affects the lungs
Gut bacteria produce metabolites such as:
-
short-chain fatty acids (SCFAs) — e.g., butyrate
-
tryptophan metabolites
-
bile-acid derivatives
These travel in the bloodstream and regulate:
-
airway inflammation
-
Th2/Th17 immune responses
-
IgE and eosinophils
-
neutrophil activity
-
mucus production
-
tolerance to allergens (including fungal allergens)
-
steroid responsiveness
Low SCFA levels are linked to:
-
more severe asthma
-
worse fungal sensitisation
-
increased ABPA flares
-
poorer lung function
-
difficulty clearing infection
How lung disease affects the gut
Chronic respiratory disease increases:
-
gut permeability (“leaky gut”)
-
microbiota disruption
-
systemic inflammation
-
digestive symptoms
-
fatigue
-
candida overgrowth
Steroids and antibiotics worsen this further.
This creates a self-reinforcing circle of inflammation and dysbiosis.
🧱 5. Biofilms: microbial fortresses driving persistent disease
Biofilms are communities of microbes encased in a sticky protective matrix.
In aspergillosis and bronchiectasis, biofilms:
-
make organisms up to 100–1000× more resistant to treatment
-
protect microbes from the immune system
-
allow bacteria and fungi to communicate and collaborate
-
swap resistance genes
-
support mixed infections (e.g., Aspergillus + Pseudomonas)
-
cause chronic symptoms even with “negative” cultures
Biofilms also change the immune system’s behaviour, driving long-term inflammation.
🔬 6. What microbiome research has revealed so far
A. Infection is rarely a single organism
Microbiomes show that infections are polymicrobial ecosystems, not isolated pathogens.
B. Diversity = resilience
Higher microbial diversity is linked to:
-
better lung stability
-
fewer flare-ups
-
lower inflammation
Low diversity correlates with:
-
severe disease
-
CPA progression
-
ABPA flares
-
worse bronchiectasis outcomes
C. Microbiome patterns can predict future illness
Research shows that flare-ups often follow:
-
a drop in diversity
-
an increase in dominant pathogens
-
changes in fungal–bacterial interactions
D. Treatment responses are microbiome-dependent
The presence of certain bacteria can make Aspergillus:
-
grow faster
-
form stronger biofilms
-
resist antifungals
-
provoke more inflammation
🫁🌱 7. What patients can do to support their lung microbiota
Just as dietary fibre supports gut microbes, there are practical steps that support a healthier lung microbial ecosystem.
These steps do not introduce microbes into the lungs; instead, they improve the environment the microbiota lives in.
⭐ 1. Keep airways clear — the foundation of lung microbial health
Biofilms and harmful microbes thrive in stagnant mucus.
Effective clearance techniques:
-
Active Cycle of Breathing Techniques (ACBT)
-
Autogenic drainage
-
Oscillating devices (Flutter, Acapella, Aerobika)
-
Nebulised saline (3–7%)
-
Huffing and controlled coughing
Clearer airways → more airflow → better microbial balance.
⭐ 2. Hydration
Hydration thins mucus, improves ciliary function, and weakens biofilms.
⭐ 3. Use inhalers correctly & control inflammation
Inflamed, narrowed airways promote dysbiosis.
Good control of:
-
asthma
-
ABPA
-
eosinophilia
reduces microbial imbalance.
⭐ 4. Improve sinus health
The sinuses drip microbes into the lungs all day long.
Sinus care (saline rinses, nasal steroids) supports lung microbiota stability.
⭐ 5. Avoid unnecessary antibiotics
Antibiotics disrupt:
-
lung microbiota
-
gut microbiota
-
fungal–bacterial balance
-
biofilm behaviour
Use them when needed — but avoid repeated unnecessary courses.
⭐ 6. Exercise
Exercise increases airflow and clearance, helping shift the lung microbiome toward a healthier, more diverse state.
⭐ 7. Reduce smoke and indoor pollutants
Pollutants:
-
paralyse cilia
-
thicken mucus
-
promote pathogenic microbes
-
reduce diversity
HEPA filtration, ventilation, and smoke avoidance all help.
⭐ 8. Manage reflux (GORD)
Micro-aspiration introduces stomach contents into the lungs, disrupting the lung microbiota.
Treating reflux supports lung microbial homeostasis.
⭐ 9. Support your gut microbiota
A healthy gut → more SCFAs → improved lung immunity → a more balanced lung microbiome.
Helpful for gut health:
-
fibre-rich foods
-
diverse diet
-
fermented foods (if tolerated)
-
avoiding unnecessary antibiotics
-
reducing alcohol
-
managing stress
🩺 8. What this means for the future of aspergillosis care
Within 5–10 years, we may routinely use:
-
microbiome sequencing in clinic
-
AI-designed “ecosystem maps” of the lungs
-
targeted therapies for mixed infections
-
inhaled agents that break down biofilms
-
gut-directed therapies to help lung disease
-
personalised airway clearance plans
-
microbial diversity scores to predict flares
This could:
-
reduce exacerbations
-
minimise antibiotic and antifungal exposure
-
improve quality of life
-
slow CPA progression
-
improve steroid responsiveness
-
reduce hospital admissions
🧠 9. Key takeaways
-
You have two important microbiomes that matter for aspergillosis:
the lung microbiome and the gut microbiome. -
They communicate through the gut–lung axis.
-
Dysbiosis (imbalance) increases inflammation and worsens fungal disease.
-
Biofilms make infections far more resistant and persistent.
-
Patients can support their lung microbiota through lifestyle steps, especially:
-
airway clearance
-
hydration
-
exercise
-
sinus care
-
avoiding unnecessary antibiotics
-
supporting gut health
-
-
Microbiome science is transforming future diagnosis and treatment strategies.
Share this post
Latest News posts
News archive
- ABPA
- Air Quality
- Airway Clearance, Diagnosis & Physiotherapy
- Antifungals
- Aspergilloma
- Aspergillus Bronchitis
- Biologics
- Blood Tests
- CPA
- Carers & Family
- Communities
- Complementary & Supplements
- Complications
- Conditions
- Diagnostics
- Environment
- Events & Recordings
- GP Guidance
- General interest
- Housing & Damp
- Imaging
- Immune System
- Lifestyle & Coping
- Living with Aspergillosis
- Mental Health
- Monitoring
- Monitoring & Safety
- NAC & Guidance
- NAC Announcements
- Other
- Other Forms Aspergillosis
- Patient Research
- Pets & Animals
- Professional Guidance
- Recordings
- Research
- Research Summaries
- SAFS / Severe Asthma
- Side Effects
- Specialists
- Steroids
- Symptoms
- Travel and Insurance
- Treatment
- Vaccines
- Weekly Updates

